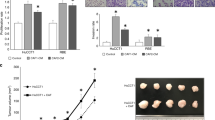
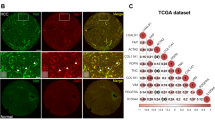
Abstract
Transforming growth factor (TGF)-β stimulates extracellular matrix (ECM) deposition during development of liver fibrosis and cirrhosis, the most important risk factor for the onset of hepatocellular carcinoma. In liver cancer, TGF-β is responsible for a more aggressive and invasive phenotype, orchestrating remodeling of the tumor microenvironment and triggering epithelial–mesenchymal transition of cancer cells. This is the scientific rationale for targeting the TGF-β pathway via a small molecule, galunisertib (intracellular inhibitor of ALK5) in clinical trials to treat liver cancer patients at an advanced disease stage. In this study, the hypothesis that galunisertib modifies the tissue microenvironment via inhibition of the TGF-β pathway is tested in an experimental preclinical model. At the age of 6 months, Abcb4ko mice—a well-established model for chronic liver disease development and progression—are treated twice daily with galunisertib (150 mg/kg) via oral gavage for 14 consecutive days. Two days after the last treatment, blood plasma and livers are harvested for further assessment, including fibrosis scoring and ECM components. The reduction of Smad2 phosphorylation in both parenchymal and non-parenchymal liver cells following galunisertib administration confirms the treatment effectiveness. Damage-related galunisertib does not change cell proliferation, macrophage numbers and leucocyte recruitment. Furthermore, no clear impact on the amount of fibrosis is evident, as documented by PicroSirius red and Gomori-trichome scoring. On the other hand, several fibrogenic genes, e.g., collagens (Col1α1 and Col1α2), Tgf-β1 and Timp1, mRNA levels are significantly downregulated by galunisertib administration when compared to controls. Most interestingly, ECM/stromal components, fibronectin and laminin-332, as well as the carcinogenic β-catenin pathway, are remarkably reduced by galunisertib-treated Abcb5ko mice. In conclusion, TGF-β inhibition by galunisertib interferes, to some extent, with chronic liver progression, not by reducing the stage of liver fibrosis as measured by different scoring systems, but rather by modulating the biochemical composition of the deposited ECM, likely affecting the fate of non-parenchymal cells.





Similar content being viewed by others
References
Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2007) Molecular biology of the cell. Garland Science, London
An SS, Kim J, Ahn K, Trepat X, Drake KJ, Kumar S et al (2009) Cell stiffness, contractile stress and the role of extracellular matrix. Biochem Biophys Res Commun 382(4):697–703. https://doi.org/10.1016/j.bbrc.2009.03.118
Bedossa P, Poynard T (1996) An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology 24(2):289–293. https://doi.org/10.1002/hep.510240201
Bergamini C, Sgarra C, Trerotoli P, Lupo L, Azzariti A, Antonaci S et al (2007) Laminin-5 stimulates hepatocellular carcinoma growth through a different function of alpha6beta4 and alpha3beta1 integrins. Hepatology 46(6):1801–1809. https://doi.org/10.1002/hep.21936
Bissell DM (2001) Exp Mol Med 33(4):179–190. (Review)
Cao Y, Agarwal R, Dituri F, Lupo L, Trerotoli P, Mancarella S et al (2017) NGS-based transcriptome profiling reveals biomarkers for companion diagnostics of the TGF-β receptor blocker galunisertib in HCC. Cell Death Dis 8(2):e2634. https://doi.org/10.1038/cddis.2017.44
Carraher CL, Schwarzbauer JE (2013) Regulation of matrix assembly through rigidity-dependent fibronectin conformational changes. J Biol Chem 288(21):14805–148014. https://doi.org/10.1074/jbc.M112.435271
Cho JJ, Hocher B, Herbst H, Jia JD, Ruehl M, Hahn EG et al (2000) An oral endothelin-A receptor antagonist blocks collagen synthesis and deposition in advanced rat liver fibrosis. Gastroenterology 118(6):1169–1178
