Abstract
Aims/hypothesis
The aim of this study was to determine the frequency of undiagnosed glucose abnormalities and the burden of cardiovascular disease (CVD) risk among south Asians and white Europeans attending a systematic screening programme for type 2 diabetes (ADDITION-Leicester) and to estimate the achievable risk reduction in individuals identified with glucose disorders.
Methods
Random samples of individuals (n = 66,320) from 20 general practices were invited for a 75 g OGTT and CVD risk assessment. Ten-year CVD risk among screen-detected people with diabetes or impaired glucose regulation (IGR) (impaired fasting glycaemia and/or impaired glucose tolerance [IGT]) was computed using the Framingham-based ETHRISK engine and achievable risk reduction was predicted using relative reductions for treatments extracted from published trials.
Results
A total of 6,041 participants (48% male, 22% south Asian) aged 40–75 years inclusive were included. Undiagnosed glucose disorders occurred more frequently in south Asians than white Europeans; age and sex adjusted odds ratios were 1.74 (95% CI 1.42–2.13) and 2.30 (95% CI 1.68–3.16) for IGT and diabetes respectively. Prevalence of any undetected glucose disorder was 17.5% in the whole cohort. Adjusted 10-year risk was similar in screen-detected people with IGR and diabetes (18.3% vs 21.6%), and was higher in south Asians across the glucose spectrum. Absolute CVD risk reductions of up to 13% in those with screen-detected type 2 diabetes and 6% in IGR are achievable using existing cardioprotective therapies.
Conclusions/interpretation
Population screening with an OGTT identifies a significant burden of modifiable CVD risk, especially within south Asian groups. Strategies enticing this population to consider screening programmes are urgently needed as significant risk reduction is possible once a glucose abnormality is identified.
Trial registration:
ClinicalTrials.gov NCT00318032
Funding:
The project is funded for support and treatment costs by NHS Department of Health Support for Science and project grants.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Screening for type 2 diabetes mellitus identifies patients who may have more to gain from aggressive cardiovascular risk management earlier in the disease trajectory [1]. Simultaneously addressing co-existent risk factors in addition to hyperglycaemia should theoretically maximise any benefits of screening, as intensive glucose lowering alone fails to rapidly improve cardiovascular mortality in individuals with established diabetes [2, 3]. Modelling studies suggest that targeted screening for type 2 diabetes is cost effective when the likely vascular benefits of optimised blood pressure and lipid control are also considered [4].
Earlier identification of common risk factor combinations accompanying diabetes, followed by intensive multifactorial intervention, may be required to improve outcomes. The UK Department of Health vascular check programme, aimed at initially screening all 40–70 year olds for hypertension (blood pressure >140/90 mmHg) and hypercholesterolaemia (total cholesterol >5.0 mmol/l, LDL-cholesterol >3.0 mmol/l) in advance of glucose testing, reinforces a model of care centred on screening for vascular risk rather than type 2 diabetes alone [5].
The extent of achievable cardiovascular risk reduction depends not only on the efficacy of available treatments, but also the level of background risk. UK screening policy recommends targeting populations known to be at high risk of diabetes, logically expecting to optimise identification of individuals at greatest risk of cardiovascular disease [6]. Ethnic minority south Asian groups may benefit greatly from such approaches, especially if latent, modifiable cardiovascular risk factors are more prevalent than in white Europeans [7, 8]. It is unknown whether screening for asymptomatic glucose disorders to reduce overall cardiovascular morbidity is feasible within UK populations of south Asian descent.
The Anglo–Danish–Dutch study of Intensive Treatment In peOple with screeN detected Diabetes in Primary Care (ADDITION) [9], a trial in three European countries with a screening phase followed by a cluster randomised controlled trial of intervention based upon the Steno study [10], recently reported its findings [11]. The UK based Leicester arm (ADDITION-Leicester) of the study, is specifically designed to address coronary heart disease (CHD) risk within a multi-ethnic (predominantly Indian south Asian) western population [12].
Here we report the uptake of screening in ADDITION-Leicester and subsequent yield by racial group (white European vs south Asian) of undiagnosed glucose disorders defined as type 2 diabetes and impaired glucose regulation (IGR: impaired fasting glucose [IFG] and/or impaired glucose tolerance [IGT]). We compare the burden of cardiovascular risk among white Europeans and south Asians, and then predict potential cardiovascular risk reduction using systematic multifactorial interventions.
