Abstract
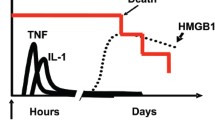
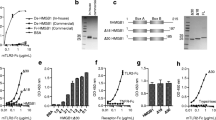
High mobility group box 1 (HMGB1), a ubiquitous DNA-binding protein, has been implicated as a proinflammatory cytokine and late mediator of lethal endotoxemia. HMGB1 is released by activated macrophages. It amplifies and extends the inflammatory response by inducing cytokine release and mediating acute lung injury, anorexia, and the inflammatory response to tissue necrosis. The kinetics of HMGB1 release provide a wide therapeutic window for endotoxemia because extracellular levels of HMGB1 begin to increase 12 to 24 h after exposure to inflammatory stimuli. Here, we demonstrate that a DNA-binding domain of HMGB1, the B box, recapitulates the cytokine activity of full length HMGB1 and efficiently activates macrophages to release tumor necrosis factor (TNF) and other proinflammatory cytokines. Truncation of the B box revealed that the TNF-stimulating activity localizes to 20 amino acids (HMGB1 amino acids 89 to 108). Passive immunization of mice with antibodies raised against B box conferred significant protection against lethal endotoxemia or sepsis, induced by cecal perforation. These results indicate that a proinflammatory domain of HMGB1 maps to the highly conserved DNA-binding B box, making this primary sequence a suitable target in the design of therapeutics.





Similar content being viewed by others
References
Bustin M. (2001) Revised nomenclature for high mobility group (HMG) chromosomal proteins. Trends Biochem. Sci. 26:152–4.
Goodwin GH, Sanders C, Johns EW. (1973) A new group of chromatin-associated proteins with a high content of acidic and basic amino acids. Eur. J. Biochem. 38:14–9.
Bianchi ME, Beltrame M, Paonessa G. (1989) Specific recognition of cruciform DNA by nuclear protein HMG1. Science 243:1056–9.
Maher JF, Nathans D. (1996) Multivalent DNA-binding properties of the HMG-1 proteins. Proc. Natl. Acad. Sci. U.S.A. 93:6717–20.
Hughes EN, Engelsberg BN, Billings PC. (1992) Purification of nuclear proteins that bind to cisplatin-damaged DNA. J. Biol. Chem. 267:13520–7.
Melvin VS, Edwards DP. (1999) Coregulatory proteins in steroid hormone receptor action: the role of chromatin high mobility group proteins HMG-1 and -2. Steroids 64:576–86.
Bustin M, Lehn DA, Landsman D. (1990) Structural features of the HMG chromosomal proteins and their gene. Biochim Biophys Acta. 1049:231–43.
Weir HM et al. (1993) Structure of the HMG box motif in the B-domain of HMG1. EMBO J. 12:1311–9.
Hardman CH et al. (1995) Structure of the A-domain of HMG1 and its interaction with DNA as studied by heteronuclear 3- and 4-dimensional NMR spectroscopy. Biochemistry 34:16596–607.
Baxevanis AD, Landsman D. (1995) The HMG-1 box protein family: classification and functional relationship. Nucleic Acids Res. 23:1604–13.
Calogero S et al. (1999) The lack of chromosomal protein Hmg1 does not disrupt cell growth but causes lethal hypoglycemia in newborn mice. Nature Genet. 22:276–80.
Wang H et al. (1999) HMG-1 as a late mediator of endotoxin lethality in mice. Science 285:248–51.
Andersson U et al. (2000) HMG-1 stimulates proinflammatory cytokine synthesis in human monocytes. J. Exp. Med. 192:565–70.
Abraham E, Arcaroli J, Carmody A, Wang H, Tracey KJ. (2000) HMG-1 as a mediator of acute lung injury. J. Immunol. 165:2950–4.
Wang H et al. (1999) Proinflammatory cytokines (tumor necrosis factor and interleukin 1) stimulate release of high mobility group protein-1 by pituicytes. Surgery 126:389–92.
Ombrellino M et al. (2000) Increased serum concentrations of high-mobility-group protein 1 in hemorrhagic shock. Lancet 354:1446–7.