de Gouville AC, Boullay V, Krysa G, Pilot J, Brusq JM, Loriolle F et al (2005) Inhibition of TGF-beta signaling by an ALK5 inhibitor protects rats from dimethylnitrosamine-induced liver fibrosis. Br J Pharmacol 145(2):166–177. https://doi.org/10.1038/sj.bjp.0706172
Dituri F, Mazzocca A, Fernando J, Papappicco P, Fabregat I, De Santis F et al (2013) Differential inhibition of the TGF-β signaling pathway in HCC cells using the small molecule inhibitor LY2157299 and the D10 monoclonal antibody against TGF-β receptor type II. PLoS One 8(6):e67109. https://doi.org/10.1371/journal.pone.0067109
Dooley S, Ten Dijke P (2012) TGF-β in progression of liver disease. Cell Tissue Res 347(1):245–256. https://doi.org/10.1007/s00441-011-1246-y (Review)
Fickert P, Fuchsbichler A, Wagner M, Zollner G, Kaser A, Tilg H et al (2004) Regurgitation of bile acids from leaky bile ducts causes sclerosing cholangitis in Mdr2 (Abcb4) knockout mice. Gastroenterology 127(1):261–274. https://doi.org/10.1053/j.gastro.2004.04.009
Franco DL, Mainez J, Vega S, Sancho P, Murillo MM, de Frutos CA et al (2010) Snail1 suppresses TGF-beta-induced apoptosis and is sufficient to trigger EMT in hepatocytes. J Cell Sci 123(Pt 20):3467–3477. https://doi.org/10.1242/jcs.068692
Fransvea E, Mazzocca A, Antonaci S, Giannelli G (2009) Targeting transforming growth factor (TGF)-βRI inhibits activation of beta1 integrin and blocks vascular invasion in hepatocellular carcinoma. Hepatology 49(3):839–850. https://doi.org/10.1002/hep.22731
George J, Roulot D, Koteliansky VE, Bissell DM (1999) In vivo inhibition of rat stellate cell activation by soluble transforming growth factor beta type II receptor: a potential new therapy for hepatic fibrosis. Proc Natl Acad Sci USA 96(22):12719–12724. https://doi.org/10.1073/pnas.96.22.12719
Giannelli G, Bergamini C, Fransvea E, Sgarra C, Antonaci S (2005) Laminin-5 with transforming growth factor-beta1 induces epithelial to mesenchymal transition in hepatocellular carcinoma. Gastroenterology 129(5):1375–1383. https://doi.org/10.1053/j.gastro.2005.09.055
Giannelli G, Villa E, Lahn M (2014) Transforming growth factor-β as a therapeutic target in hepatocellular carcinoma. Cancer Res 74(7):1890–1894. https://doi.org/10.1158/0008-5472.CAN-14-0243 (Review)
Giannelli G, Mikulits W, Dooley S, Fabregat I, Moustakas A, ten Dijke P et al (2016) The rationale for targeting TGF-β in chronic liver diseases. Eur J Clin Invest 46(4):349–361. https://doi.org/10.1111/eci.12596 (Review)
Gomori G (1950) A rapid one-step trichrome stain. Am J Clin Pathol 20(7):661–664
Hammad S, Hoehme S, Friebel A, von Recklinghausen I, Othman A, Begher-Tibbe B et al (2014) Protocols for staining of bile canalicular and sinusoidal networks of human, mouse and pig livers, three-dimensional reconstruction and quantification of tissue microarchitecture by image processing and analysis. Arch Toxicol 88(5):1161–1183. https://doi.org/10.1007/s00204-014-1243-5
Hiramatsu K, Aoyama H, Zen Y, Aishima S, Kitagawa S, Nakanuma Y (2006) Proposal of a new staging and grading system of the liver for primary biliary cirrhosis. Histopathology 49(5):466–478. https://doi.org/10.1111/j.1365-2559.2006.02537.x
Ikenaga N, Liu SB, Sverdlov DY, Yoshida S, Nasser I, Ke Q et al (2015) A new Mdr2(-/-) mouse model of sclerosing cholangitis with rapid fibrosis progression, early-onset portal hypertension, and liver cancer. Am J Pathol 185(2):325–334. https://doi.org/10.1016/j.ajpath.2014.10.013
Kim KH, Kim HC, Hwang MY, Oh HK, Lee TS, Chang YC et al (2006) The antifibrotic effect of TGF-beta1 siRNAs in murine model of liver cirrhosis. Biochem Biophys Res Commun 343(4):1072–1078. https://doi.org/10.1016/j.bbrc.2006.03.087
