Abstract
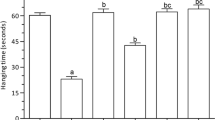
Curcumin, a major component of Indian saffron through clinical studies, revealed its neuroprotective effect in neurodegenerative diseases. However, it has not been utilized alone orally due to its low bioavailability. There are certain strategies to overcome the drawbacks such as poor absorption and low aqueous solubility. Many strategies are utilized to increase the systemic availability of curcumin. Among them, the steady intestinal and liver metabolism of curcumin by a curcumin adjuvant (enzyme inhibitor/inducer) is an important and less engrossed strategy for improving the overall systemic bioavailability of curcumin. Here, we assess the effect of probiotic Lactobacillus rhamnosus as a curcumin adjuvant (potentiate the effect of curcumin) in scopolamine-induced dementia in mice. To induce amnesia, scopolamine was used in a mouse model (1 mg/kg, daily for 10 days i.p.). After execution of behavioural tests (Morris water maze test), brains and liver were isolated for further neurochemical and histopathology examination. Our results showed a significant increase in antioxidant enzyme levels in curcumin with a probiotic group compared with curcumin alone. Besides, histopathology study results showed less neuronal damage of curcumin with probiotics as compared with the curcumin and scopolamine alone groups. Additionally, curcumin with probiotics improved memory and cognitive functions in the behavioural study with the significance of p ≤ 0.0001. In conclusion, curcumin with probiotics has greater activity as compared with curcumin alone and reverses the hallmarks of Alzheimer’s disease (AD).



Similar content being viewed by others
Data availability
Not applicable
References
Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB et al (2007) Bioavailability of curcumin: problems and promises. Mol Pharm 4(6):807–818. https://doi.org/10.1021/mp700113r
Athari NAS, Djazayeri A, Safa M, Azami K, Ahmadvand B, Sabbaghziarani F, Sharifzadeh M, Vafa M (2018) Lactobacilli and bifidobacteria ameliorate memory and learning deficits and oxidative stress in β-amyloid(1–42) injected rats. Appl Physiol Nutr Metab 43(7):718–726. https://doi.org/10.1139/apnm-2017-0648
Biernat KA, Pellock SJ, Bhatt AP, Bivins MM, Walton WG, Tran BN (2019) Structure, function, and inhibition of drug reactivating human gut microbial β-glucuronidases. Sci Rep 9(825):1–5. https://doi.org/10.1038/s41598-018-36069-w
Bondi MW, Edmonds EC, Salmon DP (2017) Alzheimer’s disease: past, present, and future. J Int Neuropsychol Soc 23(9-10):818–831. https://doi.org/10.1017/S135561771700100X
Chen KC, Baxter MG, Rodefer JS (2004) Central blockade of muscarinic cholinergic receptors disrupts affective and attentional set-shifting. Eur J Neurosci 20(4):1081–1088. https://doi.org/10.1111/j.1460-9568.2004.03548.x
Chesser AS, Pritchard SM, Johnson GV (2013) Tau clearance mechanisms and their possible role in the pathogenesis of Alzheimer disease. Front Neurosci 4:122. https://doi.org/10.3389/fneur.2013.00122
Decker AL, Duncan K (2020) Acetylcholine and the complex interdependence of memory and attention. Curr Opin Behav Sci 32:21–28. https://doi.org/10.1016/j.cobeha.2020.01.013
Di Martino RMC, Bisi A, Rampa A, Gobbi S, Belluti F (2017) Recent progress on curcumin-based therapeutics: a patent review (2012-2016). Part II: curcumin derivatives in cancer and neurodegeneration. Expert Opin Ther Pat 4;27(8): 953-965. https://doi.org/10.1080/13543776.2017.1339793
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colourimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Glenner GG, Wong CW (1984) Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochim Biophys Res Commun 120(3):885–890. https://doi.org/10.1016/s0006-291x(84)80190-4