Methods
The ADDITION-Leicester study is comprised of two stages, an initial screening phase followed by a pragmatic, single-blind, cluster randomised, parallel group trial among people with screen-detected type 2 diabetes [12]. Here we consider the screening phase.
Screening procedure
Engaging ethnic minority groups in medical research is challenging and often unpredictable [13, 14]. The design of ADDITION-Leicester paid particular attention to potential cultural sensitivities and barriers to south Asian recruitment [12]. The opinions of prominent lay representatives together with national screening experts were sought in an attempt to construct a protocol tailored to the needs of the local population. Further experiences were drawn from a preceding diabetes screening programme [15]. Attendance was encouraged through culturally sensitive promotions linked to local media (TV, radio, newspapers, flyers), publicity surrounding a mobile screening unit (‘the diabetes bus’), optional community based screening venues and special clinics employing Gujarati interpreters (the Indian dialect spoken by the majority). It was felt that these features were important in gaining the trust and support of the population.
Recruitment
The screening process of ADDITION-Leicester has been described in detail elsewhere [12]. Briefly, 20 local community practices participated in a screening programme inviting a random sample of people between 40 and 75 years inclusive (25–75 years for south Asians) to attend a single session glucose and cardiovascular risk assessment between 2005 and 2008. This included a 75 g OGTT, plasma lipid profile, and standardised blood pressure and anthropometric measurements.
Study measurements
Self-completed questionnaires were used to assess medical history, smoking status and ethnicity. Deprivation level was calculated using the Index of Multiple Deprivation [16]. Body fat percentage was measured via calibrated bioimpedance (Tanita Europe, Amsterdam, the Netherlands). Glucose was analysed in fasting and 2 h post-challenge venous samples via the hexokinase method. HbA1c was analysed using the Bio-rad Variant II HPLC system (Bio-Rad Clinical Diagnostics, Hemel Hempstead, UK). These assays were undertaken in pathology laboratories within the University Hospital of Leicester, UK, and repeat testing carried out if the coefficient of variance was ≥20%. Serum total cholesterol, HDL-cholesterol and triacylglycerol were measured by standard enzymatic techniques. LDL-cholesterol was calculated using Friedewald’s formula. Other biomedical measurements are described in the study protocol [12]
Participants were divided into diabetes, IFG or IGT categories based on current WHO criteria [17]. Here IGR refers to a composite of IFG and/or IGT. Those with results above the diagnostic threshold for diabetes were rescreened within 2 weeks to confirm the diagnosis. Participants with diabetes were subsequently entered into the trial phase of the study.
Cardiovascular risk assessment
Ten-year cardiovascular risk was assessed using two tools. The ETHRISK calculator assesses risk according to sex, age, smoking status, systolic blood pressure, HDL and total cholesterol levels adjusted for ethnicity [18]. The UK Prospective Diabetes Study (UKPDS) score uses age, duration of diabetes, sex, ethnicity, smoking status, systolic blood pressure, HbA1c, total cholesterol and HDL cholesterol [19, 20]. For both tools CVD risk was calculated using the composite of the CHD risk and stroke risk. Subjects with a known history of CVD, including myocardial infarction, stroke and angina, were excluded from CVD risk calculation. ETHRISK was used to assess CVD risk in people with both type 2 diabetes and IGR, whereas the UKPDS was only used for people with diabetes.
Prediction of CVD risk reduction
Given the anticipated high level of CVD risk in the screen-detected diabetes and IGR groups, we assessed the possible achievable CVD risk reduction if these individuals were treated with interventions known to reduce CVD risk. The predicted CVD risk reduction achievable was calculated according to the method used by Echouffo-Tcheugui et al. [21]. We updated the model to reflect the results of recent outcome studies reporting the effects of aspirin and blood glucose lowering in individuals with type 2 diabetes [22, 23]. We also included the recently reported reduction of composite CVD events in the intensive multifactorial intervention arm of the ADDITION-Europe study [11]. Briefly, we estimated the absolute risk reduction (or risk difference) that would be achieved by a preventive approach consisting of prescribing therapies targeting glycaemia, blood pressure and cholesterol for all participants with screen-detected diabetes. We extended this analysis to those with IGR anticipating comparable baseline CVD risk to those with diabetes. The estimation accounted for drugs already prescribed for each patient by assuming that the additional benefit would only be derived from the added classes of medication, but not from any adjustment of existing therapy. The estimation was undertaken under different assumptions on how single cardiovascular risk reductions combine when more than one factor is treated.