Agnello D, Wang H, Tracey KJ, Ghezzi P. (2002) HMGB1, a DNA-binding protein with cytokine activity, induces brain TNF and IL-6 production, and mediates anorexia and taste aversion. Cytokine 18:231–6.
Parkkinen J, Rauvala H. (1991) Interactions of plasminogen and tissue plasminogen activator (t-PA) with amphoterin. Enhancement of t-PA-catalyzed plasminogen activation by amphoterin. J. Biol. Chem. 266:16730–5.
Rauvala H, Pihlaakari R. (1987) Isolation and some characteristics of an adhesive factor of brain that enhances neurite outgrowth in central neurons. J. Biol. Chem. 262:16625–5.
Degryse B et al. (2001) The high mobility group (HMG) boxes of the nuclear protein HMG1 induce chemotaxis and cytoskeleton reorganization in rat smooth muscle cells. J. Cell Biol. 152:1192–206.
Gardella S et al. (2002) The nuclear protein HMGB1 is secreted by monocytes via a non-classical, vesicle-mediated secretory pathway. EMBO Rep. 3:995–1001.
Scaffidi P, Misteli T, Bianchi ME. (2002) Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature 418:191–5.
Yang H et al (2001) The HMG B box confers cytokine activity and signals via RAGE. Shock 15(suppl):27.
Sap**ton PL et al. (2002) HMGB1 B Box increases the permeability of Caco-2 enterocytic monolayers and impairs intestinal barrier function in mice. Gastroenterology 123:790–802.
Wichmann MW, Haisken JM, Ayala A, Chaudry IH. (1996) Melatonin administration following hemorrhagic shock decreases mortality from subsequent septic challenge. J. Surgery Res. 65:109–14.
Galanos C, Freudenberg MA, Reutter W. (1979) Galactosamine-induced sensitation of the lethal effects of endotoxin. Proc. Natl. Acad. Sci. U.S.A. 76:5939–43.
Lehmann V, Freudenberg MA, Galanos C. (1987) Lethal toxicity of lipopolysaccharide and tumor necrosis factor in normal and d-galactosamine-treated mice. J. Exp. Med. 165:657–63.
Bianchi M et al. (1996) Suppression of proinflammatory cytokines in monocytes by a tetravalent guanylhydrazone J. Exp. Med. 183:927–36.
Poltorak A et al. (1998) Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282:2085–8.
Hsu H, **ong J, Goeddel DV. (1995) The TNF receptor 1-associated protein TRADD signals cell death and NF-κB activation. Cell. 81:495–504.
Barnes PJ, Karin M. (1997) Nuclear factor-κB: a pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med. 336:1066–71.
Hellerbrand C, Jobin C, Licato LL, Sartor RB, Brenner DA. (1998) Cytokines induce NF-κB in activated but not in quiescent rat hepatic stellate cells. Am. J. Physiol. 275:G269–78.
Fiuza C et al. (2002) Inflammatory promoting activity of HMGB1 on human microvascular endothelial cells. Blood. Forthcoming.
Ulloa L et al. (2002) Ethyl pyruvate prevents lethality in mice with established lethal sepsis and systemic inflammation. Proc. Natl. Acad. Sci. U.S.A. 99:12351–6.
Landsman D, Bustin M. (1993) A signature for the HMG-1 box DNA-binding proteins. Bioessays 15:539–46.
Bachvarov D, Moss T. (1991) The RNA polymerase I transcription factor xUBF contains 5 tandemly repeated HMG homology boxes. Nucleic Acids Res. 19:2331–5.
Van de Wetering M, Oosterwegel M, Dooijes D, Clevers H. (1991) Identification and cloning of TCF-1, a T-lymphocyte-specific transcription factor containing a sequence specific HMG box. EMBO J. 10:123–32.
Sinclair AH et al. (1990) A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature 346:240–4.
Tracey KJ et al. 1986. Shock and tissue injury induced by recombinant human cachectin. Science 234:470–4.
Yang H, Wang H, Tracey KJ. 2001. HMG-1 rediscovered as a cytokine. Shock 15:247–53.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Kokkola, R., Tabibzadeh, S. et al. Structural Basis for the Proinflammatory Cytokine Activity of High Mobility Group Box 1. Mol Med 9, 37–45 (2003). https://doi.org/10.1007/BF03402105
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03402105