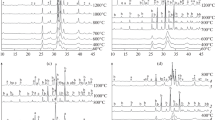
Abstract
Precipitates of calcium phosphate were obtained from aqueous media, some of which contained Mg2+ and F−. Initial solids removed as a slurry were shown by electron diffraction to be amorphous when Mg2+ and F− were not present. Subsequent ageing for 24 h in the supernatant induced a change in morphology and diffraction pattern as observed with the electron microscope. The final solid was crystalline hydroxyapatite. In the presence of F− the initial precipitate was crystalline phase. Conversely, the presence of Mg2+ in the system markedly delayed the transformation from the amorphous to the crystalline phase. When both Mg and F were present the initial phase was amorphous but transformation occurred much more rapidly than in systems with Mg alone. Examination of the solids at high magnification in the electron microscope suggested that the spheroidal structures of the amorphous phase may play a role in the transformation by aligning in a chain-like manner, which appeared to be a morphological feature of the crystalline phase.
Résumé
Des précipités de phosphate de calcium sont obtenus à partir de milieux aqueux dont certains contiennent du Mg2+ et du F−. Les premiers solides éliminés du mélange apparaissent amorphes en diffraction électronique, lorsque Mg2+ et F− ne sont pas présents. Un vieillissement de 24 heures du surnageant induit un changement de morphologie et de diffraction au microscope électronique. Le solide final est constitué, par de l'hydroxyapatite cristallin. En présence de F−, le précipité initial est constitué par une phase cristalline. Inversement, la présence de Mg2+ dans le système retarde de façon marquée la transformation de la phase amorphe en phase cristalline. Lorsque le Mg at le F sont présents simultanément, la phase initiale est amorphe, mais la transformation se fait beaucoup plus rapidement que dans des systèmes avec Mg seul. L'étude des solides, à fort grossissement au microscope électronique, suggère que les structures sphéroïdes de la phase amorphe peuvent jourer un rôle dans la transformation par alignement «en chaine», caractérisant morphologiquement la phase cristalline.
Zusammenfassung
Calciumphosphatfällungen wurden aus wässerigen Lösungen gewonnen, von welchen einige Mg2+ und F− enthielten. Wenn Mg2+ und F− nicht anwesend waren, erwiesen sich anfänglich feste Bestandteile, welche als Klumpen entfernt wurden, als amorph in der Elektronendiffraktion. Nach 24stündigem Stehen des Überstandes konnte im Elektronenmikroskop eine Veränderung der Morphologie und des Diffraktionsmusters beobachtet werden. Die verbleibenden festen Bestandteile waren kristallines Hydroxyapatit. Bei Anwesenheit von F− war der anfängliche Niederschlag kristallin. Bei Vorliegen von Mg2+ im System wurde hingegen die Umwandlung der amorphen in die kristalline Phase stark verzögert. Wenn Mg2+ und F− anwesend waren, war die Anfangsphase amorph, jedoch erfolgte die Umwandlung viel rascher als in Systemen, welche nur Mg enthielten. Die Untersuchung der festen Bestandteile bei starker Vergrößerung im Elektronenmikroskop läßt vermuten, daß die sphäroidischen Formen der amorphen Phase in der Umwandlung eine Rolle spielen könnten, indem sie sich kettenartig aneinanderreihen; dies scheint eine morphologische Eigenschaft der kristallinen Phase zu sein.
Similar content being viewed by others
References
Bachra, B. N., Fischer, H. R. A.: The effect of some inhibitors on the nucleation and crystal growth of apatite. Calcif. Tiss. Res.3, 348–357 (1969).
—, Trautz, O. R., Simon, S. L.: Precipitation of calcium carbonates and phosphates. I. Spontaneous precipitation of calcium carbonates and phosphates under physiological conditions. Arch. Biochem.103, 124–138 (1963).
———: Precipitation of calcium carbonates and phosphates. II. A precipitation diagram for the system calcium-carbonate-phosphate and the heterogeneous nucleation of solids in the metastability region. Advances in Fluorine Research3, 101–118 (1965a).