Two extreme scenarios were considered: the conservative situation in which combined therapy is only as effective as the most effective single agent and the optimistic scenario in which relative risk reductions combine in a multiplicative manner. For those with diabetes, the relative risks were based on data from a single trial [24–26], a meta-analysis of trials [23, 27] and a combination of estimates from two trials [28, 29]. Given the lack of clinical trial evidence for lipid-lowering and anti-platelet therapy in those with IGR, and the comparable relative risks in those with and without diabetes, we have used the same relative risks in the analysis of the IGR group. However, for blood pressure and glucose lowering in those with IGR, we took relative risk estimates from the Nateglinide and Valsartan in Impaired Glucose Tolerance Outcomes Research (NAVIGATOR) study, a large randomised controlled trial [24, 30]. Two further assumptions were made: (1) that adherence to treatment would be similar in routine practice to that in the trials; and (2) that the risk reductions observed in the populations recruited to the trials could be generalised to people whose diabetes was detected through screening.
Statistical methods
Data were presented as age and sex adjusted means (95% CIs). Non-normally distributed variables (IPAQ physical activity measure [31]) were base log transformed (log e ). South Asians and white Europeans were compared using logistic regression adjusted for age and sex. The prevalences of diabetes, IFG and IGT were compared by ethnicity using logistic regression, first adjusted for age and sex and then adjusted for age, sex, central obesity (using ethnicity specific cut points of waist circumference) and deprivation. Analyses were carried out using Stata (version 11.0) with statistical significance taken at p < 0.05.
For each patient, the absolute CVD risk was computed using the ETHRISK equation in Stata. Simple deterministic sensitivity analyses using extreme ranges (95% CI) of the relative risk reductions shown in Table 1 (based upon [21]) were performed to estimate the plausible range of absolute risk reduction. For each patient we calculated an absolute risk reduction as the product of the absolute risk and the relative risk reduction point estimate from the literature; a maximum reduction as the product of the absolute risk and the upper 95% CI of the relative risk reduction; and a minimum reduction as the product of the absolute risk and the lower 95% CI of the relative risk reduction. The range of reduction was indicated by the medians of the maximum and minimum absolute risk reduction values for all participants without a history of CVD. The number of individuals with screen-detected diabetes needed to treat over a 10-year period to prevent one event was derived as the reciprocal of the median absolute risk reduction. This was then repeated for people with screen-detected IGR.
Results
Screening uptake
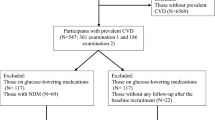
A total of 66,320 patients from 20 practices met the inclusion criteria for ADDITION-Leicester. The median number of eligible patients per practice was 1,996 (range: 707 to 14,895). Of the 30,950 patients randomly invited, 6,749 (22%) were screened (Fig. 1). The median number of patients screened per practice was 299 (range: 16–1,023). To provide a more meaningful comparison, south Asians under 40 years of age (n = 331) were excluded. This revised group consisted of 1,272 Indians (94%), 30 Pakistanis (2.2%), 5 Bangladeshis (0.4%) and 46 from other unspecified south Asian countries (3.4%). The term south Asian is used to describe this predominantly Indo-Asian group.
Table 2 shows the characteristics (age and sex) of people eligible to participate, invited and screened, by ethnic group. Those attending for screening were older compared with both the eligible population and those invited. The south Asian eligible, invited and screened populations were younger than the white European populations, reflecting the differing entry criteria. Even after removal of those aged less than 40, the remaining Asians were on average 5.6 years younger than the white Europeans. More females were screened, although the numbers of males and females eligible and invited were similar. The proportion of males and females screened were similar across ethnicities.
Characteristics of those screened
The demographics and biomedical data of those screened by ethnicity adjusted for age and sex are shown in Table 3. There were marked differences between the white European and south Asian cohorts. The south Asian cohort was more deprived, had lower BMI, waist circumference, total cholesterol, and LDL- and HDL-cholesterol values, was less likely to smoke and was less physically active than the white Europeans, but had higher HbA1c levels, medication use, 10-year modelled CVD risk and a greater frequency of previous CVD. There were no differences in body fat, blood pressure or aspirin use.
