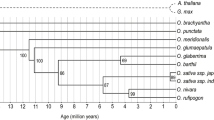
Abstract
Rice blast, caused by the fungal pathogen Magnaporthe oryzae, is a devastating disease of rice worldwide. Among the 85 mapped resistance (R) genes against blast, 13 have been cloned and characterized. However, how these genes originated and how they evolved in the Oryza genus remains unclear. We previously cloned the rice blast R-genes Pi2, Pi9, and Piz-t, and analyzed their genomic structure and evolution in cultivated rice. In this study, we determined the genomic sequences of the Pi2/9 locus in four wild Oryza species representing three genomes (AA, BB and CC). The number of Pi2/9 family members in the four wild species ranges from two copies to 12 copies. Although these genes are conserved in structure and categorized into the same subfamily, sequence duplications and subsequent inversions or uneven crossing overs were observed, suggesting that the locus in different wild species has undergone dynamic changes. Positive selection was found in the leucine-rich repeat region of most members, especially in the largest clade where Pi9 is included. We also provide evidence that the Pi9 gene is more related to its homologues in the recurrent line and other rice cultivars than to those in its alleged donor species O. minuta, indicating a possible origin of the Pi9 gene from O. sativa. Comparative sequence analysis between the four wild Oryza species and the previously established reference sequences in cultivated rice species at the Pi2/9 locus has provided extensive and unique information on the genomic structure and evolution of a complex R-gene cluster in the Oryza genus.





Similar content being viewed by others
References
Amante-Bordeos A, Sitch LA, Nelson R, Damacio RD, Oliva L, Aswidinnoor H, Leung H (1992) Transfer of bacterial blight and blast resistance from the tetraploid wild rice Oryza minuta to cultivated rice, Oryza sativa. Theor Appl Genet 84:345–354
Ameline-Torregrosa CB, Wang B, O’Bleness MS, Deshpande S, Zhu H, Roe B, Young ND, Cannon SB (2008) Identification and characterization of nucleotide-binding site-leucine-rich repeat genes in the model plant Medicago truncatula. Plant Physiol 146:5–21
Ammiraju JSS, Lu F, Sanyal A, Yu Y, Song X, Jiang J, Pontaroli AC, Rambo T, Currie J, Collura K, Talag J, Fan C, Goicoechea JL, Zuccolo A, Chen J, Bennetzen JL, Chen M, Jackson S, Wing RA (2008) Dynamic evolution of Oryza genomes is revealed by comparative genomic analysis of a genus-wide vertical data set. Plant Cell 20:3191–3209
Bai JL, Pennill A, Ning J, Lee SW, Ramalingam J, Webb CA, Zhao B, Sun Q, Nelson JC, Leach JE, Hulbert SH (2002) Diversity in nucleotide binding site-leucine-rich repeat genes in cereals. Genome Res 12:1871–1884
Ballini E, Morel JB, Droc G, Price A, Courtois B, Notteghem JL, Tharreau D (2008) A genome-wide meta-analysis of rice blast resistance genes and quantitative trait loci provides new insights into partial and complete resistance. Mol Plant Microbe Interact 21:859–868
Caracuel-Rios Z, Talbot NJ (2007) Cellular differentiation and host invasion by the rice blast fungus Magnaporthe grisea. Curr Opin Microbiol 10:339–345
Dodds PN, Lawrence GJ, Ellis JG (2001) Six amino acid changes confined to the leucine-rich repeat β-strand/β-turn motif determine the difference between the P and P2 rust resistance specificities in flax. Plant Cell 13:163–178
Ellis JG, Lawrence GJ, Luck JE, Dodds PN (1999) Identification of regions in alleles of the flax rust resistance gene L that determine differences in gene-for-gene specificity. Plant Cell 11:495–506
Ewing B, Green P (1998) Base calling of automated sequencer traces using Phred II. Error probabilities. Genome Res 8:186–194
Ewing B, Hillier L, Wendl M, Green P (1998) Base calling of automated sequencer traces using Phred I. Accuracy assessment. Genome Res 8:175–185
Fukuoka S, Saka N, Koga H, Ono K, Shimizu T, Ebana K, Hayashi N, Takahashi H, Hirochika A, Okuno K, Yano M (2009) Loss of function of a proline-containing protein confers durable disease resistance in rice. Science 325:998–1001
