Abstract
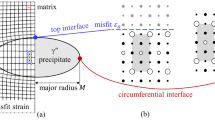
End-point thermodynamic analyses were made of the tetragonal to monoclinic transformation (t→m) occurring in ZrO2 precipitates in a Ca-PSZ alloy and particles in Al2O3-ZrO2 composites. Calculated plots of the reciprocal critical size for transformation temperature were in excellent agreement with experimental data for both systems. Contributions to the total free energy change included bulk chemical, dilatational and residual shear strain energies and also interfacial energies. The latter term consisted of contributions from the change in the chemical surface free energy, the presence of twin boundaries in the precipitate (particle)-matrix interfacial energy. The major impediment to the transformation was the shear strain energy which could not be reduced sufficiently by twinning alone. The t → m reaction proceeded spontaneously when the energy barrier was reduced by the response of the particle-matrix interface. The response comprised loss of coherency and grain boundary microcracking for the Ca-PSZ and Al2O3-ZrO2 alloys, respectively. These results are in accord with recent suggestions that either a stress-free strain or a free surface is a necessary conditions for the initiation of a martensitic transformation.
Similar content being viewed by others
References
R. H. J. Hannink, K. A. Johnston, R. T. Pascoe andR. C. Garvie,Adv. Ceram. 3 (1981) 116.
F. F. Lange,J. Mater. Sci. 17 (1982) 225.
A. G. Evans, N. Burlingame, M. Drory andW. M. Kriven,Acta. Metall. 29 (1981) 447.
N. Claussen andM. Rühle,Adv. Ceram. 3 (1981) 137.
A. H. Heuer, N. Claussen, W. M. Kriven andM. Rühle,J. Amer. Ceram. Soc. 65 (1982) 642.
G. B. Olson andM. Cohen,Ann. Rev. Mater. Sci. 11 (1981) 1.
R. C. Garvie,J. Phys. Chem. 82 (1978) 218.
R. C. Garvie, R. H. J. Hannink andC. Urbani,Ceram. Int. 6 (1980) 19.
D. M. Marsh,Proc. Roy. Soc. A279 (1964) 420.
H. Holmes, E. Fuller, Jr andR. Gammaye,J. Phys. Chem. 76 (1972) 1497.
R. H. J. Hannink,J. Mater. Sci. 13 (1978) 2487.
D. J. Green,J. Amer. Ceram. Soc. 65 (1982) 610.
N. Claussen, R. L. Cox andJ. S. Wallace,Comm. Amer. Ceram. Soc. 65 (1982) C-190.
W. J. Campbell andC. F. Grain,Adv. X-ray Anal. 5 (1962) 244.
S. M. Lang,J. Amer. Ceram. Soc. 47 (1964) 641.
M. V. Swain,J. Non-Cryst. Solids 38/39 (1980) 451.
R. W. Davidge, “Mechanical Behaviour of Ceramics” (Cambridge, London, 1979).
W. D. Kingery,J. Amer. Ceram. Soc. 37 (1954) 42.
S. M. Wiederhorn, Proceedings of the Symposium on Mechanical and Thermal Properties of Ceramics, edited by J. B. Wachtman Jr, NBS Special Publication 303 (1969) 217.
S. Prochazka, J. S. Wallace andN. Claussen,Comm. Amer. Ceram. Soc. 66 (1983) C-125.
“Engineering Properties of Selected Ceramic Materials”, edited by J. F. Lynch, C. G. Ruderer and W. H. Duckworth (American Ceramic Society, Colombus, Ohio, 1966).
J. T. Smith andC. L. Quackenbush,Bull. Amer. Ceram. Soc. 59 (1980) 529.
W. M. Kriven, Proceedings of the International Conference on Solid-Solid Phase Transformations, Pittsburgh, 1981.
M. Rühle andW. M. Kriven,Ber. Bunsenge. Phys. Chem. 17 (1983) 222.
P. C. Clapp,Phys. Status Solidi (b) 57 (1975) 561.
D. J. Cheng andR. F. Wallis,Phys. Rev. B 12 (1975) 5599.
J. M. Galligan andT. Garosshen,Nature 274 (1978) 674.
T. J. Garosshen andJ. M. Galligan,Scripta Metall. 14 (1980) 1111.
K. E. Easterling andP. R. Swann,Acta. Metall. 19 (1971) 117.
S. Burke andR. C. Garvie,J. Mater. Sci. 12 (1977) 1487.
J. W. Spretnak,Met. Trans. A 7A (1976) 158.
H. Ruf andA. G. Evans,J. Amer. Ceram. Soc. 66 (1983) 328.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Garvie, R.C., Swain, M.V. Thermodynamics of the tetragonal to monoclinic phase transformation in constrained zirconia microcrystals. J Mater Sci 20, 1193–1200 (1985). https://doi.org/10.1007/BF01026313
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01026313