Prevalence of abnormal glucose tolerance
Of the 6,041 patients included, 17.5% (n = 1,056) had abnormal glucose tolerance, comprising 3.3% (n = 196) with type 2 diabetes, 2.6% (n = 157) with IFG, 9.7% (n = 585) with IGT, and 2.0% (n = 118) with both IFG and IGT. The south Asian cohort had a significantly higher prevalence of IGT (adjusted OR 1.66, 95% CI 1.33–2.06), IGT or IFG (adjusted OR 1.53, 95% CI 1.26–1.87), IGT and IFG (adjusted OR 1.78, 95% CI 1.12–2.81) and type 2 diabetes (adjusted OR 2.18, 95% CI 1.56–3.06), compared with the white European cohort. The adjusted odds of having any glucose disorder were 1.8 times higher in the south Asians compared with the white Europeans (adjusted OR 1.80, 95% CI 1.52–2.14). High levels of 10-year CVD risk were seen across the abnormal glucose spectrum, ranging from 15.2% in the exclusive IGT white European group to 27.7% in south Asians with screen-detected diabetes. CVD risk was higher in south Asians across all glucose categories (Table 4).
Absolute risk assessment and prediction of its reduction
Table 5 compares the predicted absolute risk reduction and corresponding numbers needed to treat (NNT) in those without previous CVD who have screen-detected diabetes using the ETHRISK CVD risk engine. A statistically significant difference was seen between ethnic groups in terms of 10-year CVD risk (Table 1).
For diabetes, predicted absolute risk reductions ranged from 2.4 to 12.9. Absolute risk reductions were lower for the multifactorial intervention of ADDITION-Europe compared with the additive effects model from published studies; 3.3 (95% CI −0.01, 6.7) compared with 12.9 (95% CI 9.4–15.1) in white Europeans. Predicted absolute risk reductions were generally higher in white Europeans compared with south Asians; for example the conservative scenario gives an absolute risk reduction of 6.3 (95% CI 2.1–9.4) in white Europeans compared with 3.5 (95% CI 2.4–4.6) in south Asians.
Smaller risk reductions were seen for those with screen-detected IGR. A similar pattern of risk reduction was observed with south Asians having lower risk reductions than white Europeans (ranging from 2.5 to 5.9 for white Europeans and 2.2 to 5.2 for south Asians) resulting in larger NNT.
Discussion
Ideally, screening should deliver convincing mortality benefits before it is widely endorsed as a key CVD prevention strategy in type 2 diabetes. Such improvements probably depend upon the extent of risk reduction that could be achieved within the framework of screening. The comparisons and modelling work described here strengthen existing epidemiological data on screening and cardiovascular risk intervention in early glucose disorders. Novel aspects of this paper relate to the feasibility of screening in a multi-ethnic UK population, the characterisation of the risk profile of south Asians attending screening, the burden of CVD risk across the whole spectrum of screen-detected glucose disorders and the likely benefit of pharmacological intervention for CVD risk reduction in non-diabetes range hyperglycaemia.
Screening uptake and prevalence of glucose disorders
Despite concerted attempts to engage the population, the achieved response rate to screening by OGTT was lower than other diabetes screening studies, especially within the south Asian population [32–34]. We consider the cultural adaptations of ADDITION-Leicester a significant strength of the study, which serves to emphasise the complexities of engaging ethnic minority groups in research and health promotion activities. This should be of particular concern to health authorities and future research programmes aiming to improve health outcomes in ethnic minority groups as, despite these measures, the uptake among south Asians was low.
Previous studies adopting stepwise screening approaches with questionnaire based risk scores [35], random capillary testing [36], or a fasting plasma glucose assessment [37] report higher response rates. Assuming more simplistic strategies tend to attract larger numbers, the decision to characterise post-challenge hyperglycaemia and commit the cohort to an arduous OGTT may have affected our response rate. Whilst acknowledging this as a limitation of the study, the impracticality of screening via this method highlights the importance of capturing IGT or isolated post-challenge hyperglycaemia in other ways, for example via use of HbA1c, which is now widely included as a diagnostic test. As has been shown in previous studies, these classifications are particularly common in south Asians, and are associated with significant, often untreated, cardiovascular risk comparable to newly diagnosed type 2 diabetes [38].
Although the achieved response may influence our prevalence estimates for newly diagnosed type 2 diabetes and IGR, this source of error is unavoidable when reporting undiagnosed cases within population settings. Typical of previous diabetes screening studies, responders were older with a female preponderance which probably reflects the ability of these groups to commit to a morning 3 h appointment. More worryingly it may also indicate a generalised apathy towards screening within perceived ‘healthy’ populations. An unexpected difference in the mean age of eligible and invited populations may relate to discrepancies in the practice-sourced eligibility data and subsequent independent mailing searches conducted by the research team. This may have influenced random sampling and should be considered a potential source of error affecting an otherwise highly representative population. We also acknowledge that it may have been more appropriate to use a tool measuring an individual’s deprivation rather than the geographically determined index employed.
