Abstract
Diet plays a fundamental role in maintaining long-term health, with healthful diets being endorsed by current dietary guidelines for the prevention and management of type 2 diabetes. However, the response to dietary interventions varies widely, highlighting the need for refinement and personalisation beyond population-based ‘one size fits all’. This article reviews the clinical evidence supporting precision nutrition as a fundamental approach for dietary advice in diabetes. Further, it proposes a framework for the eventual implementation of precision nutrition and discusses key challenges for the application of this approach in the prevention of diabetes. One implication of this approach is that precision nutrition would not exclude the parallel goal of population-based healthy dietary advice. Nevertheless, the shift in prioritising precision nutrition is needed to reflect the dynamic nature of responses to dietary interventions that vary among individuals and change over the life course.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
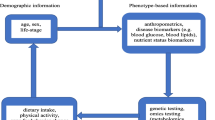
Precision nutrition is the discipline that studies the health effects of nutritional exposure in the context of differences in human biology, environment and social determinants of health. To the extent that a nutrient, food or diet is beneficial in a subgroup of the population, precision nutrition information may be clinically useful by offering nutrients, foods or diets to individuals most likely to benefit from them, while limiting those that are potentially harmful. Conversely, the promotion of certain nutrients, foods or diets in the context of upstream social determinants of health (i.e. the social, physical and economic conditions in society that impact upon health [1]), could reduce health disparities while supporting environmentally sustainable diets. As a corollary, precision nutrition advice for optimal health requires consideration of synergistic approaches targeted at the individual level and those aimed at the population level [2,3,4].
While the terms ‘personalised nutrition’ and ‘precision nutrition’ have been used interchangeably, the latter is more appropriate when defining nutritional approaches based on the combination of biological, environmental and social factors that could be targeted to individuals or populations sharing similar characteristics [5]. In recent years, precision nutrition studies have been facilitated by the growing capture of multiple data points across orthogonal axes of information, as well as the development of analytical methods that permit the interpretation of complex datasets. These have taken the form of gene × diet interaction studies or studies integrating diet with the abundance, composition and activity of the gut microbiota. Proof-of-concept approaches have been successful in multi-omics studies, in which detrimental nutritional responses can be predicted by artificial intelligence algorithms that consider inter-individual differences arising from genetics, gut microbiome, and other clinical and lifestyle characteristics [6,7,8,9,10].
To date, several reviews have been published in the topic of precision nutrition in diabetes [11,12,13]. However, none of them has explicitly focused on how emerging data from diabetes heterogeneity and disease subtypes emphasise the need for precision nutrition. In this narrative review, I summarise the clinical evidence supporting precision nutrition as a fundamental approach for dietary advice in diabetes, propose a framework for the eventual implementation of precision nutrition, and explore challenges for the implementation of this approach for the prevention of diabetes.
Precision nutrition is a fundamental approach for dietary advice
Recommendations aimed at improving overall diet quality are an essential part of preventing and treating type 2 diabetes, as reflected in current dietary guidelines. These guidelines recommend dietary patterns rich in fruits and vegetables, whole grains, nuts, legumes and seafood, and recommend reduced intakes from refined and ultra-processed foods [14,15,16,17]. Although dietary guidelines have become more evidence-driven, with data coming from large observational studies complemented with short-term human clinical trials, many areas of disagreement exist on what constitutes an optimal diet for the prevention and management of type 2 diabetes. Expert consensus decreases particularly for the beneficial effects of dairy, meat and beverages, or dietary patterns such as ketogenic diets [18,19,20].
The reasons underlying these discrepancies are complex and involve many factors, including quality and context of nutritional studies or the funding ecosystem in nutrition research [21]. Inter-individual differences also underlie the divergence regarding the beneficial effect of certain nutrients, foods or diets. Previous studies have shown that inter-individual differences in demographic, clinical, genetic, gut microbiota and lifestyle characteristics result in large variability in responses to identical foods [6, 8,9,10]. As an example, in the Personalised REsponses to DIetary Composition Trial (PREDICT), the population CV in postprandial responses of blood glucose, triacylglycerol and insulin was 68%, 103% and 59%, respectively [10], stressing the need for precision nutrition to reduce the impact of anomalous postprandial glycaemic responses on the development of obesity and diabetes. Refinement and personalisation beyond a population-based ‘one size fits all’ approach is needed to reflect the dynamics of nutritional responses that vary among individuals and change over the life course.
Most dietary interventions, some focusing on macronutrients such as carbohydrate or fat reduction and others on overall dietary patterns, result in progressive weight loss and improvement in cardiovascular risk factors over 6 months, followed by plateau and weight regain after 1 year [22, 23]. While many observers assume that voluntary resumption of old habits explains the lack of efficacy of diet interventions, strong physiological adaptations contribute to the high rate of relapse among people who have initially lost weight. Previous studies have reported that changes in mitochondrial efficiency and energy expenditure serve to attenuate the effect of energy restriction in the short term [24, 25]. Further, increased appetite has been shown to play a primordial role during weight loss maintenance [26]. This distinction implies that strategies to maintain weight lost should be fundamentally different from the ones used for initial weight loss. Further, it suggests that a better understanding of the molecular, behavioural and social drivers of appetite could potentially increase compliance with dietary interventions and prevent weight gain–weight loss cycles (Fig. 1).
