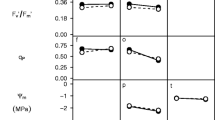
Abstract
Key message
Quantitation of leaf surface wax on a population of switchgrass identified three significant QTL present across six environments that contribute to leaf glaucousness and wax composition and that show complex genetic × environmental (G × E) interactions.
Abstract
The C4 perennial grass Panicum virgatum (switchgrass) is a native species of the North American tallgrass prairie. This adaptable plant can be grown on marginal lands and is useful for soil and water conservation, biomass production, and as a forage. Two major switchgrass ecotypes, lowland and upland, differ in a range of desirable traits, and the responsible underlying loci can be localized efficiently in a pseudotestcross design. An outbred four-way cross (4WCR) map** population of 750 F2 lines was used to examine the genetic basis of differences in leaf surface wax load between two lowland (AP13 and WBC) and two upland (DAC and VS16) tetraploid cultivars. The objective of our experiments was to identify wax compositional variation among the population founders and to map underlying loci responsible for surface wax variation across environments. GCMS analyses of surface wax extracted from 4WCR F0 founders and F1 hybrids reveal higher levels of wax in lowland genotypes and show quantitative differences of β-diketones, primary alcohols, and other wax constituents. The full map** population was sampled over two seasons from four field sites with latitudes ranging from 30 to 42 °N, and leaf surface wax was measured. We identified three high-confidence QTL, of which two displayed significant G × E effects. Over 50 candidate genes underlying the QTL regions showed similarity to genes in either Arabidopsis or barley known to function in wax synthesis, modification, regulation, and transport.





Similar content being viewed by others
Availability of data and material
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Barthlott W, Neinhuis C, Cutler D et al (1998) Classification and terminology of plant epicuticular waxes. Bot J Linn Soc 126:237–260. https://doi.org/10.1111/j.1095-8339.1998.tb02529.x
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bianchi G, Avato P, Salamini F (1979) Glossy mutants of maize. Heredity 42:391–395. https://doi.org/10.1038/hdy.1979.42
Brunken JN, Estes JR (1975) Cytological and morphological variation in Panicum virgatum L. Southwest Nat 19:379–385. https://doi.org/10.2307/3670396
Burow GB, Franks CD, **n Z (2008) Genetic and physiological analysis of an irradiated bloomless mutant (epicuticular wax mutant) of sorghum. Crop Sci 48:41–48. https://doi.org/10.2135/cropsci2007.02.0119
Buschhaus C, Jetter R (2012) Composition and physiological function of the wax layers coating arabidopsis leaves: β-amyrin negatively affects the intracuticular water barrier. Plant Physiol 160:1120–1129. https://doi.org/10.1104/pp.112.198473
Buschhaus C, Herz H, Jetter R (2007) Chemical composition of the epicuticular and intracuticular wax layers on adaxial sides of Rosa canina leaves. Ann Bot 100:1557–1564. https://doi.org/10.1093/aob/mcm255
Casler MD (2012) Switchgrass breeding, genetics, and genomics. In: Monti A (ed) Switchgrass: a valuable biomass crop for energy. Springer, London, pp 29–53
Core Team R (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ebercon A, Blum A, Jordan WR (1977) A rapid colorimetric method for epicuticular wax content of sorghum leaves. Crop Sci 17:179–180. https://doi.org/10.2135/cropsci1977.0011183X001700010047x
Eberhart SA, Newell LC (1959) Variation in domestic collections of switchgrass, Panicum virgatum L 1. Agron J 51:613–616. https://doi.org/10.2134/agronj1959.00021962005100100015x
Eigenbrode SD, Espelie KE (1995) Effects of plant epicuticular lipids on insect herbivores. Annu Rev Entomol 40:171–194. https://doi.org/10.1146/annurev.en.40.010195.001131
El-Otmani M, Arpaia ML, Coggins CW et al (1989) Developmental changes in ‘Valencia’ orange fruit epicuticular wax in relation to fruit position on the tree. Sci Hortic 41:69–81. https://doi.org/10.1016/0304-4238(89)90051-4
