Abstract
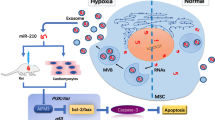
This study seeks to test our hypothesis that transgenic induction of miR-210 in mesenchymal stem cells (MSC) simulates the pro-survival effects of ischemic preconditioning (IPC) and that engraftment of PCMSC helps in the functional recovery of ischemic heart by miR-210 transfer to host cardiomyocytes through gap junctions. miR-210 expression in MSC was achieved by IPC or nanoparticle-based transfection of miR-210 plasmid (miRMSC) and functional recovery of the infarcted heart of rat transplanted with PCMSC or miRMSC was evaluated. Both PCMSC and miRMSC showed higher survival under lethal anoxia as compared to non-PCMSC and scramble-transfected MSC (ScMSC) controls with concomitantly lower CASP8AP2 expression. Similarly, both PCMSC and miRMSC survived better and accelerated functional recovery of ischemic heart post-transplantation. To validate our hypothesis that MSC deliver miR-210 to host cardiomyocytes, in vitro co-culture between cardiomyocytes and PCMSC or miRMSC (using non-PCMSC or ScMSC as controls) showed co-localization of miR-210 with gap-junctional connexin-43. miR-210 transfer to cardiomyocytes was blocked by heptanol pretreatment. Moreover, higher survival of cardiomyocytes co-cultured with PCMSC was observed with concomitant expression of CASP8AP2 as compared to cardiomyocytes co-cultured with non-PCMSC thus suggesting that miR-210 was translocated from MSC to protect host cardiomyocytes. Induction of miR-210 in MSC promoted their survival post-engraftment in the infarcted heart. Moreover, direct transfer of pro-survival miR-210 from miRMSC to host cardiomyocytes led to functional recovery of the ischemic heart.







Similar content being viewed by others
References
Lau NC, Lim LP, Weinstein EG, Bartel DP (2001) An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans. Science 294:858–862
Rane S, Sayed D, Abdellatif M (2007) MicroRNA with a MacroFunction. Cell Cycle 6:1850–1855
Semenza GL, Agani F, Feldser D, Iyer N, Kotch L, Laughner E, Yu A (2000) Hypoxia, HIF-1, and the pathophysiology of common human diseases. Adv Exp Med Biol 475:123–130
Ivan M, Harris AL, Martelli F, Kulshreshtha R (2008) Hypoxia response and microRNAs: no longer two separate worlds. J Cell Mol Med 12:1426–1431
Haider KH, Kim HW, Ashraf M (2009) Hypoxia-inducible factor-1alpha in stem cell preconditioning: mechanistic role of hypoxia-related micro-RNAs. J Thorac Cardiovasc Surg 138:257
Kulshreshtha R, Ferracin M, Negrini M, Calin GA, Davuluri RV, Ivan M (2007) Regulation of microRNA expression: the hypoxic component. Cell Cycle 6:1426–1431
Fasanaro P, D’Alessandra Y, Di Stefano V, Melchionna R, Romani S, Pompilio G, Capogrossi MC, Martelli F (2008) MicroRNA-210 modulates endothelial cell response to hypoxia and inhibits the receptor tyrosine kinase ligand Ephrin-A3. J Biol Chem 283:15878–15883
Mathew LK, Simon MC (2009) mir-210: a sensor for hypoxic stress during tumorigenesis. Mol Cell 35:737–738
Huang X, Le QT, Giaccia AJ (2010) MiR-210—micromanager of the hypoxia pathway. Trends Mol Med 16:230–237
Lu G, Haider HK, Jiang S, Ashraf M (2009) Sca-1+ stem cell survival and engraftment in the infarcted heart: dual role for preconditioning-induced connexin-43. Circulation 119:2587–2596
Suzuki Y, Kim HW, Ashraf M, Haider H (2011) Diazoxide potentiates mesenchymal stem cell survival via NF-kappaB-dependent miR-146a expression by targeting Fas. Am J Physiol Heart Circ Physiol 299:H1077–H1082
Lu G, Jiang S., Ashraf, M., Haider, KH (2012) Sub-cellular preconditioning of stem cells: mitochondrial targeting of connexin-43 transgene is cytoprotective and involves shift of balance between mitochondrial Bak and Bcl-xL. Regen Med 7(3):323–334
Haider H, Ashraf M (2010) Preconditioning and stem cell survival. J Cardiovasc Transl Res 3:89–102
Kim HW, Haider HK, Jiang S, Ashraf M (2009) Ischemic preconditioning augments survival of stem cells via miR-210 expression by targeting caspase-8-associated protein 2. J Biol Chem 284:33161–33168