Precision nutrition stresses the need to understand better how people interact with food to enhance compliance with dietary interventions. Short-term signalling from the gastrointestinal tract, which includes hormones such as glucagon-like peptide 1 (GLP1) or glucokinase (GCK), and long-term signalling from the adipose tissue due to the action of leptin or insulin are sensed by the brain and modulate appetite. Characterisation of these nutrient-sensing pathways through genetic, epigenetic and transcriptome profiling could identify individuals sharing similar molecular characteristics underlying appetite control (represented as Groups 1, 2 and 3). Further, integrating molecular aspects with environmental and social pressures influencing food intake could identify people who are more vulnerable to our modern food environment. This is particularly important in the context of diet-induced body-weight loss and maintenance, in which physiological adaptations targeted to increase appetite contribute to the high rate of relapse among people who have initially lost weight. Precision nutrition offers an opportunity to improve compliance with dietary interventions and prevent weight gain–weight loss cycles. This figure is available as part of a downloadable slideset
Diabetes heterogeneity stresses the need for precision nutrition
Albeit with the common final denominator of hyperglycaemia, diabetes is a complex disease with substantial aetiological and clinical heterogeneity [27]. People at increased risk of type 2 diabetes can range from those with a clinical phenotype characterised by increased adiposity to those who may have primary defects in insulin secretion pathways with normal weight. Consequently, individuals with hyperglycaemia differ considerably in their propensity to respond to preventive interventions, clinical presentation or the likelihood of develo** complications.
During the last decade, complementary studies have been conducted to discriminate major diabetes subtypes that might enable targeting of therapeutic strategies. A study using information from six clinical variables measured in patients with newly diagnosed diabetes identified five clusters of individuals with different clinical presentation [28]. The five diabetes subtypes (severe autoimmune diabetes, severe insulin-deficient diabetes, severe insulin-resistant diabetes, mild obesity-related diabetes, and mild age-related diabetes) also showed varying degrees of risk of diabetic complications. For example, individuals with severe insulin-resistant diabetes were at increased risk of diabetic kidney disease, while those with severe insulin-deficient diabetes had the highest risk of retinopathy.
Beyond the use of clinical information to identify diabetes subtypes, the stable nature of genetic susceptibility offers a useful tool to aid in the characterisation of individuals at increased risk of diabetes. Two recent genetic studies have identified clusters of genetic variants that increased diabetes risk through specific intermediary processes [29, 30]. These included two processes related to insulin deficiency due to either beta cell dysfunction or impaired proinsulin synthesis, three processes linked to insulin resistance including obesity-mediated insulin resistance, adverse body fat distribution and disrupted liver lipid metabolism, and a group of genetic variants only identified in the Mahajan study [30] with less precise phenotypic characterisation. The generation of ‘pathway-specific’ polygenic scores with cluster-specific variants showed that individuals whose diabetes risk is driven by adverse body fat distribution were at increased risk of coronary artery disease, while those with beta cell dysfunction were at increased risk of stroke [29]. These findings support the notion that differential aspects of diabetes pathophysiology and clinical outcomes could be detected by pathway-specific polygenic scores, although further work is needed to define their use for precision nutrition.
Despite the heterogeneity that characterises diabetes, there is a notion that weight loss should be considered a central target for diabetes prevention regardless of the mechanisms influencing disease risk [31]. While recent studies support the benefits of losing body weight on the risk of develo** diabetes [32], diabetes-related cardiovascular complications [33, 34] or diabetes remission [35], there is still enormous variability in individual response to weight-loss interventions. For example, in the Diet Intervention Examining The Factors Interacting with Treatment Success (DIETFITS) study, a randomised clinical trial to investigate the effect of a healthy low-fat diet vs a healthy low-carbohydrate diet on weight change at 12 months, weight change varied widely within each study group, ranging from a loss of approximately 30 kg to a gain of approximately 10 kg [36]. The study showed that neither genetic variation underlying fat and carbohydrate metabolism nor insulin secretion interacted with diet, suggesting that these factors did not significantly explain weight loss variability. To identify factors associated with long-term weight-loss success, a post hoc analysis of the Diabetes Prevention Program Outcomes Study showed that losing more than 5% of body weight during the first year of the intervention, achieved either by an intensive lifestyle intervention or metformin, was an independent predictor of long-term weight loss success and diabetes risk after 15 years of follow-up [37]. These findings reinforce the continued effort to identify molecular, environmental and social characteristics underlying the variable response to diabetes prevention interventions.
Evidence of precision nutrition studies in diabetes
Gene × diet interaction studies in diabetes
The foundation for a new era of precision nutrition has been set by recent technological advances in omics and wearable monitoring that enable deep molecular and physiological profiling. Previous studies have yielded convincing examples of how environmental pressures have enriched the human genome with specific variants that could affect an individual’s response to therapeutic interventions [38]. One such study, among people of Mexican or Latin American descent, identified a common risk haplotype in the SLC16A11 locus associated with ∼20% increased risk of type 2 diabetes [39]. Carriers of the risk haplotype have low plasma levels of monocarboxylate transporter 11 (the protein encoded by SLC16A11) in the plasma membrane of hepatocytes and, under energy overload, there is a metabolic shift towards triacylglycerol accumulation [40]. While population-wide dietary advice to avoid energy-dense foods is beneficial in reducing the risk of obesity and diabetes, limiting energy intake could be particularly valuable among carriers of this risk haplotype.