Gülz P-G, Boor G (2014) Seasonal variations in epicuticular wax ultrastructures of Quercus robur leaves. Z Für Naturforschung C 47:807–814. https://doi.org/10.1515/znc-1992-11-1205
Hallam ND, Chambers TC (1970) The leaf waxes of the genus Eucalyptus L’Héritier. Aust J Bot 18:335–386. https://doi.org/10.1071/bt9700335
Hen-Avivi S, Savin O, Racovita RC et al (2016) A metabolic gene cluster in the wheat w1 and the barley cer-cqu loci determines β-diketone biosynthesis and glaucousness. Plant Cell 28:1440–1460. https://doi.org/10.1105/tpc.16.00197
Holloway PJ (1969) The effects of superficial wax on leaf wettability. Ann Appl Biol 63:145–153. https://doi.org/10.1111/j.1744-7348.1969.tb05475.x
Holmes MG, Keiller DR (2002) Effects of pubescence and waxes on the reflectance of leaves in the ultraviolet and photosynthetic wavebands: a comparison of a range of species. Plant, Cell Environ 25:85–93. https://doi.org/10.1046/j.1365-3040.2002.00779.x
Jeffree CE, Baker EA, Holloway PJ (1975) Ultrastructure and Recrystallization of plant epicuticular waxes. New Phytol 75:539–549. https://doi.org/10.1111/j.1469-8137.1975.tb01417.x
Koch K, Ensikat H-J (2008) The hydrophobic coatings of plant surfaces: epicuticular wax crystals and their morphologies, crystallinity and molecular self-assembly. Micron 39:759–772. https://doi.org/10.1016/j.micron.2007.11.010
Koch K, Barthlott W, Koch S et al (2006) Structural analysis of wheat wax (Triticum aestivum, c.v. ‘Naturastar’ L.): from the molecular level to three dimensional crystals. Planta 223:258–270. https://doi.org/10.1007/s00425-005-0081-3
Koornneef M, Hanhart CJ, Thiel F (1989) A genetic and phenotypic description of eceriferum (cer) mutants in Arabidopsis thaliana. J Hered 80:118–122. https://doi.org/10.1093/oxfordjournals.jhered.a110808
Lenth R (2020) emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.4.8. https://CRAN.R-project.org/package=emmeans
Lovell J, Healy A, Schmutz J, Juenger, T (2020) Switchgrass v5 4-way (AP13 × DAC, WBC × VS16) genetic map v2, Dryad, Dataset. https://doi.org/10.5061/dryad.ghx3ffbjv
Lowry DB, Taylor SH, Bonnette J et al (2015) QTLs for biomass and developmental traits in switchgrass (Panicum virgatum). Bioenerg Res 8(4):1856–1867. https://doi.org/10.1007/s12155-015-9629-7
Lowry DB, Lovell JT, Zhang L et al (2019) QTL × environment interactions underlie adaptive divergence in switchgrass across a large latitudinal gradient. Proc Natl Acad Sci 116:12933–12941. https://doi.org/10.1073/pnas.1821543116
Lundqvist U, Wettstein DV (1962) Induction of eceriferum mutants in barley by ionizing radiations and chemical mutagens. Hereditas 48:342–362. https://doi.org/10.1111/j.1601-5223.1962.tb01818.x
Luo Z, Tomasi P, Fahlgren N, Abdel-Haleem H (2019) Genome-wide association study (GWAS) of leaf cuticular wax components in Camelina sativa identifies genetic loci related to intracellular wax transport. BMC Plant Biol 19:187. https://doi.org/10.1186/s12870-019-1776-0
Macey MJK (1970) The effect of light on wax synthesis in leaves of Brassica oleracea. Phytochemistry 9:757–761. https://doi.org/10.1016/S0031-9422(00)85178-X
Malosetti M, Ribaut J-M, van Eeuwijk FA (2013) The statistical analysis of multi-environment data: modeling genotype-by-environment interaction and its genetic basis. Front Physiol. https://doi.org/10.3389/fphys.2013.00044
Milano ER, Lowry DB, Juenger TE (2016) The genetic basis of upland/lowland ecotype divergence in switchgrass (Panicum virgatum). G3 Genes Genomes Genet 6:3561–3570. https://doi.org/10.1534/g3.116.032763
Nødskov Giese B (1975) Effect of light and temperature on the composition of epicuticular wax of barley leaves. Phytochemistry 14:921–929. https://doi.org/10.1016/0031-9422(75)85160-0
Riederer M, Schreiber L (2001) Protecting against water loss: analysis of the barrier properties of plant cuticles. J Exp Bot 52:2023–2032. https://doi.org/10.1093/jexbot/52.363.2023
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Schneider LM, Adamski NM, Christensen CE et al (2016) The Cer-cqu gene cluster determines three key players in a β-diketone synthase polyketide pathway synthesizing aliphatics in epicuticular waxes. J Exp Bot. https://doi.org/10.1093/jxb/erw105