After age and sex adjustment, the odds ratios for undiagnosed glucose disorders, whether fasting or post-challenge, were twice as high in south Asians. A Leicester based study has previously reported similar findings within diagnosed diabetes cases and our data extend this trend into undiagnosed diabetes and IGR ranges [39]. A prevalence of 4.5% for undiagnosed type 2 diabetes is similar to the Indian ethnic boost sample of the Health Survey of England [40].
Whilst the prevalence of IGR was expected to be 15–20%, previously undiscovered diabetes frequency was lower than anticipated in both groups. Although this observation could be partly explained by the sampling and ascertainment issues already described, it is also plausible that the number of undiagnosed cases is in decline as a greater effort is made to identify the condition. Although designed prior to its conception, the last year of the screening phase in ADDITION-Leicester coincided with the implementation of the NHS pay-for-performance quality and outcomes framework (QoF) [41]. This centrally devolved strategy rewards general practitioners for identifying and maintaining good glycaemic control in people with type 2 diabetes and may have contributed to increased opportunistic screening activity in primary care over recent years. Our results may reflect a declining population prevalence of undiagnosed diabetes in centres more actively engaged in earlier detection practices. It may also simply be that this relatively discrete Indian population is not as susceptible to glucose disorders as other diasporic south Asian groups.
Cardiovascular risk in south Asians
There have been a number of UK based surveys quantifying cardiovascular risk within dispersed south Asian communities. To date the largest remains the Southall Diabetes study, an ongoing programme describing traditional risk factors and incident vascular events in 3,000 Bangladeshis and white Europeans [42–44]. Similar to our findings, the initial cross-sectional analysis of this cohort demonstrated a lower mean plasma cholesterol and smoking tendency in combination with a much higher prevalence of diabetes in south Asians (19%). Consistently higher rates of CHD in prospective analyses have lead to the assumption that hyperglycaemia and a predisposition to central obesity and its pro-inflammatory consequences probably account for excess vascular disease in this group [44]. Similarly, the more recent Newcastle Heart project used a robust population based approach to identify new cases of diabetes and compare cardiovascular risk in a mixed ethnicity sample of 325 south Asians and 425 white Europeans [45]. This study employed the OGTT and found more IGT and diabetes in Indians (approximately twofold), Pakistanis (twofold) and Bangladeshis (more than threefold), together with higher rates of CHD compared with white Europeans.
The major finding of these studies is replicated here, with an increased CHD risk and particular predisposition to glucose disorders characterising our Indo-Asian population. Whilst there have undoubtedly been advances in the awareness and management of cardiovascular disease over the last 20 years, the intervening period since publication of the Southall study appears not to have significantly changed the risk profile of south Asians residing in the UK. The time may have come to consider changing largely reactive current therapeutic approaches and develop culturally sensitive screening programmes aimed at much earlier identification of glucose disorders and vascular disease prevention. Implementation of interventions in people with screen-detected non-diabetic hyperglycaemia may be particularly advantageous in ethnic minority populations known to be susceptible to type 2 diabetes and CHD [46].
Burden of CVD risk in a screened population
This study reflects others demonstrating a significant burden of CVD risk in responders to population based screening programmes for diabetes [1, 35, 47, 48]. In contrast to other programmes, ADDITION-Leicester provides additional information on CVD risk burden relating specifically to IGT. Importantly, CVD risk appears amplified by any degree of glucose impairment, as illustrated by the similarity between 10-year global CVD risk estimates in IGR and screen-detected diabetes (ETHRISK 18.0% vs 21.1%).
CVD modelled risk reduction
Our modelling suggests that significant CVD risk reduction using existing cardioprotective therapies (up to 12%) would be achievable within the diabetes, and to a lesser extent the IGR, range (up to 6%). Interestingly, the effect is attenuated when risk reduction achieved for the multifactorial intervention of the ADDITION-Europe study is considered. This may reflect lower CVD risk in screen-detected ‘newly diagnosed’ diabetes compared with ‘established’ conventionally diagnosed cases typically recruited in earlier intervention studies.