Gong CX, Liu F, Iqbal K (2018) Multifactorial hypothesis and multi-targets for Alzheimer’s disease. J Alzheimers Dis 64(s1):S107–S117. https://doi.org/10.3233/JAD-179921
Goverdhan P, Sravanthi A, Mamatha T (2012) Neuroprotective effects of meloxicam and selegiline in scopolamine induced cognitive impairment and oxidative stress. Int J Alzheimers Dis 2012:1–8. https://doi.org/10.1155/2012/974013
Gupta SC, Kismali G, Aggarwal BB (2013) Curcumin, a component of turmeric: from farm to pharmacy. Biofactors 39(1):2–13. https://doi.org/10.1002/biof.1079
Heger M, van Golen RF, Broekgaarden M, Michel MC (2014) The molecular basis for the pharmacokinetics and pharmacodynamics of curcumin and its metabolites in relation to cancer. Pharmacol Rev 66(1):222–307. https://doi.org/10.1124/pr.110.004044
Kajander K, Myllyluoma E, Rajlic-Stojanovic M, Kyronpalo SS, Rasmussen M, Jarvenpaa SS, Zoetendal EG et al (2008) Clinical trial: multispecies probiotic supplementation alleviates the symptoms of IBS and stabilises intestinal microbiota. Aliment Pharmacol Ther 27(1):48–57. https://doi.org/10.1111/j.1365-2036.2007.03542.x
Kametani F, Hasegawa M (2018) Reconsideration of amyloid hypothesis and tau hypothesis in Alzheimer’s disease. Front Neurosci 12:25. https://doi.org/10.3389/fnins.2018.00025
Karlinsky H (1986) Alzheimer’s disease in Down’s syndrome: a review. J Am Geriatr Soc 34(10):728–734. https://doi.org/10.1111/j.1532-5415.1986.tb04304.x
Konar A, Gupta R, Shukla K, Maloney B, Khanna V, Wadhwa R, Lahiri DK (2019) M1 muscarinic receptor is a key target of neuroprotection, neuroregeneration and memory recovery by i-Extract from Withaniasomnifera. Sci Rep 9:13990. https://doi.org/10.1038/s41598-019-48238-6
Kunnumakkara AB, Harsha C, Banik K, Vikkurthi R, Sailo BL, Bordoloi D, Gupta SC, Aggarwal BB (2019) Is curcumin bioavailability a problem in humans: lessons from clinical trials. Expert Opin Drug Metab Toxicol 15(9):705–733. https://doi.org/10.1080/17425255.2019.1650914
Li HL, Liu C, De Couto G, Ouzounian M, Sun M, Wang AB et al (2008) Curcumin prevents and reverses murine cardiac hypertrophy. J Clin Invest 118(3):879–893. https://doi.org/10.1172/JCI32865
Luck H, Bergmeyer HU (1971) Catalase. In: Bergmeyer HW (ed) Methods of enzymatic analysis. Academic Press, NewYork, pp 885–894
Maurer SV, Williams CL (2017) The cholinergic system modulates memory and hippocampal plasticity via its interactions with non-neuronal cells. Front Immunol 8:1489. https://doi.org/10.3389/fimmu.2017.01489
Nunez J (2008) Morris water maze experiment. J Vis Exp 19:897. https://doi.org/10.3791/897
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95(2):351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Ozawa H, Imaizumi A, Sumi Y, Hashimoto T, Kanai M, Makino Y, Tsuda T, Takahashi N, Kakeya H (2017) Curcumin β-D-glucuronide plays an important role to keep high levels of free-form curcumin in the blood. Biol Pharm Bull 40(9):1515–1524. https://doi.org/10.1248/bpb.b17-00339
Patel C, Patel P, Acharya S (2020) Therapeutic prospective of a spore-forming probiotic—Bacillus clausii UBBC07 against acetaminophen-induced uremia in rats. Probiotics Antimicro Prot 12:253–258. https://doi.org/10.1007/s12602-019-09540-x
Pham PL, Dupont I, Roy D, Lapointe G, Cerning J (2000) Production of exopolysaccharide by Lactobacillus rhamnosus R and analysis of its enzymatic degradation during prolonged fermentation. Appl Environ Microbiol 66(6):2302–2310. https://doi.org/10.1128/aem.66.6.2302-2310.2000
Prasad S, Tyagi AK, Aggarwal BB (2014) Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: the golden pigment from golden spice. Cancer Res Treat 46(1):2–18. https://doi.org/10.4143/crt.2014.46.1.2
Rinwa P, Kumar A (2012) Piperine potentiates the protective effects of curcumin against chronic unpredictable stress-induced cognitive impairment and oxidative damage in mice. Brain Res 1488:38–50. https://doi.org/10.1016/j.brainres.2012.10.002