Beyond specific genetic variants, a critical question is whether polygenic scores, which provide a quantitative measure of genetic risk to a disease, could be relevant to precision nutrition. In a prospective study including male health professionals, the association between a polygenic score for type 2 diabetes and the risk of the disease was amplified by a western dietary pattern [41]. However, no evidence of significant interactions was detected between genetic risk and the Mediterranean diet on the development of diabetes in the InterAct study [42]. Null findings were also reported in an individual-participant-data meta-analysis to investigate interactions between genetic risk and diet fat quality [43], and in a UK Biobank study showing that diet was associated with incident diabetes within and across genetic risk groups [44]. While it is possible that genetic and dietary factors interact in ways that can be difficult to disentangle with current methods, these studies suggest that genetic variation alone is not enough to target dietary recommendations and that more holistic approaches through the integration of biological, environmental and social metrics are needed to target dietary advice.
Diet and gut microbiome studies in diabetes
In parallel with the identification of genetic profiles for precision nutrition, there is a growing interest in discovering gut microbiota signatures that interact or mediate the effect of diet in diabetes. The Men’s Lifestyle Validation Study, a substudy of the long-running Health Professionals Follow-Up Study, represents one of the most comprehensive biorepositories to date to identify such signatures. By leveraging longitudinal metagenomics and metatranscriptomics profiles from 307 healthy men, a recent study showed that the cardioprotective benefits of the Mediterranean diet are particularly relevant among individuals with decreased abundance of Prevotella copri in the gut [45]. The study documented that higher adherence to Mediterranean diet was associated with an 18% lower risk of myocardial infarction among those with decreased abundance of P. copri (95% CI 0.69, 0.95) and a non-significant 30% increase in myocardial infarction risk among P. copri carriers (95% CI 0.83, 2.07). While P. copri encompasses four distinct clades that are widespread in non-westernised populations [46], the study confirmed that P. copri in this study population consisted entirely of clade A. In a separate study, higher habitual intake of red meat was significantly associated with increased HbA1c levels only among participants with a microbial profile enriched with trimethylamine N-oxide (TMAO)-producing bacteria [47]. Of these microorganisms, Alistipes shahii magnified the positive association between red meat intake and HbA1c levels, in which red meat was associated with higher HbA1c among participants with microbial profile enriched with A. shahii and lower HbA1c among those without A. shahii. Elsewhere, a gut microbiome signature including 21 fibre-fermentation bacteria mediated 60% of the beneficial effect of overall diet quality, measured by the Alternate Healthy Eating Index 2010, on the risk of diabetes [48]. While these studies are preliminary and lack independent replication, the discovery of gut microbiota profiles modulating the effect of dietary and lifestyle exposures, and their incorporation into risk scores, could help advance precision nutrition in diabetes.
Integrated omics and diet studies in diabetes
Beyond single omics approaches, clinical studies collecting personal, dense and dynamic data are inspiring new venues for complex disease prevention and treatment through better understanding of inter-individual variability. The Personalised Nutrition Project showed large inter-individual variability in glycaemic responses to exact same meals and demonstrated the accuracy of a machine learning algorithm to predict postprandial glycaemic response to real-life meals [6]. The algorithm, which was validated in an independent cohort of 100 participants, predicted individual postprandial glycaemic responses better than models based on other approaches such as carbohydrate counting or glycaemic index scores. In a follow-up double-blinded randomised crossover trial of 26 individuals, algorithm-informed dietary advice resulted in improved glycaemic variables after 1 week [6]. The recent PREDICT study showed similar wide variation in postprandial responses to identical foods and demonstrated that people who display poor metabolic responses to a given food are likely to respond poorly to other similar foods [10]. Meal composition and genetic factors were the main predictors of postprandial glycaemic responses, while serum lipid and glycaemic markers explained the larger variation in postprandial lipid responses. These studies highlight the potential of precision nutrition to reduce the unfavourable impact of postprandial glycaemic responses on health outcomes.
A framework for the eventual implementation of precision nutrition in diabetes
Major scientific guidelines already emphasise flexible and culturally adapted healthy dietary patterns for the prevention and management of type 2 diabetes [14,15,16,17] but there are likely to be additional benefits of a precision nutrition approach based on differences in genetics, gut microbiome composition and other clinical, social and environmental factors. A recent clinical trial including adults with impaired fasting glucose randomised to either a personalised diet based on predicted glucose responses or a Mediterranean diet for 6 months, and an additional 6 month follow-up, showed that the personalised diet was superior to the Mediterranean diet in terms of HbA1c reduction and daily time spent with glucose levels >7.8 mmol/l (140 mg/dl) [49]. These findings imply that diet advice focused on favourable postprandial glycaemic responses is effective in improving glycaemic control in prediabetes.
A framework for the eventual implementation of precision nutrition in diabetes should be grounded on the set of physiological processes that contribute to the development of the disease (Fig. 2). The rationale for this approach is based on McCarthy’s ‘Palette’ model of type 2 diabetes, in which the aetiological processes that contribute to diabetes risk are the same across individuals (base colours) but different individuals have different components of each process (mixture and saturation of hues) that results in a heterogeneous disease risk, presentation and progression [50]. These physiological processes and components are highly influenced by environmental, social and clinical factors such as ageing or body-weight gain. Thus, genetic information for physiological processes underlying diabetes risk needs to be combined with orthogonal axes of molecular, environmental and social data collected over time. In combining this information, it is important to preserve complexity by tracking each process involved separately and to consider gradation of risk rather than collapsing rich quantitative information into rigid clinical categories [51]. This approach is consistent with the observed large inter-individual variable response to healthful dietary interventions, in which most people benefit from the healthy diet irrespective of underlying susceptibility to disease, but refinement is necessary among those who might benefit less. For example, the Diabetes Prevention Program showed that a lifestyle intervention is effective in all study subgroups but that its effect is significantly attenuated among individuals with impaired fasting glucose [32]. Similarly, a post hoc analysis of the Look AHEAD (Action for Health in Diabetes) study showed that individuals with a diabetes phenotype characterised by poor glucose control are less likely to benefit from a lifestyle intervention in terms of body-weight loss and cardiovascular protection [52], implying that targeted advice based on underlying alterations could optimise the health-enhancing effects of diet.