Kocabayoglu P, Friedman SL (2013) Cellular basis of hepatic fibrosis and its role in inflammation and cancer. Front Biosci (Schol Ed) 5:217–230 (Review)
Lammert F, Wang DQ, Hillebrandt S, Geier A, Fickert P, Trauner M et al (2004) Spontaneous cholecysto- and hepatolithiasis in Mdr2-/- mice: a model for low phospholipid-associated cholelithiasis. Hepatology 39(1):117–128. https://doi.org/10.1002/hep.20022
Luangmonkong T, Suriguga S, Bigaeva E, Boersema M, Oosterhuis D, de Jong KP et al (2017) Evaluating the antifibrotic potency of galunisertib in a human ex vivo model of liver fibrosis. Br J Pharmacol 174(18):3107–3117. https://doi.org/10.1111/bph.13945
Mauad TH, van Nieuwkerk CM, Dingemans KP, Smit JJ, Schinkel AH, Notenboom RG et al (1994) Mice with homozygous disruption of the mdr2 P-glycoprotein gene. A novel animal model for studies of nonsuppurative inflammatory cholangitis and hepatocarcinogenesis. Am J Pathol 145(5):1237–1245
Nieto MA, Huang RY, Jackson RA, Thiery JP (2016) EMT: 2016. Cell 166(1):21–45. https://doi.org/10.1016/j.cell.2016.06.028 (Review)
Okuno M, Akita K, Moriwaki H, Kawada N, Ikeda K, Kaneda K et al (2001) Prevention of rat hepatic fibrosis by the protease inhibitor, camostat mesilate, via reduced generation of active TGF-beta. Gastroenterology 120(7):1784–17800. https://doi.org/10.1053/gast.2001.24832
Popov Y, Patsenker E, Fickert P, Trauner M, Schuppan D (2005) Mdr2 (Abcb4)-/- mice spontaneously develop severe biliary fibrosis via massive dysregulation of pro- and antifibrogenic genes. J Hepatol 43(6):1045–1054. https://doi.org/10.1016/j.jhep.2005.06.025
Qi Z, Atsuchi N, Ooshima A, Takeshita A, Ueno H (1999) Blockade of type beta transforming growth factor signaling prevents liver fibrosis and dysfunction in the rat. Proc Natl Acad Sci 96(5):2345–2349. https://doi.org/10.1073/pnas.96.5.2345
Rodon J, Carducci MA, Sepulveda-Sánchez JM, Azaro A, Calvo E, Seoane J et al (2015) First-in-human dose study of the novel transforming growth factor-β receptor I kinase inhibitor LY2157299 monohydrate in patients with advanced cancer and glioma. Clin Cancer Res 21(3):553–560. https://doi.org/10.1158/1078-0432.CCR-14-1380
Rygiel KA, Robertson H, Marshall HL, Pekalski M, Zhao L, Booth TA et al (2008) Epithelial-mesenchymal transition contributes to portal tract fibrogenesis during human chronic liver disease. Lab Invest 88(2):112–123. https://doi.org/10.1038/labinvest.3700704
Santamato A, Fransvea E, Dituri F, Caligiuri A, Quaranta M, Niimi T et al (2011) Hepatic stellate cells stimulate HCC cell migration via laminin-5 production. Clin Sci (Lond) 121(4):159–168. https://doi.org/10.1042/CS20110002
Serova M, Tijeras-Raballand A, Dos Santos C, Albuquerque M, Paradis V, Neuzillet C et al (2015) Effects of TGF-beta signalling inhibition with galunisertib (LY2157299) in hepatocellular carcinoma models and in ex vivo whole tumor tissue samples from patients. Oncotarget 6(25):21614–21627. https://doi.org/10.18632/oncotarget.4308
Vartak N, Damle-Vartak A, Richter B, Dirsch O, Dahmen U, Hammad S et al (2016) Cholestasis-induced adaptive remodeling of interlobular bile ducts. Hepatology 63(3):951–964. https://doi.org/10.1002/hep.28373
Williams MJ, Clouston AD, Forbes SJ (2014) Links between hepatic fibrosis, ductular reaction, and progenitor cell expansion. Gastroenterology 146(2):349–356. https://doi.org/10.1053/j.gastro.2013.11.034
Yoshida K, Murata M, Yamaguchi T, Matsuzaki K, Okazaki K (2016) Reversible human TGF-β signal shifting between tumor suppression and fibro-carcinogenesis: implications of smad phospho-isoforms for hepatic epithelial-mesenchymal transitions. J Clin Med 5(1):E7. https://doi.org/10.3390/jcm5010007 (Review)
Acknowledgements
We thank Christof Dormann and Friedrich Behne for their excellent technical support.