A particular strength of this data is the accurate capture of IGR in combination with relative risk estimates extracted from recently published IGR specific intervention trials. Identifying cases at an even earlier stage of disease by screening below the diabetes diagnostic level would appear to have beneficial effects and increase the time available to achieve effective CVD risk modification. These effects would appear to be equally beneficial to both south Asians and white Europeans within IGR and diabetes ranges. The effect of diabetes treatment in IGR was based on a pharmacological intervention using nateglinide [24, 30]. Nateglinide was chosen as a representative example illustrating the potential effects of multifactorial approaches to CVD prevention, and should not be regarded as advocating the use of this particular drug in IGR. In fact, our estimates of CVD risk reduction using nateglinide are conservative, given that this drug did not significantly reduce the incidence of CVD events or diabetes in the 5-year NAVIGATOR trial. CVD risk or diabetes incidence reduction might be higher if lifestyle interventions [49], or possibly other drugs such as acarbose [50], were to be used in practice. However, meaningful translation of intensive lifestyle interventions showing benefit in the research setting into everyday clinical practice remains a major challenge. Future work will aim to incorporate the CVD effects of emerging lifestyle interventions representative of ‘real world’ IGR populations. Whilst inferring benefit, it must also be acknowledged that modelling analyses of this kind make a number of key assumptions with respect to the applicability of data across populations and are therefore no substitute for randomised controlled trial evidence.
Conclusion
A screening strategy for type 2 diabetes incorporating an OGTT captures a significant and potentially reversible burden of CVD risk in a multi-ethnic UK population. Susceptibility to these complications is potentiated within WHO defined glucose disorders with similar CVD risks for impaired glucose regulation and screen-detected diabetes categories. South Asian people attending the programme have approximately twice the odds of an undetected glucose abnormality and significantly greater overall CVD risk, yet are more likely to be prescribed statin or anti-hypertensive drugs than their white European counterparts. Novel culturally sensitive approaches to cardiovascular health screening may be needed as unidentified traditional CVD risk factors remain prevalent within this group.
Abbreviations
- ADDITION:
-
Anglo–Danish–Dutch study of Intensive Treatment In peOple with screeN detected diabetes in primary care
- CHD:
-
Coronary heart disease
- CVD:
-
Cardiovascular disease
- IFG:
-
Impaired fasting glycaemia
- IGT:
-
Impaired glucose tolerance
- IGR:
-
Impaired glucose regulation (IFG and/or IGT)
- NAVIGATOR:
-
Nateglinide and Valsartan in Impaired Glucose Tolerance Outcomes Research
- NNT:
-
Number needed to treat
- UKPDS:
-
UK Prospective Diabetes Study
References
Sandbaek A, Griffin SJ, Rutten G et al (2008) Stepwise screening for diabetes identifies people with high but modifiable coronary heart disease risk. The ADDITION study. Diabetologia 51:1127–1134
Duckworth W, Abraira C, Moritz T et al (2009) Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 360:129–139
The ADVANCE, Collaborative G (2008) Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 358:2560–2572
Waugh N, Scotland G, McNamee P (2007) Screening for type 2 diabetes: literature review and economic modelling. Health Technology Assessment 11: iii–iv, ix–xi, 1–125
Department of Health (2009) Putting prevention first. NHS Health Check: vascular risk assessment and management. Gateway Reference 11473. Available from www.dh.gov.uk/prod_consum_dh/groups/dh_digitalassets/documents/digitalasset/dh_098410.pdf. Accessed 1 April 2011
Department of Health (1998) RCGP summary paper: first report of the National Screening Committee. DoH, London
Riste L, Khan F, Cruickshank K (2001) High prevalence of type 2 diabetes in all ethnic groups, including Europeans, in a British inner city: relative poverty, history, inactivity, or 21st century Europe? Diabetes Care 24:1377–1383
Forouhi NG, Merrick D, Goyder E et al (2006) Diabetes prevalence in England, 2001—estimates from an epidemiological model. Diabet Med 23:189–197
Lauritzen T, Griffin S, Borch-Johnsen K, Wareham NJ, Wolffenbuttel BHR, Rutten G (2000) The ADDITION study: proposed trial of the cost-effectiveness of an intensive multifactorial intervention on morbidity and mortality among people type 2 diabetes detected by screening. Int J Obes 24(Suppl 3):S6–S11
