Abstract
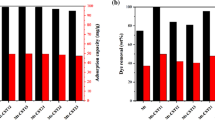
The removal of heavy metal ions (Pb+2, Cu+2 and Zn+2) from solutions with high crosslinked amphoteric starch containing the sulfonate anionic group and the tertiary ammonium cationic group was investigated. The adsorption capacity of sodium tertiary amine sulfunate starch is 0.050 meq/g. The adsorption process has been found to be concentration and pH dependent and exothermic, and follows the Langmuir isothermal adsorption. The heat of adsorption (ΔH) of Pb+2, Cu+2 and zn+2 ions is equal to −10.85. −16.20 and −20.00 Kcal/mole, respectively. The amount of adsorbed metal ions on the adsorbent decreases when NaCl or Na2SO4 is added to the solution.
Similar content being viewed by others
References
J. W. Patterson,Industrial Wastewater Treatment Technology, 2nd ed., Butterworth Publication, Stonehaw. (1985).
J.M. Randall, W. Reuter and A.C. Waiss,J. Appl Polym. Sci.,19, 1563 (1975).
J.M. Randall, E. Hautala, and G. McDonlad,J. Appl., Polym. Sci.,22, 379 (1978).
P. Kuman and S.S. Dara,J Polym. Sci., Polym. Chem. Ed.,19, 397 (1481).
M. Friedman, C.S. Harisen, W.H. Ward, and H.P. Lundgsen,J Appl. Polym. Sci.,17, 377 (1973).
M. Friedman and M.S. Masri,J. Appl Polym. Sci.,18. 2367 (1974).
M. Friedman and M.S. Masri,Environ. Sci. Technol.,7. 951 (1973).
M. Friedman and A.C. Waiss,Environ, Sci. Technol.,6, 451 (1977).
E.J. Roberts and S.P. Rowland,Environ. Sci Technol.,7, 552 (1973).
R.E. Wing, W.M. Doane, and C.R. Russell,J. Appl. Polym. Sci.,19, 847 (1975).
J. Mohammad and V. Tare,J. Appl. Polym. Sci.,42, 317 (1991).
R.E. Wing, W.E. Rayford, W.M. Doane and C.R. Russell,J. Appl. Polym. Sci.,22, 1405 (1979).
V.N. Singh, I.S. Singh, and U.S. Rai,Indian J. Chem.,21A. 247 (1982).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chan, WC. Removal of heavy metal ions with water-insoluble amphoteric sodium tertiary amine sulfonate starches. J Polym Res 1, 221–226 (1994). https://doi.org/10.1007/BF01374099
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01374099