Abstract
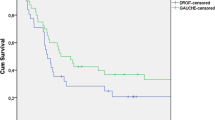
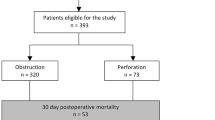
BACKGROUND: Large-bowel cancers that present as obstructing lesions have a poor prognosis. However, little is known of the reasons for the dismal survival and of failure patterns after potentially curative treatment. METHOD: An observational study and multivariate analysis were conducted to identify determinants of survival and to compare recurrence patterns between obstructing and nonobstructing tumors after primary resection and anastomosis as curative treatment. RESULTS: Over a period of ten years (1980–1989), 528 patients with colonic cancer were treated at one institution. The cancer was obstructing in 179 cases and nonobstructing in 349. One-stage primary resection and anastomosis as curative treatment were performed in 107 obstructed and 256 nonobstructed patients. Three hundred thirty-six potentially cured survivors (94 in the former group and 242 in the latter) were followed for a median of 55 months. During follow-up, local recurrence occurred in 37 patients (12 obstructed (12.8 percent) and 25 nonobstructed (10.4 percent),P=0.44) and metastatic disease in 68 (25 obstructed (27.6 percent) and 43 nonobstructed (17.8 percent),P=0.029). Multivariate analysis of survival showed that age over 70 years, Dukes stage, histologic grade, and recurrence were the only prognostic factors. No statistically significant determinant turned out for local recurrence, whereas at multivariate analysis for metastatic and overall relapse, Dukes stage, positive nodes, and obstruction remained independent prognostic factors. CONCLUSIONS: After one-stage emergency curative treatment, patients presenting with obstructing tumors of the colon have a smaller survival probability than that of patients with nonobstructing lesions. Local recurrence pattern is similar between groups. Conversely, obstruction, along with pathologic stage and positive nodes, carries a significantly higher risk of metastatic tumor recurrence and death.
Similar content being viewed by others
References
Welch JP, Donaldson GA. Management of severe obstruction of the large bowel due to malignant disease. Am J Surg 1974;127:492–9.
Ohman U. Prognosis in patients with obstructing colorectal carcinoma. Am J Surg 1982;143:742–7.
Phillips RK, Hittinger R, Fry JS, Fielding LP. Malignant large bowel obstruction. Br J Surg 1985;72:296–302.
Hughes ES. Mortality of acute large bowel obstruction. Br J Surg 1966;53:593–4.
Irvin TT, Greaney MG. The treatment of colonic cancer presenting with intestinal obstruction. Br J Surg 1977;64:741–4.
Chapuis PH, Dent OF, Fisher R,et al. A multivariate analysis of clinical and pathological variables in prognosis after resection of large bowel cancer. Br J Surg 1985;72:698–702.
Fielding LP, Wells BW. Survival after primary and after staged resection for large bowel obstruction caused by cancer. Br J Surg 1974;61:16–8.
Kasperk R, Braun J, Schumpelick V. Obstructive colorectal carcinoma. Principles and technique in 134 cases. Zentralbl Chir 1992;117:67–72.
Serpell JW, McDermott FT, Katrivessis H, Hughes ES. Obstructing carcinomas of the colon. Br J Surg 1989;76:965–9.
Umpleby HC, Williamson RC. Survival in acute obstructing colorectal carcinoma. Dis Colon Rectum 1984;27:299–304.
Wolmark N, Wieand HS, Rockette HE,et al. The prognostic significance of tumor location and bowel obstruction in Dukes B and C colorectal cancer. Findings from the NSABP clinical trials. Ann Surg 1983;198:743–52.
Dutton JW, Hreno A, Hampson LG. Mortality and prognosis of obstructing carcinoma of the large bowel. Am J Surg 1976;131:36–41.
Fielding LP, Stewart-Brown S, Blesovsky L. Large bowel obstruction caused by cancer: a prospective study. Br J Surg 1979;2:515–7
Dukes C. The classification of cancer of the rectum. J Pathol Bacteriol 1932;35:323–32.
Brown SC, Walsh S, Sykes PA. Operative mortality and surgery for colo-rectal cancer. Br J Surg 1988;75:645–7.
Mantel N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 1966;50:163–70.
Cox DR. Regression models and life tables. J R Stat Soc B 1972;34:187–220.
Marubini E, Valsecchi MG. Analysing survival data from clinical trials and observational studies. Chichester: J. Wiley and Sons, 1995:123–7.
Garcia-Valdecasas JC, Llovera JM, deLacy AM,et al. Obstructing colorectal carcinomas: prospective study. Dis Colon Rectum 1991;34:759–62.
Mulcahy HE, Skelly MM, Husain A, O'Donoghue DP. Long-term outcome following curative surgery for malignant large bowel obstruction. Br J Surg 1996;83:46–50.
Willett C, Tepper JE, Cohen A, Orlow E, Welch C. Obstructive and perforative colonic carcinoma: patterns of failure. J Clin Oncol 1985;3:379–84.
Sjodahl R, Franzén T, Nystrom PO. Primary versus staged resection for acute obstructing colorectal carcinoma. Br J Surg 1992;79:685–8.
The SCOTIA Study Group. Single-stage treatment for malignant left-sided colonic obstruction: a prospective randomized clinical trial comparing subtotal colectomy with segmental resection following intraoperative irrigation. Br J Surg 1995;82:1622–7.
Waldron RP, Donovan IA, Drumm J, Mottram SN, Tedman S. Emergency presentation and mortality from colorectal cancer in the elderly. Br J Surg 1986;73:214–6.
Huddy SP, Shorthouse AJ, Marks CG. The surgical treatment of intestinal obstruction due to left sided carcinoma of the colon. Ann R Coll Surg Engl 1988;70:40–3.
Kronborg O. The missing randomized trial of two surgical treatments for acute obstruction due to carcinoma of the left colon and rectum. An interim report. Int J Colorectal Dis 1986;1:162–6.
Phillips RK, Hittinger R, Blesovsky L, Fry JS, Fielding LP. Local recurrence following “curative” surgery for large bowel cancer. I. The overall picture. Br J Surg 1984;71:12–6.
McArdle CS, Hole D. Impact of variability among surgeons on postoperative morbidity and mortality and ultimate survival. BMJ 1991;302:1501–5.
Aldridge MC, Phillips RK, Hittinger R, Fry JS, Fielding LP. Influence of tumour site on presentation, management and subsequent outcome in large bowel cancer. Br J Surg 1986;73:663–70.
Moertel CG, Fleming TR, Macdonald JS,et al. Levamisole and fluorouracil for adjuvant therapy of resected colon carcinoma. N Engl J Med 1990;322:352–8.
International Multicentre Pooled Analysis of Colon Cancer Trials (IMPACT) investigators. Efficacy of adjuvant fluorouracil and folinic acid in colon cancer. Lancet 1995;345:939–44.
Swiss Group for Clinical Cancer Research (SAKK). Long term results of single course of adjuvant intraportal chemotherapy for colorectal cancer. Lancet 1995;345:349–53.
Author information
Authors and Affiliations
About this article
Cite this article
Setti Carraro, P.G., Segala, M., Cesana, B.M. et al. Obstructing colonic cancer: Failure and survival patterns over a ten-year follow-up after one-stage curative surgery. Dis Colon Rectum 44, 243–250 (2001). https://doi.org/10.1007/BF02234300
Issue Date:
DOI: https://doi.org/10.1007/BF02234300