A framework for the eventual implementation of precision nutrition in diabetes. Precision nutrition in diabetes should be grounded on the set of physiological processes that contribute to the development of the disease. This concept is illustrated using a model of three hypothetical diabetes components (shown in purple, yellow and grey, with different tones and shades to denote different mechanisms affecting each process) present many years before the development of the disease. Deep phenotypic, environmental and social data collected at different time points would be fed into a predictive algorithm to identify nutrients, foods and diets with optimal, neutral or detrimental profiles (denoted as green, yellow and red). For example, purple individuals would benefit the most from peppers, while people whose diabetes risk is driven by grey would benefit from lettuce. With the proposed framework, precision nutrition advice needs to be readjusted with data collected at different time points through the life course. Further, rapid changes in body weight or physical activity levels are likely to modify food responses, reinforcing the notion of dynamic nutritional advice. This figure is available as part of a downloadable slideset
The dynamic and adaptable nature of human metabolism implies that the set of physiological processes underlying disease risk would have different impacts along the life course [53]. A recent study suggests that changes in components of body composition at different stages of the life course are a source of metabolic variability associated with diabetes risk [54]. Consequently, the order in which pathophysiological stages appear may not be the same for everyone [55], highlighting the need of multi-dimensional, high-resolution and time-series data to characterise metabolic status and inform targeted dietary advice. In general, early beta cell failure is characterised by a reduction in the first phase of glucose-stimulated insulin release, leading to hyperinsulinaemia and diabetes. While dietary advice should be targeted at preserving beta cell function among people whose diabetes risk is mainly driven through alterations in insulin synthesis and secretion, for someone with established beta cell insufficiency weight loss could be more relevant to delay progression to diabetes. There is consistent evidence showing that glucose variability and postprandial glycaemic peaks induce glucotoxicity effects and consequent beta cell dysfunction [56]. Therefore, dietary advice to avoid foods or meals that produce large glycaemic fluctuations could be particularly beneficial in individuals with early beta cell failure.
Previous studies have shown that weight loss increases beta cell function because of an increase in both beta cell glucose sensitivity and whole-body insulin sensitivity [57]. Short-term weight loss can be achieved through different healthful diets, as significant weight loss is often observed when people are randomised to a range of different diets [23]. To maintain weight lost, a personalised approach to mitigate physiological adaptations related to increased appetite would be necessary. Physiological adaptations underlying increased energy intake are heterogeneous and may change over time [58], implying that frequent assessment of fluctuations in psychological, social or molecular factors is necessary to capture this dynamic adaptation and offer targeted dietary advice. There is early evidence that reducing blood glucose dips is associated with lower energy intake [59], suggesting that the promotion of nutrients, foods and meals that mitigate glycaemic dips, rather than glycaemic peaks, could be relevant for counterbalancing increased energy intake and achieving long-term body weight maintenance.
Practical considerations and challenges for implementing precision nutrition in diabetes
There are important considerations when redefining dietary advice for diabetes prevention and management based on precision nutrition (see text box). First, precision nutrition is not a competitor but an addition to what we know about healthy eating for the prevention and management of diabetes. In the same way that promotion of physical activity and weight loss do not replace the need to adhere to a healthy diet, precision nutrition advice would not exclude the parallel goal of healthy dietary patterns nor replace other evidence-based strategies for decreasing the risk of diabetes, such as novel weight-loss or glucose-lowering medications. However, the shift in emphasis to prioritise precision nutrition is important to reflect the dynamic and divergent nature of responses to dietary interventions.

Second, precise tools are needed for the eventual implementation of precision nutrition. The use of objective biomarkers of diet is necessary to mitigate random and systematic error from self-reported dietary assessment methods [60]. Further, scientific scrutiny should precede the introduction of emerging tools, such as continuous glucose monitoring (CGM) devices, for precision nutrition. A recent domiciled-feeding study including 16 healthy adults connected to two CGM devices showed that foods categorised as favourable by one device were not always considered favourable by the other device [61]. While these results call into question the use of CGM devices for precision nutrition applications, a post hoc study within the PREDICT cohort, including 34 individuals connected to two CGM devices, provided evidence that most of the glycaemic rankings were concordant between devices [62]. Discordant rankings were mainly observed for foods or meals with lower carbohydrate content. Technological advances to improve interstitial glucose-sensing accuracy are needed, especially during the postprandial state in which rapid changes in glucose, blood flood rate or temperature could impact measurement precision. In addition, there is a need to develop new technologies that go beyond changes in blood glucose levels and monitor other components that change in response to diet.
Third, clinical trials specifically designed to capture the nuances of dynamic nutritional responses are needed for the eventual implementation of precision nutrition. Most precision nutrition evidence comes from post hoc analysis of clinical trials and observational studies, and independent replication in new intervention studies is usually missing. Due to their short follow-up periods, these studies do not adequately capture the dynamic complexity of dietary and behavioural modification. It is unlikely that our increasing understanding of the pathophysiology of diabetes will translate into precision nutrition unless clinical trials overcome three main aspects: (1) the implementation of repeated-crossover clinical studies to investigate an individual’s response to diet at different cycles [63]; (2) the specification of a clear and precise outcome definition that goes beyond rigid disease categories [64]; and (3) the development of reproducible algorithms and statistical methods that could help solve challenges related to complex datasets [64].