———: Precipitation of calcium carbonates and phosphates. III. The effect of magnesium and fluoride ions on the spontaneous precipitation of calcium carbonates and phosphates. Arch. oral. Biol.10, 731–738 (1965b).
Eanes, E. D.: Thermochemical studies on amorphous calcium phosphate. Calcif. Tiss. Res.5, 133–145 (1970).
—, Gillessen, I. H., Posner, A. S.: Intermediate states in the precipitation of hydroxyapatite. Nature (Lond.)208, 365–367 (1965).
—, Posner, A. S.: Intermediate phases in the basic solution preparations of alkaline earth phosphates. Calcif. Tiss. Res.2, 38–48 (1968).
——: Kinetics and mechanism of conversion of noncrystalline calcium phosphate to crystalline hydroxyapatite. Trans. N. Y. Acad. Sci.28, 233–241 (1965).
— Zipkin, I., Harper, R. A., Posner, A. S.: Small-angle X-ray diffraction analysis of the effect of fluoride on human bone apatite. Arch. oral Biol.10, 161–173 (1965).
Harper, R. A., Posner, A. S.: Measurement of non-crystalline calcium phosphate in bone mineral. Proc. Soc. exp. Biol. (N. Y.)122, 137–141 (1966).
Hohling, H. J., Theman, H., Vahl, J.: In: Third European Symposium on Calcified Tissues (ed. Fleisch, H., Blackwood, H. J. J. and Owen, M.), p. 146. Berlin-Heidelberg-New York: Springer 1966.
Menczel, J., Posner, A. S., Schraer, H., Pakis, G., Likins, R. C.: Comparative fixation of Sr89 and Ca54 by calcified tissues as related to fluoride induced changes in crystallinity. Proc. Soc. exp. Biol. (N. Y.)110, 609–613 (1962).
Molnar, Z.: Development of the parietal bone of young mice. I. Crystals of bone mineral in frozen-dried preparations. J. Ultrastruct. Res.3, 39–45 (1959).
Neuman, W. F., Toribara, T. Y., Mulryan, B. J.: Synthetic hydroxyapatite crystals. I. Sodium and potassium fixation. Arch. Biochem.98, 234–290 (1962).
Posner, A. S., Eanes, E. D., Harper, R. A., Zipkin, I.: X-ray diffraction, analysis of the effect of fluoride on human bone apatite. Arch. oral Biol.8, 549–570 (1963).
Robinson, R. A., Watson, M. L.: Crystal-collagen relationship in bone as observed in the electron microscope. III. Crystals and collagen morphology as a function of age. Ann. N. Y. Acad. Sci.60, 596–628 (1955).
Schraer, H., Posner, A. S., Schraer, R., Zipkin, I.: Effect of fluoride on bone “crystallinity” in the growing rat. Biochim. biophys. Acta (Amst.)64, 565–567 (1962).
Termine, J. D., Posner, A. S.: Infrared analysis of rat bone: age dependency of amorphous and crystalline mineral fractions. Science153, 1523–1525 (1966).
——: Amorphous/crystalline interrelationships in bone mineral. Calcif. Tiss. Res.1, 8–23 (1967).
—, Wuthier, R. E., Posner, A. S.: Amorphous-crystalline mineral changes during endochondral and periosteal bone formation. Proc. Soc. exp. Biol. (N. Y.)125, 4–9 (1967).
Watson, M. L., Robinson, R. A.: Collagen-crystal relationships in bone. Amer. J. Anat.93, 25–60 (1953).
Weber, J. C., Eanes, E. D., Gerdes, R. J.: Electron microscope study of noncrystalline calcium phosphate. Arch. Biochem.120, 723–724 (1967).
Young, R. A., Lugt, W. van der, Elliott, J. C.: Mechanism for fluorine inhibition of diffusion in hydroxyapatite. Nature (Lond.)223, 729–730 (1969).
Zipkin, I., Posner, A. S., Eanes E. D.: The effect of fluoride on the X-ray-diffraction pattern of the apatite of human bone. Biochim. biophys. Acta (Amst.)59, 255–258 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
West, V.C. Observations on phase transformation of a precipitated calcium phosphate. Calc. Tis Res. 7, 212–219 (1971). https://doi.org/10.1007/BF02062608
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02062608