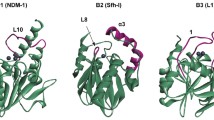
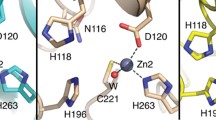
Abstract.
During the past few years the results from molecular biological, biochemical, chemical, physical and theoretical approaches expanded the knowledge about metallo-β-lactamases considerably. The main reason for the attracted interest is a persisting medical problem. Bacteria expressing metallo-β-lactamases can be resistant to treatment with all the known β-lactam antibiotics, and they are additionally invulnerable to combined treatment with inhibitors for the wider-spread serine-β-lactamases. However, clinically useful inhibitors for metallo-β-lactamases are not yet available. In spite of the rapidly expanding knowledge base a central question is still controversially discussed: is it the mononuclear, the binuclear or the metal-free state which might serve as the physiologically relevant target for inhibitor design? A summary of the present views of the roles and coordination geometries of metal ion(s) in metallo-β-lactamases is combined with a discussion of the possibly variable metal ion content under physiological conditions.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Received 19 May 2004; received after revision 6 July 2004; accepted 13 July 2004
Rights and permissions
About this article
Cite this article
Heinz, U., Adolph, H.W. Metallo-β-lactamases: two binding sites for one catalytic metal ion?. CMLS, Cell. Mol. Life Sci. 61, 2827–2839 (2004). https://doi.org/10.1007/s00018-004-4214-9
Issue Date:
DOI: https://doi.org/10.1007/s00018-004-4214-9