Fourth, a key consideration for precision nutrition in diabetes is whether deep phenoty** is better than a simple clinical determination of insulin resistance/secretion. Recent studies have provided evidence that deep ‘omics’ profiling could detect subtle changes in health markers at the earliest possible time point [65, 66], allowing anticipatory prevention strategies targeted towards disrupted processes. The identification of actionable biomarkers of early alterations in insulin homeostasis is an unmet need in diabetes given that clinical tests such as the OGTT cannot describe the nuances of glycaemic and insulin fluctuations [67]. While the costs of deep phenoty** are greater than those of routine clinical testing, diabetes is now a national priority and the most expensive chronic illness in the USA. Cost–effect studies have consistently shown that investing in diabetes prevention is cost-effective [68], implying that the costs of acquiring deep phenotype data would be amortised by enhanced prevention. However, no comprehensive attempts have been made to rigorously evaluate the cost-effectiveness of precision nutrition in diabetes.
Fifth, equitable access to effective precision nutrition advice should be ensured [69]. Among the nearly half-billion people with diabetes globally, three out of every four people live in low- and middle-income countries, and the prevalence of the disease in high-income countries tends to be higher among ethnic minorities or people living in areas with higher deprivation [70]. There is a need to embrace diversity in the design, implementation and clinical validation of studies to reduce diabetes disparities and bias towards underrepresented minorities. Further, addressing upstream social determinants of health, particularly those related to the food environment, is critical for hel** people sustain targeted dietary advice [71]. Our food system is overflowing with energy-dense and highly palatable foods that are almost perfectly designed to maximise food reward properties and promote overconsumption. Policies that affect food pricing, sales and advertising have been successful in reducing consumption of unhealthy foods [72, 73], implying that the success of precision nutrition will be parallel to the implementation of strategies that modify the structural and environmental context in which eating behaviours occur.
Finally, important questions about the efficacy, sustainability and delivery of precision nutrition need to be answered before translating precision-based strategies into clinical practice or guidelines. Long-term clinical trials specifically designed to assess head-to-head comparisons between precision nutrition interventions and population-wide approaches are needed. One of the main reasons for low compliance with dietary recommendations is the physiological adaptations targeted at increasing energy intake. The development and validation of clinical algorithms that could predict and mitigate processes underlying increased appetite or food reward is needed. Another important question is how precision nutrition recommendations would be delivered. Right now, the gold standard for delivering dietary advice is to follow a standardised programme consisting of group counselling combined with individualised advice. Using advanced methods of communication and education through personal websites or medical portals, precision nutrition aims to engage individuals in their own care by incorporating real-time collection of data and providing real-time feedback to reinforce positive behaviours. In a new era of remote clinical testing and citizen science, in which various commercial companies are offering precision nutrition advice based on clinical, lifestyle, genetic and gut microbiome variation, it is important to address the current challenges by establishing a solid evidence base, protocols and guidelines that could benefit individuals and populations.
Conclusion
Early evidence stresses the potential of precision nutrition in diabetes. This approach would address the inter-individual response observed in diet and weight-loss interventions, target main pathophysiological processes underlying the continuum of glycaemic alterations, and reap the benefits of population-based dietary advice for the prevention of diabetes and related complications. Shifting from population-based ‘one size fits all’ dietary advice to targeted recommendations would recognise individual-level factors as predominant drivers of the effect of food intake on disease risk while considering the importance of social and environmental pressures on sha** how people interact with and respond to foods. Precision nutrition is still in its infancy and only time and much more research will tell if the initial prospects can be realised, and whether precision nutrition could be implemented as a frontline intervention for the prevention and management of diabetes.
Abbreviations
- CGM:
-
Continuous glucose monitoring
- PREDICT:
-
Personalised REsponses to DIetary Composition Trial
References
U.S. Department of Health and Human Services, Office of Disease Prevention and Health Promotion. Healthy People 2030: Social Determinants of Health. Available at https://health.gov/healthypeople/objectives-and-data/social-determinants-health. Accessed 14 Feb 2022
