Summary
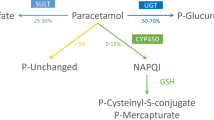
Gilbert’s syndrome (GS) is an inherited bilirubin UDP-glucuronosyl transferase deficiency. The object of this study was to investigate the possible effects of this disorder on the metabolism of a drug, such as paracetamol, which is basically eliminated by hepatic glucuronidation. We studied 32 healthy volunteers and 18 people with GS, all of whom were given 1.5 g of paracetamol orally. In the 24 h urine collected, we determined the elimination of free paracetamol, the conjugates (glucuronide, sulphate) and the oxidation products (cysteine, mercapturic acid) by high pressure liquid chromatography (HPLC). The results are given as a percentage of the total quantity of paracetamol eliminated. The patients with GS were divided into 2 subgroups (GS-I and GS-II) according to whether glucuronidation was more or less than 50%. The overall results of the GS group showed no significant difference in the urinary elimination of metabolites as compared to the control group. However, in subgroup GS-I, a reduction in glucuronidation (P=0.0012) and an increase in oxidation (P=0.0051) was seen, as compared with the other 2 groups. There was inverse correlation between the glucuronide produced by conjugation and the oxidation products (r=−0.8718;P<0.005). People with GS are a heterogeneous group with respect to the metabolism of paracetamol. In one subgroup this was normal. In the other subgroup there was a marked reduction in glucuronidation and an increase in oxidation. These changes could mean that people in this subgroup are more liable to liver damage after an overdose of paracetamol.
Similar content being viewed by others
References
Thomson J.S., Prescott L.F. (1966): Liver damage and impaired glucose tolerance after paracetamol overdose. BMJ 2: 506–507.
Mitchell J.R., Thorgeirsson S.S., Potter W.Z., Jollow D.J., Keiser H. (1974): Acetaminophen-induced hepatic injury: protective role of glutathione in man and rationale for therapy. Clin. Pharmacol. Ther., 16: 676–684.
Prescott L.F. (1983): Paracetamol overdosage. Pharmacological considerations and clinical management. Drugs, 25: 290–314.
Aono S., Adachi V., Uyama E. et al. (1995): Analysis of genes for bilirubin UDP-glucuronosyltransferase in Gilbert’s syndrome. Lancet, 345: 958–959.
Ebner T., Burchell B. (1993): Substrate specificities of two stably expressed human liver UDP-glucuronosyltransferases of the UGT1 gene family. Drug Metab. Dispos., 21: 50–55.
Owens I.S., Ritter J.K. (1992): The novel bilirubin/phenol UDP-glucuronosyltransferase UGT1 gene locus: implications for multiple nonhemolytic familial hyperbilirubinemia phenotypes. Pharmacogenetics, 2: 93–108.
de Morais S.M.F., Wells P.G. (1989): Enhanced acetaminophen toxicity in rats with bilirubin glucuronyl transferase deficiency. Hepatology, 10: 163–167.
Schmid R., Hammaker L. (1959): Glucuronide formation in patients with constitutional hepatic dysfunction (Gilbert’s disease). N. Engl. J. Med., 260: 1310–1314.
Douglas A.P., Savage R.L., Rawlins M.D. (1978): Paracetamol (Acetaminophen) kinetics in patients with Gilbert’s syndrome. Eur. J. Clin. Pharmacol., 13: 209–212.
Ullrich D., Sieg A., Blume R., Bock K.W., Schröter W., Bircher J. (1987): Normal pathways for glucuronidation, sulphation and oxidation of paracetamol in Gilbert’s syndrome. Eur. J. Clin. Invest., 17: 237–240.
de Morais S.M.F., Uetrecht J.P., Wells P.G. (1992): Decreased glucuronidation and increased bioactivation of acetaminophen in Gilbert’s syndrome. Gastroenterology, 102: 577–586.
Esteban A., Pérez-Mateo M. (1993): Gilbert’s disease: a risk factor for paracetamol overdosage? J. Hepatol., 18: 257–258.
Esteban A., Graells M., Satorre J., Pérez-Mateo M. (1992): Determination of paracetamol and its four major metabolites in mouse plasma by reversed-phase ion-pair high-performance liquid chromatography. J. Chromatogr., 573: 121–126.