Haider KH, Idris NM, Kim HW, Ahmed RP, Shujia J, Ashraf M (2010) MicroRNA-21 is a key determinant in IL-11/Stat3 anti-apoptotic signalling pathway in preconditioning of skeletal myoblasts. Cardiovasc Res 88:168–178
Ye L, Haider H, Tan R, Toh W, Law PK, Tan W, Su L, Zhang W, Ge R, Zhang Y et al (2007) Transplantation of nanoparticle transfected skeletal myoblasts overexpressing vascular endothelial growth factor-165 for cardiac repair. Circulation 116:I113–I120
Chan SY, Loscalzo J (2010) MicroRNA-210: a unique and pleiotropic hypoxamir. Cell Cycle 9:1072–1083
Zhu XM, Han T, Sargent IL, Yin GW, Yao YQ (2009) Differential expression profile of microRNAs in human placentas from preeclamptic pregnancies vs normal pregnancies. Am J Obstet Gynecol 200(661):e1–e7
Camps C, Buffa FM, Colella S, Moore J, Sotiriou C, Sheldon H, Harris AL, Gleadle JM, Ragoussis J (2008) hsa-miR-210 is induced by hypoxia and is an independent prognostic factor in breast cancer. Clin Cancer Res 14:1340–1348
Giannakakis A, Sandaltzopoulos R, Greshock J, Liang S, Huang J, Hasegawa K, Li C, O’Brien-Jenkins A, Katsaros D, Weber BL et al (2008) miR-210 links hypoxia with cell cycle regulation and is deleted in human epithelial ovarian cancer. Cancer Biol Ther 7:255–264
Pulkkinen K, Malm T, Turunen M, Koistinaho J, Yla-Herttuala S (2008) Hypoxia induces microRNA miR-210 in vitro and in vivo ephrin-A3 and neuronal pentraxin 1 are potentially regulated by miR-210. FEBS Lett 582:2397–2401
Crosby ME, Kulshreshtha R, Ivan M, Glazer PM (2009) MicroRNA regulation of DNA repair gene expression in hypoxic stress. Cancer Res 69:1221–1229
Hu S, Huang M, Li Z, Jia F, Ghosh Z, Lijkwan MA, Fasanaro P, Sun N, Wang X, Martelli F et al (2010) MicroRNA-210 as a novel therapy for treatment of ischemic heart disease. Circulation 122:S124–S131
Muralidharan-Chari V, Clancy JW, Sedgwick A, D’Souza-Schorey C (2010) Microvesicles: mediators of extracellular communication during cancer progression. J Cell Sci 123:1603–1611
Valiunas V, Polosina YY, Miller H, Potapova IA, Valiuniene L, Doronin S, Mathias RT, Robinson RB, Rosen MR, Cohen IS et al (2005) Connexin-specific cell-to-cell transfer of short interfering RNA by gap junctions. J Physiol 568:459–468
Kizana E, Cingolani E, Marban E (2009) Non-cell-autonomous effects of vector-expressed regulatory RNAs in mammalian heart cells. Gene Ther 16:1163–1168
Hosoda T, Zheng H, Cabral-da-Silva M, Sanada F, Ide-Iwata N, Ogorek B, Ferreira-Martins J, Arranto C, D’Amario D, Del Monte F et al (2011) Human cardiac stem cell differentiation is regulated by a mircrine mechanism. Circulation 123:1287–1296
Lim PK, Bliss SA, Patel SA, Taborga M, Dave MA, Gregory LA, Greco SJ, Bryan M, Patel PS, Rameshwar P (2011) Gap junction-mediated import of microrna from bone marrow stromal cells can elicit cell cycle quiescence in breast cancer cells. Cancer Res 71:1550–1560
Barcaroli D, Bongiorno-Borbone L, Terrinoni A, Hofmann TG, Rossi M, Knight RA, Matera AG, Melino G, De Laurenzi V (2006) FLASH is required for histone transcription and S-phase progression. Proc Natl Acad Sci USA 103:14808–14812
Milovic-Holm K, Krieghoff E, Jensen K, Will H, Hofmann TG (2007) FLASH links the CD95 signaling pathway to the cell nucleus and nuclear bodies. EMBO J 26:391–401
Kim HW, Mallick F, Durrani S, Ashraf M, Jiang S, Haider KH (2012) Concomitant activation of miR-107/PDCD10 and hypoxamir-210/Casp8ap2 and their role in cytoprotection during ischemic preconditioning of stem cells. Antioxid Redox Signal. doi:10.1089/ars.2012.4518
Acknowledgments
This work was supported by NIH Grants# [R37 HL074272; HL-087246(M.A) and HL-087288; HL-089535; HL106190-01(Kh.H.H)].
Disclosure statement
Nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 4436 kb)
Rights and permissions
About this article
Cite this article
Kim, H.W., Jiang, S., Ashraf, M. et al. Stem cell-based delivery of Hypoxamir-210 to the infarcted heart: implications on stem cell survival and preservation of infarcted heart function. J Mol Med 90, 997–1010 (2012). https://doi.org/10.1007/s00109-012-0920-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-012-0920-1