Gillies CL, Abrams KR, Lambert PC et al (2007) Pharmacological and lifestyle interventions to prevent or delay type 2 diabetes in people with impaired glucose tolerance: systematic review and meta-analysis. BMJ 334(7588):299. https://doi.org/10.1136/bmj.39063.689375.55
Unwin N, Whiting D, Roglic G (2010) Social determinants of diabetes and challenges of prevention. Lancet 375(9733):2204–2205. https://doi.org/10.1016/S0140-6736(10)60840-9
White M (2016) Population approaches to prevention of type 2 diabetes. PLoS Med 13(7):e1002080. https://doi.org/10.1371/journal.pmed.1002080
Chung WK, Erion K, Florez JC et al (2020) Precision medicine in diabetes: a consensus report from the American Diabetes Association (ADA) and the European Association for the Study of diabetes (EASD). Diabetologia 63(9):1671–1693. https://doi.org/10.1007/S00125-020-05181-W
Zeevi D, Korem T, Zmora N et al (2015) Personalised nutrition by prediction of glycemic responses. Cell 163(5):1079–1094. https://doi.org/10.1016/j.cell.2015.11.001
Thaiss CA, Itav S, Rothschild D et al (2016) Persistent microbiome alterations modulate the rate of post-dieting weight regain. Nature 540(7634):544–551. https://doi.org/10.1038/nature20796
Korem T, Zeevi D, Zmora N et al (2017) Bread affects clinical parameters and induces gut microbiome-associated personal glycemic responses. Cell Metab 25(6):1243–1253.e5. https://doi.org/10.1016/j.cmet.2017.05.002
Mendes-Soares H, Raveh-Sadka T, Azulay S et al (2019) Assessment of a personalized approach to predicting postprandial glycemic responses to food among individuals without diabetes. JAMA Netw Open 2(2):e188102. https://doi.org/10.1001/jamanetworkopen.2018.8102
Berry SE, Valdes AM, Drew DA et al (2020) Human postprandial responses to food and potential for precision nutrition. Nat Med 26(6):964–973. https://doi.org/10.1038/s41591-020-0934-0
Wang DD, Hu FB (2018) Precision nutrition for prevention and management of type 2 diabetes. Lancet Diabetes Endocrinol 6(5):416–426. https://doi.org/10.1016/S2213-8587(18)30037-8
Bashiardes S, Godneva A, Elinav E, Segal E (2018) Towards utilization of the human genome and microbiome for personalized nutrition. Curr Opin Biotechnol 51:57–63. https://doi.org/10.1016/J.COPBIO.2017.11.013
de Toro-Martín J, Arsenault BJ, Després JP, Vohl MC (2017) Precision nutrition: a review of personalized nutritional approaches for the prevention and management of metabolic syndrome. Nutrients 9(8):913. https://doi.org/10.3390/NU9080913
U.S. Department of Agriculture and U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 2020–2025. 9th Edition. December 2020. Available at http://www.dietaryguidelines.gov. Accessed 28 Jan 2022
Ley SH, Hamdy O, Mohan V, Hu FB (2014) Prevention and management of type 2 diabetes: dietary components and nutritional strategies. Lancet 383(9933):1999–2007. https://doi.org/10.1016/S0140-6736(14)60613-9
Mozaffarian D (2016) Dietary and policy priorities for cardiovascular disease, diabetes, and obesity: a comprehensive review. Circulation 133(2):187–225. https://doi.org/10.1161/CIRCULATIONAHA.115.018585
Evert AB, Dennison M, Gardner CD et al (2019) Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care 42(5):731–754. https://doi.org/10.2337/dci19-0014
Forouhi NG, Krauss RM, Taubes G, Willett W (2018) Dietary fat and cardiometabolic health: evidence, controversies, and consensus for guidance. BMJ 361:k2139
Trepanowski JF, Kroeger CM, Barnosky A et al (2017) Effect of alternate-day fasting on weight loss, weight maintenance, and cardioprotection among metabolically healthy obese adults: a randomized clinical trial. JAMA Intern Med 177(7):930–938. https://doi.org/10.1001/JAMAINTERNMED.2017.0936
Schulze MB, Martínez-González MA, Fung TT, Lichtenstein AH, Forouhi NG (2018) Food based dietary patterns and chronic disease prevention. BMJ 361:k239. https://doi.org/10.1136/bmj.k2396
Spector TD, Gardner CD (2020) Challenges and opportunities for better nutrition science-an essay by Tim Spector and Christopher Gardner. BMJ 369:m2470. https://doi.org/10.1136/BMJ.M2470
Johnston BC, Kanters S, Bandayrel K et al (2014) Comparison of weight loss among named diet programs in overweight and obese adults: a meta-analysis. JAMA 312(9):923–933. https://doi.org/10.1001/jama.2014.10397
Ge L, Sadeghirad B, Ball GDC et al (2020) Comparison of dietary macronutrient patterns of 14 popular named dietary programmes for weight and cardiovascular risk factor reduction in adults: systematic review and network meta-analysis of randomised trials. BMJ 369:m696. https://doi.org/10.1136/BMJ.M696
Martins C, Gower BA, Hill JO, Hunter GR (2020) Metabolic adaptation is not a major barrier to weight-loss maintenance. Am J Clin Nut 112(3):558–565. https://doi.org/10.1093/AJCN/NQAA086
Hall KD, Sacks G, Chandramohan D et al (2011) Quantification of the effect of energy imbalance on bodyweight. Lancet 378(9793):826–837. https://doi.org/10.1016/S0140-6736(11)60812-X
Guo J, Brager DC, Hall KD (2018) Simulating long-term human weight-loss dynamics in response to calorie restriction. Am J Clin Nut 107(4):558. https://doi.org/10.1093/AJCN/NQX080
Tuomi T, Santoro N, Caprio S, Cai M, Weng J, Groop L (2014) The many faces of diabetes: a disease with increasing heterogeneity. Lancet 383(9922):1084–1094. https://doi.org/10.1016/S0140-6736(13)62219-9
Ahlqvist E, Storm P, Käräjämäki A et al (2018) Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol 6(5):361–369. https://doi.org/10.1016/S2213-8587(18)30051-2