Critchley J.A.J.H., Nimmo G.R., Gregson C.A., Woolhouse N.M., Prescott L.F. (1986): Inter-subject and ethnic differences in paracetamol metabolism. Br. J. Clin. Pharmacol., 22: 649–657.
Lee H.S., Ti T.Y., Koh Y.K., Prescott L.F. (1992): Paracetamol elimination in Chinese and Indians in Singapore. Eur. J. Clin. Pharmacol., 43: 81–84.
Patel M., Tang B.K., Kalow W. (1992): Variability of acetaminophen metabolism in Caucasians and Orientals. Pharmacogenetics, 2: 38–45.
Esteban A., Pérez-Mateo M. (1993): El metabolismo del paracetamol en voluntarios sanos: estudio de una muestra en población española. Gastroenterol. Hepatol., 16: 55–60.
Esteban A., Calvo R., Pérez-Mateo M. (1996): Paracetamol metabolism in two ethnically different Spanish populations. Eur. J. Drug Metab. Pharmacokinet., 21: 233–239.
Clements J.A., Critchley J.A.J.H., Prescott L.F. (1984): The role of sulphate conjugation in the metabolism and disposition of oral and intravenous paracetamol in man. Br. J. Clin. Pharmacol., 18: 481–485.
Mucklow J.C., Fraser H.S., Bulpitt J., Kahn C., Mould G., Dollery C.T. (1980): Environmental factors affecting paracetamol metabolism in London factory and office workers. Br. J. Clin. Pharmacol., 10: 67–74.
Prescott L.F. (1987): Environmental modulation of paracetamol toxicity. In: Plaa G.L., Du Souich P., Erill S. (Eds) Interactions between drugs and chemicals in industrial societies. Esteve Foundation Symposia, vol 2. Amsterdam: Elsevier, 161–174.
Nash R.M., Stein L., Penno M.B., Passananti G.T., Vesell E.S. (1984): Sources of interindividual variations in acetaminophen and antipyrine metabolism. Clin. Pharmacol. Ther., 36: 417–430.
Bosma P.J., Chowdhury J.R., Huang T.J.et al. (1992): Mechanisms of inherited deficiencies of multiple UDP-glucuronosyltransferase isoforms in two patients with Crigler-Najjar syndrome, type I. FASEB J., 6: 2859–2863.
Ritter J.K., Yeatman M.T., Ferreira P., Owens I.S. (1992): Identification of a genetic alteration in the code for bilirubin UDP-glucuronosyltransferase in the UGT1 gene complex of a Crigler-Najjar type I patient. J. Clin. Invest., 90: 150–155.
Bosma P.J., Chowdhury J.R., Bakker C.et al. (1995): The genetic basis of the reduced expression of bilirubin UDP-glucuronosyltransferase 1 in Gilbert’s syndrome. N. Engl. J. Med., 333: 1171–1175.
Mathew P. (1996): The genetic basis of Gilbert’s disease [comment]. N. Engl. J. Med., 334: 802–803.
Monaghan G., Ryan M., Seddon R., Hume R., Burchell B. (1996): Genetic variation in bilirubin UDP-glucuronosyltransferase gene promoter and Gilbert’s syndrome. Lancet, 347: 578–581.
Sato H., Adachi Y., Koiwai O. (1996): The genetic basis of Gilbert’s syndrome [comment]. Lancet, 347: 557–558.
Bock K.W., Forster A., Gschaidmeier H.et al. (1993): Paracetamol glucuronidation by recombinant rat and human phenol UDP-glucuronosyltransferases. Biochem. Pharmacol., 45: 1809–1814.
Bock K.W., Schrenk D., Forster A.et al. (1994): The influence of environmental and genetic factors on CYP2D6, CYP1A2 and UDP-glucuronosyltransferases in man using sparteine, caffeine, and paracetamol as probes. Pharmacogenetics, 4: 209–218.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Esteban, A., Pérez-Mateo, M. Heterogeneity of paracetamol metabolism in Gilbert’s syndrome. Eur. J. Drug Metab. Pharmacokinet. 24, 9–13 (1999). https://doi.org/10.1007/BF03190005
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190005