Rukmini MS, D'souza B, D'souza V (2004) Superoxide dismutase and catalase activities and their correlation with malondialdehyde in schizophrenic patients. Indian J Clin Biochem 19(2):114. https://doi.org/10.1007/BF02894268
Sanders ME (2008) Probiotics: definition, sources, selection, and uses. Clin Infect Dis 46(2):S58–S61. https://doi.org/10.1086/523341
Sathianathan R, Kantipudi SJ (2018) The dementia epidemic: impact, prevention, and challenges for India. Indian J Psychiatry 60(2):165–167. https://doi.org/10.4103/psychiatry.IndianJPsychiatry_261_18
Spencer DG, Lal H (1983) Effects of anticholinergic drugs on learning and memory. Drug Dev Res 3(6):489–502. https://doi.org/10.1002/ddr.430030602
Tang KS (2019) The cellular and molecular processes associated with scopolamine-induced memory deficit: a model of Alzheimer’s biomarkers. Life Sci 233:116695. https://doi.org/10.1016/j.lfs.2019.116695
Tang M, Taghibiglou C (2017) The mechanisms of action of curcumin in Alzheimer’s disease. J Alzheimers Dis 58(4):1003–1016. https://doi.org/10.3233/JAD-170188
Tsukada H, Nishiyama S, Fukumoto D, Ohba H, Sato K, Kakiuchi T (2004) Effects of acute acetylcholinesterase inhibition on the cerebral cholinergic neuronal system and cognitive function: functional imaging of the conscious monkey brain using animal PET in combination with microdialysis. Synapse 52(1):1–10. https://doi.org/10.1002/syn.10310
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1(2):848–858. https://doi.org/10.1038/nprot.2006.116
Weller J, Budson A (2018) Current understanding of Alzheimer’s disease diagnosis and treatment. F1000 Faculty Rev 7:1161. https://doi.org/10.12688/f1000research.14506.1
Westerik N, Kort R, Sybesma W, Reid G (2018) Lactobacillus rhamnosus probiotic food as a tool for empowerment across the value chain in Africa. Front Microbiol 9:1501. https://doi.org/10.1186/1475-2859-13-S1-S7
Weydert CJ, Cullen JJ (2010) Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat Protoc 5(1):51–66. https://doi.org/10.1038/nprot.2009.197
World Health Organiztion (2019) Dementia, key facts. https://www.who.int/news-room/fact-sheets/detail/dementia Accessed 10 March 2020
Zhou JS, Shu Q, Rutherfurd KJ, Prasad J, Gopal PK, Gill HS (2000) Acute oral toxicity and bacterial translocation studies on potentially probiotic strains of lactic acid bacteria. Food Chem Toxicol 38(2-3):153–161. https://doi.org/10.1016/s0278-6915(99)00154-4
Code availability
Not applicable
Author information
Authors and Affiliations
Contributions
All data were generated in-house, and no paper mill was used. The experiments performed were planned and conceptualized by SP and CP. Experimental methodology was executed by SP. GraphPad Prism 7 software used to interpret results was done by SP and CP. Data curation was done by SP, SA, and CP. Investigation was done by SP and CP. CP and SP wrote and prepared the original draft. CP reviewed and edited the paper. SA supervised the experiment.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
IAEC (Institutional Animal Ethics Committee) approved the experimental protocol SSR/IAEC/2019/03.
Consent to participate
Not applicable
Consent for publication
Authors give consent for information to be published in the journal- Naunyn-Schmiedeberg's Archives of Pharmacology
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Patel, C., Pande, S. & Acharya, S. Potentiation of anti-Alzheimer activity of curcumin by probiotic Lactobacillus rhamnosus UBLR-58 against scopolamine-induced memory impairment in mice. Naunyn-Schmiedeberg's Arch Pharmacol 393, 1955–1962 (2020). https://doi.org/10.1007/s00210-020-01904-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01904-3