Udler MS, Kim J, von Grotthuss M et al (2018) Type 2 diabetes genetic loci informed by multi-trait associations point to disease mechanisms and subtypes: a soft clustering analysis. PLoS Med 15(9):e1002654. https://doi.org/10.1371/journal.pmed.1002654
Mahajan A, Wessel J, Willems SM et al (2018) Refining the accuracy of validated target identification through coding variant fine-map** in type 2 diabetes. Nat Gen 50(4):559–571. https://doi.org/10.1038/S41588-018-0084-1
Lingvay I, Sumithran P, Cohen RV, le Roux CW (2022) Obesity management as a primary treatment goal for type 2 diabetes: time to reframe the conversation. Lancet 399(10322):394–405. https://doi.org/10.1016/S0140-6736(21)01919-X
Knowler WC, Barrett-Connor E, Fowler SE et al (2002) Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 346(6):393–403. https://doi.org/10.1056/NEJMoa012512
Gregg EW, Jakicic JM, Blackburn G et al (2016) Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: a post-hoc analysis of the look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol 4(11):913–921. https://doi.org/10.1016/S2213-8587(16)30162-0
Ma C, Avenell A, Bolland M et al (2017) Effects of weight loss interventions for adults who are obese on mortality, cardiovascular disease, and cancer: systematic review and meta-analysis. BMJ 359:j4849. https://doi.org/10.1136/BMJ.J4849
Lean MEJ, Leslie WS, Barnes AC et al (2019) Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol 7(5):344–355. https://doi.org/10.1016/S2213-8587(19)30068-3
Gardner CD, Trepanowski JF, Del Gobbo LC et al (2018) Effect of low-fat vs low-carbohydrate diet on 12-month weight loss in overweight adults and the association with genotype pattern or insulin secretion: the DIETFITS randomized clinical trial. JAMA 319(7):667–679. https://doi.org/10.1001/JAMA.2018.0245
Apolzan JW, Venditti EM, Edelstein SL et al (2019) Long-term weight loss with metformin or lifestyle intervention in the diabetes prevention program outcomes study. Ann Inter Med 170(10):682–690. https://doi.org/10.7326/M18-1605
Fan S, Hansen ME, Lo Y, Tishkoff SA (2016) Going global by adapting local: a review of recent human adaptation. Science 354(6308):54–59. https://doi.org/10.1126/SCIENCE.AAF5098
The SIGMA Type 2 Diabetes Consortium, Williams AL, Jacobs SBR et al (2014) Sequence variants in SLC16A11 are a common risk factor for type 2 diabetes in Mexico. Nature 506(7486):97–101. https://doi.org/10.1038/nature12828
Ainbinder A, Zhao L, Glover P et al (2021) Mct11 deficiency alters hepatic glucose metabolism and energy homeostasis. bioRxiv 2021.09.08.459307. https://doi.org/10.1101/2021.09.08.459307
Qi L, Cornelis MC, Zhang C, van Dam RM, Hu FB (2009) Genetic predisposition, Western dietary pattern, and the risk of type 2 diabetes in men. Am J Clin Nut 89(5):1453–1458. https://doi.org/10.3945/ajcn.2008.27249
Langenberg C, Sharp SJ, Franks PW et al (2014) Gene-lifestyle interaction and type 2 diabetes: the EPIC InterAct case-cohort study. PLoS Med 11(5):e1001647. https://doi.org/10.1371/journal.pmed.1001647
Merino J, Guasch-Ferre M, Ellervik C et al (2019) Quality of dietary fat and genetic risk of type 2 diabetes: individual participant data meta-analysis. BMJ 366:l4392. https://doi.org/10.1136/bmj.l4292
Said MA, Verweij N, van der Harst P (2018) Associations of combined genetic and lifestyle risks with incident cardiovascular disease and diabetes in the UK biobank study. JAMA Cardiol 3(8):693–702. https://doi.org/10.1001/JAMACARDIO.2018.1717
Abdu-Ali GS, Mehta RS, Lloyd-Price J et al (2018) Metatranscriptome of human faecal microbial communities in a cohort of adult men. Nat Microbiol 3(3):356–366. https://doi.org/10.1038/S41564-017-0084-4
Tett A, Huang KD, Asnicar F et al (2019) The Prevotella copri complex comprises four distinct clades underrepresented in westernized populations. Cell Host Microbe 26(5):666–679.e7. https://doi.org/10.1016/J.CHOM.2019.08.018
Li J, Li Y, Ivey KL et al (2022) Interplay between diet and gut microbiome, and circulating concentrations of trimethylamine N-oxide: findings from a longitudinal cohort of US men. Gut 71(4):724–733. https://doi.org/10.1136/GUTJNL-2020-322473
Qi Q, Li J, Yu B et al (2021) Host and gut microbial tryptophan metabolism and type 2 diabetes: an integrative analysis of host genetics, diet, gut microbiome and circulating metabolites in cohort studies. Gut. Online ahead of print. https://doi.org/10.1136/GUTJNL-2021-324053
Ben-Yacov O, Godneva A, Rein M et al (2021) Personalised postprandial glucose response-targeting diet versus Mediterranean diet for glycemic control in prediabetes. Diabetes Care 44(9):1980–1991. https://doi.org/10.2337/DC21-0162
McCarthy MI (2017) Painting a new picture of personalised medicine for diabetes. Diabetologia 60(5):793–799. https://doi.org/10.1007/s00125-017-4210-x
McCarthy M, Birney E (2021) Personalized profiles for disease risk must capture all facets of health. Nature 597(7875):175–177. https://doi.org/10.1038/D41586-021-02401-0
Bancks MP, Chen H, Balasubramanyam A et al (2021) Type 2 diabetes subgroups, risk for complications, and differential effects due to an intensive lifestyle intervention. Diabetes Care 44(5):1203–1210. https://doi.org/10.2337/DC20-2372
Goodpaster BH, Sparks LM (2017) Metabolic flexibility in health and disease. Cell Metab 25(5):1027–1036. https://doi.org/10.1016/J.CMET.2017.04.015