Gaede P, Vedel P, Larsen N, Jensen GVH, Parving H-, Pedersen O (2003) Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 348:383–393
Griffin SJ, Borch-Johnsen K, Wareham NJ, et al (2011) A randomised trial of the effect of early intensive multifactorial therapy on 5-year cardiovascular outcomes in individuals with screen-detected type 2 diabetes: the ADDITION-Europe study. Lancet (in press)
Webb D, Khunti K, Srinivasan B et al (2010) Rationale and design of the ADDITION-Leicester study, a systematic screening programme and randomised controlled trial of multi-factorial cardiovascular risk intervention in people with type 2 diabetes mellitus detected by screening. BMC Trials 11:16
Khunti K, Kumar S, Brodie J (2009) Diabetes UK and South Asian Health Foundation Recommendations on Diabetes Research Priorities for British South Asians, 1st edn. Diabetes UK, London
Mason S, Hussain-Gambles M, Leese B, Atkin K, Brown J (2003) Representation of South Asian people in randomised clinical trials: analysis of trials’ data. BMJ 326:1244–1245
Gray LJ, Tringham JR, Davies MJ et al (2010) Screening for type 2 diabetes in a multiethnic setting using known risk factors to identify those at high risk: a cross-sectional study. Vasc Health Risk Manage 6:837–842
Communities and Local Government (2007) The English Indices of Deprivation 2004 (revised). Available from www.communities.gov.uk/publications/communities/englishindices. Accessed 1 April 2011
Alberti KGMM, Zimmet PZ (1998) Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabet Med 15:539–553
Brindle P, May M, Gill P et al (2006) Primary prevention of cardiovascular disease: a web-based risk score for seven British black and minority ethnic groups. Heart 92:1595–1602
Kothari V, Stevens RJ, Adler AI, Stratton IM, Manley SE, Andrew Neil H et al (2002) UKPDS 60: risk of stroke in type 2 diabetes estimated by the UK Prospective Diabetes Study risk engine. Stroke 33:1776–1781
Stevens RJ, Kothari V, Adler AI, Stratton IM, Holman RR (2001) The UKPDS risk engine: a model for the risk of coronary heart disease in type II diabetes (UKPDS 56). Clin Sci 101:671–679
Echouffo-Tcheugui JB, Sargeant LA, Prevost AT et al (2008) How much might cardiovascular disease risk be reduced by intensive therapy in people with screen-detected diabetes? Diabet Med 25:1433–1439
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HAW (2008) 10-Year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 359:1577–1589
Antithrombotic Trialists’ (ATT) Collaboration (2009) Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet 373:1849–1860
Holman RR, Haffner SM, McMurray JJ et al (2010) Effect of nateglinide on the incidence of diabetes and cardiovascular events. N Engl J Med 362:1463–1476
UK Prospective Diabetes Study Group (1998) Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ 317:703–713
UK Prospective Diabetes Study (UKPDS) study group (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352:837–853
Antithrombotic Trialists’ (ATT) Collaboration (2002) Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 324:71–86
Heart Protection Study Collaborative Group (2003) MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5,963 people with diabetes: randomised placebo-controlled trial. Lancet 361:2005–2016
Colhoun HM, Betteridge DJ, Durrington PN et al (2004) Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomised placebo-controlled trial. Lancet 364:685–696
Califf RM, McMurray JJ, Holman RR et al (2010) Effect of valsartan on the incidence of diabetes and cardiovascular events. N Engl J Med 362:1477–1490
Craig CL, Marshall AL, Sjostrom M et al (2003) International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc 35:1381–1395
Lawrence JM, Bennett P, Young A, Robinson AM (2001) Screening for diabetes in general practice: cross sectional population study. BMJ 323:548–551
Christensen JO, Sandbaek A, Lauritzen T, Borch-Johnsen K (2004) Population-based stepwise screening for unrecognised Type 2 diabetes is ineffective in general practice despite reliable algorithms. Diabetologia 47:1566–1573
Janssen PG, Gorter KJ, Stolk RP, Akarsubasi M, Rutten GE (2009) Three years follow-up of screen-detected diabetic and non-diabetic subjects: who is better off? The ADDITION Netherlands study. BMC Fam Pract 9:67
Spijkerman AM, Adriaanse MC, Dekker JM et al (2002) Diabetic patients detected by population-based stepwise screening already have a diabetic cardiovascular risk profile. Diabetes Care 25:1784–1789
