Abstract
Objective
Tinnitus is defined as an imaginary subjective perception in the absence of an external sound. Convergent evidence proposes that tinnitus perception includes auditory, attentional and emotional components. The aim of this study was to investigate the thalamic, auditory and limbic interactions associated with tinnitus-related distress by Diffusion Tensor Imaging (DTI).
Methods
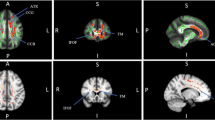
A total of 36 tinnitus patients, 20 healthy controls underwent an audiological examination, as well as a magnetic resonance imaging protocol including structural and DTI sequences. All participants completed the Tinnitus Handicap Inventory (THI) and Visual Analog Scales (VAS) related with tinnitus. The fractional anisotropy (FA) and apparent diffusion coefficient (ADC) values were obtained for the auditory cortex (AC), inferior colliculus (IC), lateral lemniscus (LL), medial geniculate body (MGB), thalamic reticular nucleus (TRN), amygdala (AMG), hippocampus (HIP), parahippocampus (PHIP) and prefrontal cortex (PFC).
Results
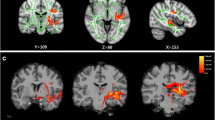
In tinnitus patients the FA values of IC, MGB, TRN, AMG, HIP decreased and the ADC values of IC, MGB, TRN, AMG, PHIP increased significantly. The contralateral IC-LL and bilateral MGB FA values correlated negatively with hearing loss. A negative relation was found between the AMG-HIP FA values and THI and VAS scores. Bilateral ADC values of PHIP and PFC significantly correlated with the attention deficiency—VAS scores.
Conclusion
In conclusion, this is the first DTI study to investigate the grey matter structures related to tinnitus perception and the significant correlation of FA and ADC with clinical parameters suggests that DTI can provide helpful information for tinnitus. Magnifying the microstructures in DTI can help evaluate the three faces of tinnitus nature: hearing, emotion and attention.





Similar content being viewed by others
References
Andersson G, Lyttkens L, Hirvela C, Furmark T, Tillfors M, Fredrikson M. Regional cerebral blood flow during tinnitus: a PET case study with lidocaine and auditory stimulation. Acta Otolaryngol. 2000;120:967–72.
Jastreboff PJ. Phantom auditory perception (tinnitus): mechanisms of generation and perception. Neurosci Res. 1990;8:221–54.
Muhlau M, Rauschecker JP, Oestreicher E, Gaser C, Rottinger M, Wohlschlager AM, Simon F, Etgen T, Conrad B, Sander D. Structural brain changes in tinnitus. Cereb Cortex. 2006;16:1283–88.
Rauschecker JP, Leaver AM, Mühlau M. Tuning out the noise: limbic-auditory interactions in tinnitus. Neuron. 2010;66:819–26.
Schneider P, Andermann M, Wengenroth M, Goebel R, Flor H, Rupp A, Diesch E. Reduced volume of Heschl’s gyrus in tinnitus. Neuroimage. 2009;45(3):927–39.
Mirz F, Gjedde A, Sodkilde-Jrgensen H, Pedersen CB. Functional brain imaging of tinnitus-like perception induced by aversive auditory stimuli. Neuroreport. 2000;11:633–37.
Landgrebe M, Langguth B, Rosengarth K, Braun S, Koch A, Kleinjung T, May A, de Ridder D, Hajak G. Structural brain changes in tinnitus: grey matter decrease in auditory and non-auditory brain areas. Neuroimage. 2009;213–18.
Crippa A, Lanting CP, Van Dijk P, Roerdink JB. A diffusion tensor imaging study on the auditory system and tinnitus. Open Neuroimag J. 2010;4:16–25.
Kleinjung T, Eichhammer P, Landgrebe M, Sand P, Hajak G, Steffens T, Strutz J, Langguth B. Combined temporal and prefrontal transcranial magnetic stimulation for tinnitus treatment: a pilot study. Otolaryngol Head Neck Surg. 2008;138:497–501.
De Ridder D, Fransen H, Francois O, Sunaert S, Kovacs S, Van De Heyning P. Amygdalohippocampal involvement in tinnitus and auditory memory. Acta Otolaryngol Suppl. 2006;(556):50–3.
Vanneste S, Plazier M, der Loo Ev, de Heyning PV, Congedo M, De Ridder D The neural correlates of tinnitus-related distress. Neuroimage. 2010;52(2):470–80.
Basser PJ, Pierpaoli C. Microstructural and physiological features of tissues elucidated by quantitative diffusion tensor MRI. J Magn Reson B. 1996;111:209–19.
Mori S, Zhang J. Principles of diffusion tensor imaging and its applications to basic neuroscience research. Neuron. 2006;51(5):527–39.
Peng SJ, Harnod T, Tsai JZ, Ker MD, Chiou JC, Chiueh H, Wu CY, Hsin YL. Evaluation of subcortical grey matter abnormalities in patients with MRI-negative cortical epilepsy determined through structural and tensor magnetic resonance imaging. BMC Neurol. 2014;14:104.
Cappellani R, Bergsland N, Weinstock-Guttman B, Kennedy C, Carl E, Ramasamy DP, Hagemeier J, Dwyer MG, Patti F, Zivadinov R. Diffusion tensor MRI alterations of subcortical deep gray matter in clinically isolated syndrome. J Neurol Sci. 2014;338(1–2):128–34.