Wells JCK (2019) The diabesity epidemic in the light of evolution: insights from the capacity-load model. Diabetologia 62(10):1740–1750. https://doi.org/10.1007/S00125-019-4944-8
Johnson JD (2021) On the causal relationships between hyperinsulinaemia, insulin resistance, obesity and dysglycaemia in type 2 diabetes. Diabetologia 64(10):2138–2146. https://doi.org/10.1007/S00125-021-05505-4
Kohnert KD, Augstein P, Zander E et al (2009) Glycemic variability correlates strongly with postprandial β-cell dysfunction in a segment of type 2 diabetic patients using oral hypoglycemic agents. Diabetes Care 32(6):1058–1062. https://doi.org/10.2337/DC08-1956
Yoshino M, Kayser BD, Yoshino J et al (2020) Effects of diet versus gastric bypass on metabolic function in diabetes. N Engl J Med 383(8):721–732. https://doi.org/10.1056/NEJMOA2003697
Greenway FL (2015) Physiological adaptations to weight loss and factors favouring weight regain. Int J Ob 39(8):1188–1196. https://doi.org/10.1038/IJO.2015.59
Wyatt P, Berry SE, Finlayson G et al (2021) Postprandial glycaemic dips predict appetite and energy intake in healthy individuals. Nat Metab 3(4):523–529. https://doi.org/10.1038/S42255-021-00383-X
Freedman LS, Schatzkin A, Midthune D, Kipnis V (2011) Dealing with dietary measurement error in nutritional cohort studies. J Natl Cancer Inst 103(14):1086–1092. https://doi.org/10.1093/JNCI/DJR189
Howard R, Guo J, Hall KD (2020) Imprecision nutrition? Different simultaneous continuous glucose monitors provide discordant meal rankings for incremental postprandial glucose in subjects without diabetes. Am J Clin Nutr 112(4):1114–1119. https://doi.org/10.1093/ajcn/nqaa198
Merino J, Linenberg I, Bermingham KM et al (2022) Validity of continuous glucose monitoring for categorizing glycemic responses to diet: implications for use in personalized nutrition. Am J Clin Nutr. Online ahead of print. https://doi.org/10.1093/AJCN/NQAC026
Potter T, Vieira R, de Roos B (2021) Perspective: application of N-of-1 methods in personalized nutrition research. Adv Nutr 12(3):579–589. https://doi.org/10.1093/ADVANCES/NMAA173
Senn S (2018) Statistical pitfalls of personalised medicine. Nature 563(7733):619–621. https://doi.org/10.1038/D41586-018-07535-2
Schüssler-Fiorenza Rose SM, Contrepois K, Moneghetti KJ et al (2019) A longitudinal big data approach for precision health. Nat Med 25(5):792–804. https://doi.org/10.1038/s41591-019-0414-6
Zhou W, Sailani MR, Contrepois K et al (2019) Longitudinal multi-omics of host–microbe dynamics in prediabetes. Nature 569(7758):663–671. https://doi.org/10.1038/s41586-019-1236-x
Hall H, Perelman D, Breschi A et al (2018) Glucotypes reveal new patterns of glucose dysregulation. PLoS Biol 16(7):e2005143. https://doi.org/10.1371/journal.pbio.2005143
Diabetes Prevention Program Research Group (2012) The 10-year cost-effectiveness of lifestyle intervention or metformin for diabetes prevention: an intent-to-treat analysis of the DPP/DPPOS. Diabetes Care 35(4):723–730. https://doi.org/10.2337/dc11-1468
The Global Nutrition Report’s Independent Expert Group https://globalnutritionreport.org/reports/2020-global-nutrition-report/2020-global-nutrition-report-context-covid-19/. Accessed 6 Jan 2022
International Diabetes Federation. IDF Diabetes Atlas. http://www.diabetesatlas.org. Accessed 14 Feb 2022
Popkin BM, Barquera S, Corvalan C et al (2021) Towards unified and impactful policies to reduce ultra-processed food consumption and promote healthier eating. Lancet Diabetes Endocrinol 9(7):462–470. https://doi.org/10.1016/S2213-8587(21)00078-4
Pell D, Mytton O, Penney TL et al (2021) Changes in soft drinks purchased by British households associated with the UK soft drinks industry levy: controlled interrupted time series analysis. BMJ 372:n254. https://doi.org/10.1136/bmj.n254
Sánchez-Romero LM, Canto-Osorio F, González-Morales R et al (2020) Association between tax on sugar sweetened beverages and soft drink consumption in adults in Mexico: open cohort longitudinal analysis of health workers cohort study. BMJ 369:m1311. https://doi.org/10.1136/BMJ.M1311
Acknowledgements
I thank J. C. Florez (Massachusetts General Hospital, USA) for critical reading of this manuscript and P. W. Franks (Lund University, Sweden) for informative and informal discussion on this topic. The research was partially supported by the ADA and the National Institutes of Health (NIH). The views expressed are those of the author and not necessarily those of the ADA or the NIH.
Author’s relationships and activities
JM is an Associate Editor for Diabetologia but played no role in the evaluation of this manuscript. The author declares that there are no other relationships or activities that might bias, or be perceived to bias, this work.
Contribution statement
JM is the sole author of this article and is the final guarantor of the work.
Funding
Work in the author’s laboratory on precision nutrition in diabetes is in part supported by funding from the ADA (7-21-JDFM-005), and the National Institutes of Health (P30 DK040561 and UG1 HD107691).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PPTX 758 kb)
Rights and permissions
About this article
Cite this article
Merino, J. Precision nutrition in diabetes: when population-based dietary advice gets personal. Diabetologia 65, 1839–1848 (2022). https://doi.org/10.1007/s00125-022-05721-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-022-05721-6