Funding
This work was supported by the BMBF program LiSyM (SD: Grant PTJ-FKZ: 031 L0043), e:Bio-Modull-II: MS_DILI and by the Italian Ministry of Health, Ricerca Corrente 2018.
Author information
Authors and Affiliations
Contributions
SH, JW and AD conceived the study, performed data analyses and wrote the manuscript. EC, MLC and AI performed the pathological evaluation. MPE, SD and GG performed critical revision of the manuscript. SD and GG provided supervisory support and corrected the manuscript. All authors read the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
204_2018_2231_MOESM1_ESM.pptx
Fig. S1 Liver cell proliferation, resident macrophage and leucocyte numbers upon Galunisertib treatment in 26-weeks-old Abcb4ko mice. a) Representative images of Ki-67 (proliferation marker), F4/80 (resident macrophages) and CD45 (leukocyte common marker) positive signals in liver tissues upon Galunisertib or vehicle administration. Scale bars are 100 µm. b and c) show numbers of proliferating hepatocytes and non-parenchymal cells, as indicated by Ki-67 positivity. d and e) quantification of F4/80 positive areas and number of CD45 positive cells, among different groups. Data are means ± SD of 5–7 mice per group. N.S.: P > 0.05; **: P < 0.01 compared with untreated livers unless otherwise indicated. (PPTX 4035 KB)
204_2018_2231_MOESM2_ESM.pptx
Body and liver weight as well as liver enzymes upon Galunisertib treatment in 26-weeks-old Abcb4ko mice. a–c mouse and liver weight, and liver as percentage of total body weight. No significant difference is present due to Galunisertib administration. d–f) liver enzymes in Galunisertib, vehicle and untreated mice. e) H&E staining across different groups. Scale bars are 200 µm. Data are means ± SD of 5–7 mice per group. N.S.: P > 0.05; *: P < 0.05; **: P < 0.01 compared with untreated livers unless otherwise indicated. (PPTX 1695 KB)
204_2018_2231_MOESM3_ESM.pptx
Negative controls for IHC as used in this study. In the same staining batch, negative controls by omitting first antibodies were included. Scale bars are 100 µm. (PPTX 5991 KB)
204_2018_2231_MOESM4_ESM.pptx
Abcb4ko mouse age, gender and number. Experiment 1 was used to investigate chronic liver disease development and progression. Experiment 2 was performed to test the anti-fibrotic efficiency of Galunisertib. (PPTX 36 KB)
204_2018_2231_MOESM5_ESM.pptx
PBC scoring of liver fibrosis in 12 to 84 weeks old Abcb4ko mice. Correlation analysis is based on Test z versus 12-weeks-old mice (PPTX 69 KB)
Rights and permissions
About this article
Cite this article
Hammad, S., Cavalcanti, E., Werle, J. et al. Galunisertib modifies the liver fibrotic composition in the Abcb4Ko mouse model. Arch Toxicol 92, 2297–2309 (2018). https://doi.org/10.1007/s00204-018-2231-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2231-y