Sargent LA, Simmons RK, Barling RS et al (2010) Who attends a UK diabetes screening programme? Findings from the ADDITION-Cambridge study. Diabet Med 27:995–1003
Bullimore SP, Keyworth C (1997) Finding diabetics—a method of screening in general practice. Br J Gen Pract 47:371–373
Joshi P, Islam S, Pais P et al (2007) Risk factors for early myocardial infarction in South Asians compared with individuals in other countries. J Am Med Assoc 297:286–294
Samanta A, Burden AC, Fent B (1987) Comparative prevalence of non-insulin-dependent diabetes mellitus in Asian and white Caucasian adults. Diab Res Clin Pract 4:1–6
NHS: The Information Centre (2006) Health survey for England 2004: health of ethnic minorities—full report. Available from www.ic.nhs.uk/pubs/hse04ethnic. Accessed 1 April 2011
Department of Health (2004) Quality and Outcomes Framework (QoF): guidance. Department of Health, London
McKeigue PM, Shah B, Marmot MG (1991) Relation of central obesity and insulin resistance with high diabetes prevalence and cardiovascular risk in South Asians. Lancet 337:382–386
McKeigue PM, Ferrie JE, Pierpoint T, Marmot MG (1993) Association of early-onset coronary heart disease in South Asian men with glucose intolerance and hyperinsulinemia. Circulation 87:152–161
Forouhi NG, Sattar N, Tillin T, McKeigue PM, Chaturvedi N (2006) Do known risk factors explain the higher coronary heart disease mortality in South Asian compared with European men? Prospective follow-up of the Southall and Brent studies, UK. Diabetologia 49:2580–2588
Bhopal R, Unwin N, White M et al (1999) Heterogeneity of coronary heart disease risk factors in Indian, Pakistani, Bangladeshi, and European origin populations: cross sectional study. BMJ 319:215–220
Gholap N, Davies MJ, Patel K, Sattar N, Khunti K (2011) Type 2 diabetes mellitus and cardiovascular disease in South Asians. Prim Care Diab. doi:10.1016/j.pcd.2010.08.002
Jorgensen T, Borch-Johnsen K, Thomsen TF, Ibsen H, Glumer C, Pisinger C (2003) A randomized non-pharmacological intervention study for prevention of ischaemic heart disease: baseline results Inter99. Eur J Cardiovasc Prev Rehabil 10:377–386
de Vegt F, Dekker JM, Ruhe HG et al (1999) Hyperglycaemia is associated with all-cause and cardiovascular mortality in the Hoorn population: the Hoorn study. Diabetologia 42:926–931
Knowler WC, Barrett-Connor E, Fowler SE et al (2002) Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 346:393–403
Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M (2003) Acarbose treatment and the risk of cardiovascular disease and hypertension in patients with impaired glucose tolerance: the STOP-NIDDM trial. J Am Med Assoc 290:486–494
Acknowledgements
We are greatly indebted to the 20 general practices participating in the study together with the entire diabetes team in Leicester. We would like to thank the Novo Nordisk Research Foundation for supporting D. R. Webb with a competitive research fellowship. We also acknowledge the contribution of the following individuals to the success of the project: E. Healey, J Stockman, M Radia, N Radia, F Ablett and S Sehmi. The project is funded for support and treatment costs by NHS Department of Health Support for Science and project grants. The study is hosted by the University Hospitals of Leicester, UK, and registered with an internationally recognised trials registry.
Duality of interest
M. J. Davies has received funds for research, honoraria for speaking at meetings and has served on advisory boards for Eli Lilly, sanofi-aventis, MSD and Novo Nordisk. K. Khunti is an advisory board member for Novo Nordisk, Novartis, Eli Lilly, MSD and Boehringer Ingelheim; has received lecture fees from Novo Nordisk, Eli Lilly, sanofi-aventis, Novartis, Takeda and MSD; and has received funds for research from Novo Nordisk, Eli Lilly, Novartis, Roche and MSD. S. J. Griffin has received speaker fees from Eli Lilly, GSK, MSD, Colgate Palmolive and Unilever, and research support from Novo Nordisk. N. J. Wareham has served on advisory panels for Unilever and GSK, and received support from Novo Nordisk. All other authors have nothing to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Webb, D.R., Gray, L.J., Khunti, K. et al. Screening for diabetes using an oral glucose tolerance test within a western multi-ethnic population identifies modifiable cardiovascular risk: the ADDITION-Leicester study. Diabetologia 54, 2237–2246 (2011). https://doi.org/10.1007/s00125-011-2189-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-011-2189-2