Serrano M, Coluccia F, Torres M et al (2014) The cuticle and plant defense to pathogens. Front Plant Sci. https://doi.org/10.3389/fpls.2014.00274
Smith LL, Smith KE (2000) United States Patent: PP11202 - Panicum plant named ‘Dallas Blues’. http://patft.uspto.gov/netacgi/nph-Parser?Sect1=PTO1&Sect2=HITOFF&d=PALL&p=1&u=%2Fnetahtml%2FPTO%2Fsrchnum.htm&r=1&f=G&l=50&s1=PP11202.PN.&OS=PN/PP11202&RS=PN/PP11202. Accessed 12 Aug 2020
Tulloch AP, Hoffman LL (1980) Epicuticular wax of Panicum virgatum. Phytochemistry 19:837–839. https://doi.org/10.1016/0031-9422(80)85122-3
Uddin MN, Marshall DR (1988) Variation in epicuticular wax content in wheat. Euphytica 38:3–9. https://doi.org/10.1007/BF00024805
Valladares F, Sanchez-Gomez D, Zavala MA (2006) Quantitative estimation of phenotypic plasticity: bridging the gap between the evolutionary concept and its ecological applications. J Ecol 94:1103–1116. https://doi.org/10.1111/j.1365-2745.2006.01176.x
VanWallendael A, Bonnette J, Juenger TE, et al (2020) Geographic variation in the genetic basis of resistance to leaf rust between locally adapted ecotypes of the biofuel crop switchgrass (Panicum virgatum). bioRxiv 619148. https://doi.org/10.1101/619148
von Wettstein-Knowles P (1974) Ultrastructure and origin of epicuticular wax tubes. J Ultrastruct Res 46:483–498. https://doi.org/10.1016/S0022-5320(74)90069-0
von Wettstein‐Knowles P(2016) Plant Waxes. In: eLS. American Cancer Society, pp 1–13
VSN International (2019) Genstat for Windows, 20th edn. VSN International, Indore
Wang Y, Wang J, Chai G et al (2015) Developmental changes in composition and morphology of cuticular waxes on leaves and spikes of glossy and glaucous wheat (Triticum aestivum L.). PLoS ONE 10:e0141239. https://doi.org/10.1371/journal.pone.0141239
Weaver JM, Lohrey G, Tomasi P et al (2018) Cuticular wax variants in a population of switchgrass (Panicum virgatum L.). Ind Crops Prod 117:310–316. https://doi.org/10.1016/j.indcrop.2018.02.081
Yeats TH, Rose JKC (2013) The formation and function of plant cuticles. Plant Physiol 163:5–20. https://doi.org/10.1104/pp.113.222737
Acknowledgements
Mention of trade names or commercial products in this publication is solely for the purpose of specific information and does not imply recommendation or endorsement by the US Department of Agriculture. We would like to acknowledge the following people: Lia Poasa for assistance with the care of the plants in the greenhouse; Sheyla Aucar, Selina Chen, and April Nguyen for help with colorimetric wax measurements; Melissa Stefanek and Kyren Tomasi for their laboratory assistance with sample processing and calibration curve development for compound quantifications. We thank the Joint Genome Institute and collaborators for prepublication access to the Panicum virgatum AP13 genome reference. Funding was provided by Agricultural Research Service (Grant Nos. 2020-21410-007-00-D, 2030-21430-014-00-D), National Science Foundation (Grant Nos. IOS-0922457, IOS-1444533), U.S. Department of Energy (Grant Nos. DE-SC0014156, DE-AC02-05CH11231).
Author information
Authors and Affiliations
Contributions
JB collected and analyzed data as well as wrote the manuscript. PT performed wax compositional analyses. JL, JS, and AH created the 4WCR genetic map. LZ performed QTL analysis and co-wrote the manuscript. CT assisted with wax quantitation, co-wrote the manuscript, and coordinated the study. LC and PC performed wax quantitation, DW and TW conducted the SEM, JT coordinated the research and co-wrote the manuscript, and JB coordinated field sampling.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Additional information
Communicated by Herman J. van Eck.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bragg, J., Tomasi, P., Zhang, L. et al. Environmentally responsive QTL controlling surface wax load in switchgrass. Theor Appl Genet 133, 3119–3137 (2020). https://doi.org/10.1007/s00122-020-03659-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-020-03659-0