Geffroy V, Macadre C, David P, Pedrosa-Harand A, Sevignac M, Dauga C, Langin T (2009) Molecular analysis of a large subtelomeric nucleotide-binding-site-leucine-rich-repeat family in two representative genotypes of the major gene pools of Phaseolus vulgaris. Genetics 181:405–419
Gordon D, Abajian C, Green P (1998) Consed, a graphical tool for sequence finishing. Genome Res 8:195–202
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Series 41:95–98
Hichey DA, Bally-Cuif L, Abukashawa S, Payant V, Benkel BF (1991) Concerted evolution of duplicated protein-coding genes in Drosophila. Proc Natl Acad Sci USA 88:1611–1615
Huang CL, Hwang SY, Chiang YC, Lin TP (2008) Molecular evolution of the Pi-ta gene resistant to rice blast in wild rice (Oryza rufipogon). Genetics 179:1527–1538
Hulbert SH, Webb CA, Smith SM, Sun Q (2001) Resistance gene complexes: evolution and utilization. Annu Rev Phytopathol 39:285–312
Kruijt M, Brandwagt BF, De Wit PJGM (2004) Rearrangements in the Cf-9 disease resistance gene cluster of wild tomato have resulted in three genes that mediate Avr9 responsiveness. Genetics 168:1655–1663
Kumar S, Tamura K, Jakobsen IB, Nei M (2001) MEGA2: molecular evolutionary genetics analysis software. Bioinformatics 17:1244–1245
Lee S, Costanzo S, Jia Y, Olsen KM, Caicedo AL (2009) Evolutionary dynamics of the genomic region around the blast resistance gene Pi-ta in AA genome Oryza Species. Genetics 183:1315–1325
Leister D (2004) Tandem and segmental gene duplication and recombination in the evolution of plant disease resistance gene. Trends Genet 20:116–122
Liu J, Liu X, Dai L, Wang G (2007) Recent progress in elucidating the structure, function and evolution of disease resistance genes in plants. J Genet Genomics 34:765–776
Lu F, Ammiraju JS, Sanyal A, Zhang S, Song R, Chen J, Li G, Sui Y, Song X, Cheng Z, de Oliveira AC, Bennetzen JL, Jackson SA, Wing RA, Chen M (2009) Comparative sequence analysis of MONOCULM1-orthologous regions in 14 Oryza genomes. Proc Natl Acad Sci USA 106:2071–2076
Ma J, Bennetzen JL (2004) Rapid recent growth and divergence of rice nuclear genomes. Proc Natl Acad Sci USA 101:12404–12410
Maricio R, Stahl EA, Korves T (2003) Natural selection for polymorphism in the disease resistance gene Rps2 of Arabidopsis thaliana. Genetics 163:735–746
Martin GB, Bogdanove AJ, Sessa G (2003) Understanding the functions of plant disease resistance proteins. Annu Rev Plant Biol 54:23–61
McDowell JM, Dhandaydham M, Long TA, Aarts MG, Goff S, Holub EB, Dangl JL (1998) Intragenic recombination and diversifying selection contribute to the evolution of downy mildew resistance at the RPP8 locus of Arabidopsis. Plant Cell 10:1861–1874
McHale L, Tan X, Koehl P, Michelmore RW (2006) Plant NBS-LRR proteins: adaptable guards. Genome Biol 7:212
Meyers BC, Chin DB, Shen KA, Sivaramakrishnan S, Lavelle DO, Zhang Z, Michelmore RW (1998a) The major resistance gene cluster in lettuce is highly duplicated and spans several megabases. Plant Cell 10:1817–1832
Meyers BC, Shen KA, Rohani P, Gaut BS, Michelmore RW (1998b) Receptor-like genes in the major resistance locus of lettuce are subject to divergent selection. Plant Cell 10:1833–1846
Meyers BC, Kozik A, Griego A, Kuang H, Michelmore RW (2003) Genome-wide analysis of NBS-LRR-encoding genes in Arabidopsis. Plant Cell 15:809–834
Monosi B, Wisser RJ, Pennill L, Hulbert SH (2004) Full-genome analysis of resistance gene homologues in rice. Theor Appl Genet 109:1434–1447
Noel L, Moores TL, Van der Biezen EA, Parniske M, Daniels MJ, Parker JE, Jones JD (1999) Pronounced intraspecific haplotype divergence at the RPP5 complex disease resistance locus of Arabidopsis. Plant Cell 11:2099–2111
Parniske M, Jones JD (1999) Recombination between diverged clusters of the tomato Cf-9 plant disease resistance gene family. Proc Natl Acad Sci USA 96:5850–5855
Perriere G, Gouy M (1996) WWW-Query: an on-line retrieval system for biological sequence banks. Biochimie 78:364–369
Qu S, Liu G, Zhou B, Bellizzi M, Zeng L, Dai L, Han H, Wang GL (2006) The broad-spectrum blast resistance gene Pi9 encodes a nucleotide-binding site-leucine-rich repeat protein and is a member of a multigene family in rice. Genetics 172:1901–1914
