Abstract
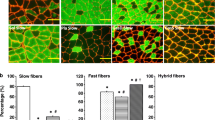
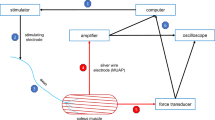
The effects of denervation and hindlimb suspension induced disuse on concentrations of ATP, phosphocreatine (PC), and fiber type profile were investigated in slow twitch soleus and fast twitch extensor digitorum longus (EDL) muscles. The results show that the soleus and EDL muscles differ in their dependency on loadbearing as a stimulus for maintaining normal energy metabolism and the biochemical and morphological characteristics of muscle fibers. As determined by R-P methodology, suspension reduced ATP and PC concentrations of the soleus to 26% and 56%, respectively, while, in EDL only, PC is reduced to 71% of control with no change in ATP. Both muscles, however, show identical losses in ATP and PC following denervation. The energy charge, an indicator of Pi availability in muscle was reduced significantly in both denervated muscles to 82% and 85% in soleus and EDL, respectively. No significant reduction of the energy charge was seen in the muscles from suspended rats. Thus, in parallel with the indirect regulation through muscle loadbearing, the nerve can effectively modulate the levels of high-energy phosphates more directly by some regulatory mechanisms independent of muscle type. Denervation and suspension disuse increased the proportion of type 2 fibers in the soleus with a concomitant decrease in type 1 fibers and a relative rise in the number of very small diameter fibers. The EDL showed only variation in fiber size.
Similar content being viewed by others
References
Morey, E. R. 1979. Spaceflight and bone turnover: correlation with a new rat model of weightlessness. Bio. Science 29:168–179.
Musacchia, X. J., and Deavers, D. R. 1980. A new rat model for studies of hypokinesia in antiorthostasis. Physiologist 23(suppl):s91-s92.
Gazenko, O. G., Genin, A. M., Ilyin, E. A., Oganov, V. S., and Serova, L. V. 1980. Adaptation to weightlessness and its physiological mechanisms (results of animal experiments aboard biosatellites). Physiologist 23(suppl):s11-s15.
Oganov, V. S. 1981. Results of biosatellite studies of gravity-dependent changes in the musculo-skeletal system of mammals. Physiologists 23(suppl):s55-s58.
Gupta, R. C., Misulis, K. E., and Dettbarn, W.-D. 1985. Changes in the cholinergic system of rat sciatic nerve and skeletal muscle following suspension induced disuse. Exp. Neurol. 89:622–633.
Steffen, J. M., and Musacchia, X. J. 1984. Effects of hypokinesia and hypodynamia on protein, RNA, DNA in rat hindlimb muscles. Am. J. Physiol. 247:R728-R732.
Booth, F. W., and Kelson, J. R. 1973. Effects of hindlimb immobilization on contractile and histochemical properties of skeletal muscle. Pflüger's Arch. 342:231–238.
Corley, K. N. Kowalchuk, N., and McComas, A. J. 1984. Contrasting effects of suspension on hind limb muscles in hamster. Exp. Neurol. 85:30–40.
Dettbarn, W.-D., and Misulis, K. E. 1983. Physiological changes in fast and slow muscle with simulated weightlessness. NASA Conf. Publ. 2299:35–36.
Edgerton, V. R., Barnard, R. J., Peter, J. B., Maier, A., and Simpson, D. R. 1975. Properties of immobilized hind-limb muscles of theGalago senegalensis. Exp. Neurol. 46:115–131.
Templeton, G. H., Padalino, M., Manton, J., Glasberg, M., Silver, C. J., Silver, P., DeMartino, G., Leconey, T., Klug, G., Hagler, H., and Sutko, J. L. 1984. Influence of suspension hypokinesia on rat soleus muscle. J. Appl. Physiol.: Respirat. Environ. Exercise Physiol. 56(2):278–286.
Witzmann, F. A., Kim, D. H., and Fitts, R. M. 1985. Effect of hindlimb immobilization on the fatigability of skeletal muscle. J. Appl. Physiol.: Respirat. Environ. Exercise Physiol. 54:1242–1248.
Dettbarn, W.-D. 1981. A distinct difference between slow and fast muscle in acetylcholinesterase recovery after reinnveration in the rat. Exp. Neurol. 74:33–50.
Gupta, R. C., and Dettbarn, W.-D. 1987. Alterations of high energy phosphate compounds in the skeletal muscles of rats intoxicated with diisopropylphosphorofluoridate (DFP) and soman. Fund. Appl. Toxicol. 8:400–407.
Hull-Ryde, E. A., Cummings, R. G., and Lowe, J. E. 1983. Improved methods for high energy nucleotide analysis of canine cardiac muscle using reversed phase high performance liquid chromatography. J. Chromat. 275:411–417.
Mabuchi, K., and Sreter, F. A. 1980. Actomyosin ATPase I. Quantitative measurement of activity in cryostat sections. Muscle & Nerve 3:227–232.
Mabuchi, K., and Sreter, F. A. 1980. Actomyosin ATPase II. Fiber ty** by histochemical ATPase reactions. Muscle & Nerve 3:233–239.
Alford, E. K., Roy, R. R., Hodgson, J. A., and Edgerton, A. R. 1987. Electromyography of rat soleus medial gastrocnemius and tibialis anterior during hindlimb suspension, Exp. Neurol. 96:635–649.
Austin, N. A., and Woledge, R. C. 1978. Energy changes and muscular contraction. Physiol. Rev. 58:690–761.
Woledge, R. C. 1971. Heat production and chemical change in muscle. Prog. Biophys. Mol. Biol. 22:39–74.
Bessman, S. P., and Yeiger, P. J. 1981. Transport of energy in muscle, the phosphorylcreatine shuttle. Science 211:448–452.
Petrofsky, J. S., and Fitch, C. D. 1980. Contractile characteristics of skeletal muscles depleted of phosphocreatine. Pflüger's Arch. 384:123–129.
Mainwood, G. W., and DeZepetnek, J. T. 1985, Post tetanic response in creatine depleted rat EDL muscle. Muscle & Nerve 8:774–782.
Max, S. R. 1972. Disuse atrophy of skeletal muscle: loss of functional activity of mitochondria. Biochem. Biophys. Res. Commun. 1394–1398.
Rifenberick, D. H., Gamble, J. G., and Max, S. R. 1973. Response of mitochondrial enzymes to decreased muscular activity. Am. J. Physiol. 225:1295–1299.
Dulhunty, A. F. 1985. Excitation-contraction coupling and contractile properties in denervated rat EDL and soleus muscles. J. Muscle Res. Cell Mot. 6:207–225.
Brooke, M. H., Williamson, E., and Kaiser, K. 1971. The behavior of four fiber types in develo** and reinnervated muscle. Arch. Neurol. 25:360–366.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gupta, R.C., Misulis, K.E. & Dettbarn, W.D. Activity dependent characteristics of fast and slow muscle: biochemical and histochemical considerations. Neurochem Res 14, 647–655 (1989). https://doi.org/10.1007/BF00964874
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00964874