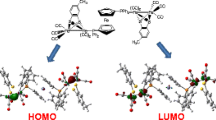
Abstract.
A key step in the biological nitrogen fixation problem is the transfer of protons and electrons onto inert molecular nitrogen. A first intermediate will then be diazene (diimide), N2H2, which is thermodynamically unstable with respect to dissociation into N2 and H2 in the gas phase. Thus, diazene must be stabilized such that the reduction of a complex binding an activated nitrogen becomes energetically feasible. A considerable contribution to this stabilization has been attributed to hydrogen bonds of the type N–H···S. We investigate the strength of these hydrogen bonds in two model compounds. Since the contribution of an intramolecular hydrogen bridge to the total binding energy of a molecule is not a well-defined concept, it is necessary to define a suitable descriptor for this quantity. We present a new approach of estimating hydrogen-bond energies from two-center shared-electron numbers obtained from density functional calculations. Our approach is particularly designed for highly complex systems such as transition-metal complexes with large coordination spheres.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 28 March 2001 / Accepted: 13 June 2001 / Published online: 11 October 2001
Rights and permissions
About this article
Cite this article
Reiher, M., Sellmann, D. & Hess, B. Stabilization of diazene in Fe(II)–sulfur model complexes relevant for nitrogenase activity. I. A new approach to the evaluation of intramolecular hydrogen bond energies. Theor Chem Acc 106, 379–392 (2001). https://doi.org/10.1007/s002140100287
Issue Date:
DOI: https://doi.org/10.1007/s002140100287