Shang J, Tao Y, Chen X, Zou Y, Lei C, Wang J, Li X, Zhao X, Zhang M, Lu Z, Xu J, Cheng Z, Wan J, Zhu L (2009) Identification of a new rice blast resistance gene, Pid3, by genomewide comparison of paired nucleotide-binding site-leucine-rich repeat genes and their pseudogene alleles between the two sequenced rice genomes. Genetics 182:1303–1311
Shimamoto K, Kyozuka J (2002) Rice as a model for comparative genomics of plants. Annu Rev Plant Biol 53:399–419
Song WY, Pi LY, Wang GL, Gardner J, Holsten T, Ronald P (1997) Evolution of the rice Xa21 disease resistance gene family. Plant Cell 9:1279–1287
Sun Q, Collins NC, Ayliffe M, Smith SM, Drake J, Pryor T, Hulbert SH (2001) Recombination between paralogues at the rp1 rust resistance locus in maize. Genetics 158:423–438
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The ClustalX windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucl Acids Res 25:4876–4882
Valent B (1990) Rice blast as a model system for plant pathology. Phytopathology 80:33–36
Vitte C, Panaud O (2003) Formation of solo-LTRs through unequal homologous recombination counterbalances amplifications of LTR retrotransposons in rice Oryza sativa L. Mol Biol Evol 20:528–540
Walsh JB (1987) Sequence-dependent gene conversion: can duplicated genes diverge fast enough to escape conversion? Genetics 117:543–557
Wang X, Jia Y, Shu QY, Wu D (2008) Haplotype diversity at the Pi-ta locus in cultivated rice and its wild relatives. Phytopathology 98:1305–1311
Webb CA, Richter TE, Collins NC, Nicolas M, Trick HN, Pryor T, Hulbert SH (2002) Genetic and molecular characterization of the maize rp3 rust resistance locus. Genetics 162:381–394
Wei F, Wing RA, Roger PW (2002) Genome dynamics and evolution of the Mla (Powdery mildew) resistance locus in barley. Plant Cell 14:1903–1917
Wicker T, Yahiaoui N, Keller B (2007) Contrasting rates of evolution in Pm3 loci from three wheat species and rice. Genetics 177:1207–1216
Wing RA, Ammiraju JS, Luo M, Kim H, Yu Y, Kudrna D, Zuccolo A, Ammiraju JS, Luo M, Nelson W, Ma J, SanMiguel P, Hurwitz B, Ware D, Brar D, Mackill D, Soderlund C, Stein L, Jackson S (2005) The oryza map alignment project: the golden path to unlocking the genetic potential of wild rice species. Plant Mol Biol 59:53–62
Wisser RJ, Qi S, Hulbert SH, Kresovich S, Nelson RJ (2005) Identification and characterization of regions of the rice genome associated with broad-spectrum, quantitative disease resistance. Genetics 169:2277–2293
**ao S, Emerson B, Ratanasut K, Patrick E, O’Neill C, Bancroft I, Turner JG (2004) Origin and maintenance of a broad-spectrum disease resistance locus in Arabidopsis. Mol Biol Evol 21:1661–1672
Yang Z (1997) PAML: a program package for phylogenetic analysis by maximum likelihood. Comput Appl Biosci 13:555–556
Yang Z, Nielsen R, Goldman N, Pedersen AM (2000) Codon substitution models for heterogeneous selection pressure at amino acid sites. Genetics 155:431–449
Yoshida K, Miyashita NT (2009) DNA polymorphism in the blast disease resistance gene Pita of the wild rice Oryza rufipogon and its related species. Genes Genet Syst 84(2):121–136
Zhou B, Qu S, Liu G, Dolan M, Sakai H, Lu G, Bellizzi M, Wang GL (2006) The eight amino-acid differences within three leucine-rich repeats between Pi2 and Piz-t resistance proteins determine the resistance specificity to Magnaporthe grisea. Mol Plant Microbe Interact 19:1216–1228
Zhou B, Dolan M, Sakai H, Wang GL (2007) The genomic dynamics and evolutionary mechanism of the Pi2/9 locus in rice. Mol Plant Microbe Interact 20:63–71
Acknowledgments
This project was supported by the “973” Project (2006CB101904), the “948” Project (2006-G61), the Henye Project of Ministry of Agriculture, National Natural Science Foundation of China (30828022), and the NSF-Plant Genome Research Program (#0605017).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by E. Guiderdoni.
L. Dai and J. Wu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dai, L., Wu, J., Li, X. et al. Genomic structure and evolution of the Pi2/9 locus in wild rice species. Theor Appl Genet 121, 295–309 (2010). https://doi.org/10.1007/s00122-010-1310-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-010-1310-0