Cavallari M, Moscufo N, Meier D, Skudlarski P, Pearlson GD, White WB, Wolfson L, Guttmann CR. Thalamic fractional anisotropy predicts accrual of cerebral white matter damage in older subjects with small-vessel disease. J Cereb Blood Flow Metab. 2014;34(8):1321–7.
Pal D, Trivedi R, Saksena S, Yadav A, Kumar M, Pandey CM, Rathore RK, Gupta RK. Quantification of age- and gender-related changes in diffusion tensor imaging indices in deep grey matter of the normal human brain. J Clin Neurosci. 2011;18(2):193–6.
Smits M, Kovacs S, de Ridder D, Peeters RR, van Hecke P, Sunaert S. Lateralization of functional magnetic resonance imaging (fMRI) activation in the auditory pathway of patients with lateralized tinnitus. Neuroradiology. 2007;49(8):669–79.
Yoo DS, Choi WY, Lee SY, Jeong JW, Lee JW, Kim S, Chang Y. Quantitative analysis of white matter on DTI images of patients with tinnitus: preliminary report. Conf Proc IEEE Eng Med Biol Soc. 2006;1:1870–2.
Husain FT, Medina RE, Davis CW, Szymko-Bennett Y, Simonyan K, Pajor NM, Horwitz B. Neuroanatomical changes due to hearing loss and chronic tinnitus: a combined VBM and DTI study. Brain Res. 2011;1369:74–88.
Aldhafeeri FM, Mackenzie I, Kay T, Alghamdi J, Sluming V. Neuroanatomical correlates of tinnitus revealed by cortical thickness analysis and diffusion tensor imaging. Neuroradiology. 2012;54(8):883–92.
Seydell-Greenwald A, Raven EP, Leaver AM, Turesky TK, Rauschecker JP. Diffusion imaging of auditory and auditory-limbic connectivity in tinnitus: preliminary evidence and methodological challenges. Neural Plast. 2014;2014:145943.
Newman CW, Jacobson GP, Spitzer JB. Development of the Tinnitus Handicap Inventory. Arch Otolaryngol Head Neck Surg. 1996;122:143–8.
Netsch T, van Muiswinkel A. Quantitative evaluation of image-based distortion correction in diffusion tensor imaging. IEEE Trans Med Imaging. 2004;23(7):789–98.
Shrout PE, Fleiss JL. Intraclass correlations: uses in assessing rater reliability. Psychol Bull. 1979;86:420–28.
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159–74.
Benson RR, Gattu R, Cacace AT. Left hemisphere fractional anisotropy increase in noise-induced tinnitus: a diffusion tensor (DTI) study of white matter tracts in the brain. Hear Res. 2014;309:8–16.
Lin Y, Wang J, Wu C, Wai Y, Yu J, Ng S. Diffusion tensor imaging of the auditory pathway in sensorineural hearing loss: changes in radial diffusivity and diffusion anisotropy. J Magn Reson Imaging. 2008;28(3):598–603.
Wu CM, Ng SH, Liu TC. Diffusion tensor imaging of the subcortical auditory tract in subjects with long-term unilateral sensorineural hearing loss. Audiol Neurootol. 2009;14(4):248–53.
Emmorey K, Allen JS, Bruss J, Schenker N, Damasio H. A morphometric analysis of auditory brain regions in congenitally deaf adults. Proc Natl Acad Sci USA. 2003;100(17):10049–54.
Langguth B, Landgrebe M, Kleinjung T, Sand GP, Hajak G. Tinnitus and depression. World J Biol Psychiatry. 2011;12:489–500.
Kreyberg S, Torvik A, Bjorneboe A, Wiik-Larsen W, Jacobsen D. Trimethyltin poisoning: report of a case with postmortem examination. Clin Neuropathol. 1992;11:256–59.
Corkin S, Amaral DG, Gonzalez RG, Johnson KA, Hyman BT. H. M.’s medial temporal lobe lesion: findings from magnetic resonance imaging. J Neurosci. 1997;17:3964–79.
Yu YF, Zhai F, Dai CF, Hu JJ. The relationship between age-related hearing loss and synaptic changes in the hippocampus of C57BL/6 J mice. Exp Gerontol. 2011;46:716–22.
Boutros NN, Mears R, Pflieger ME, Moxon KA, Ludowig E, Rosburg T. Sensory gating in the human hippocampal and rhinal regions: regional differences. Hippocampus. 2008;18:310–31.
Bickford PC, Luntz-Leybman V, Freedman R. Auditory sensory gating in the rat hippocampus: modulation by brainstem activity. Brain Res. 1993;607:33–8.
Diederen KM, Neggers SF, Daalman K, Blom JD, Goekoop R, Kahn RS, Sommer IE. Deactivation of the parahippocampal gyrus preceding auditory hallucinations in schizophrenia. Am J Psychiatry. 2010;167:427–35.
Hwang JH, Chou PH, Wu CW, Chen JH, Liu TC. Brain activation in patients with idiopathic hyperacusis. Am J Otolaryngol. 2009;30(6):432–4.
Leaver AM, Seydell-Greenwald A, Turesky TK, Morgan S, Kim HJ, Rauschecker JP. Cortico-limbic morphology separates tinnitus from tinnitus distress. Front Syst Neurosci. 2012;6:21.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper was presented as oral presentation at "XX". Symposium of Neuroradiologicum, 2014.
Rights and permissions
About this article
Cite this article
Gunbey, H., Gunbey, E., Aslan, K. et al. Limbic-Auditory Interactions of Tinnitus: An Evaluation Using Diffusion Tensor Imaging. Clin Neuroradiol 27, 221–230 (2017). https://doi.org/10.1007/s00062-015-0473-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-015-0473-0