Abstract
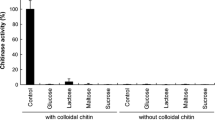
An extracellular chitin deacetylase activity has been purified to homogeneity from autolyzed cultures of Aspergillus nidulans. This enzyme is an acidic glycoprotein with a pI of 2.75 and a 28% (wt/wt) carbohydrate content. The apparent M r of the enzyme estimated by SDS/PAGE and Superose 12 (f.p.l.c.) was around 27,000. The enzyme had an optimum pH at 7.0 and was stable in the pH range 4.0–7.5. Its optimum temperature of reaction was 50°C, and it was stable from 30° to 100°C after 1 h of preincubation. The enzyme hydrolyzed glycol chitin and oligomers of N-acetylgucosamine and to a lesser extent chitin, colloidal chitin, carboxymethylchitin, and an α-1 → 3,1 → 6-N-acetylgalactosamine-galactan among other substances with amido groups, but the enzyme did not hydrolyze peptide bonds. The role of this enzyme could be deacetylation of chitin oligosaccharides during autolysis, after action of endochitinase on cell walls.
Similar content being viewed by others
Literature Cited
Araki Y, Ito E (1974) A pathway of chitosan formation in Mucor rouxii: enzymatic deacetylation of chitin. Biochem Biophys Res Commun 3:669–675
Bartnicki-García S (1968) Cell wall chemistry, morphogenesis and taxonomy of fungi. Annu Rev Microbiol 22:87–108
Calvo-Méndez C, Ruiz-Herrera J (1987) Biosynthesis of chitosan in membrane fractions from Mucor rouxii by the concerted action of chitin synthetase and a particulate deacetylase. Exp Mycol 11:128–140
Carney SL (1986) Proteoglycans. In: Chaplin MF, Kennedy JF (eds.) Carbohydrate analysis, a practical approach. Oxford-Washington D.C.:IRL Press, pp 97–141
Davis LL, Bartnicki-Garcia S (1984) Chitosan synthesis by the tandem action of chitin synthetase and chitin deacetylase from Mucor rouxii. Biochemistry 23:1065–1073
Eyler RW, Klug ED, Diephuis F (1947) Determination of degree of substitution of sodium carboxymethylcellulose. Anal Chem 19:24–27
Gooday GW, Trinci APJ (1980) Wall structure and biosynthesis in fungi: In: Gooday GW, Lloyd D, Trinci APJ (eds) The eukaryotic microbial cell. Symp. Soc. Gen. Microbiol. Vol. 30. Cambridge: Cambridge University Press, pp 207–251
Hadwiger LA, Beckman JM, Adams MJ (1981) Localization of fungal components in the pea-Fusarium interaction detected immunochemically with anti-chitosan and anti-fungal cell wall antisera. Plant Physiol 67:170–175
Hadwiger LA, Line RF (1981) The presence of “chitosan like” compounds in wheat-Puccinia striformis interactions. Physiol Plant Pathol 19:249–255
Henkeshoven J, Dernick R (1985) Simplified methods for silver staining of proteins in polyacrylamide gels and the mechanism of silver staining. Electrophoresis 6:103–112
Jeuniaux C (1966) Chitinases. Methods Enzymol 8:644–650
Jiménez MH, Weill CE (1982) A sensitive, fluorimetric analysis of amino sugars. Carbohydr Res 101:133–137
John M, Röhrir H, Schmidt J, Wieneke U, Shell J (1993) Rhizobium NodB protein involved in modulation signal synthesis is a chitooligosaccharide deacetylase. Proc Natl Acad Sci USA 90:625–629
Kauss H, Jeblick W, Young DH (1983) Chitin deacetylase from the plant pathogen Collecotrichum lindemuthianum. Plant Sci Lett 28:231–236
Kauss H, Bauch B (1988) Chitin deacetylase from Colletotrichum lindemuthianum. Methods Enzymol 161:518–523
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lowry OH, Rosebrough J, Farr AL, Randall RJ (1981) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Molano J, Polacheck I, Duran A, Cabib E (1979) An endochitinase from wheat germ. Activity on nascent and performed chitin. J Biol Chem 254:4901–4907
Reyes F, Calatayud J, Martínez MJ (1989) Endochitinase from Aspergillus nidulans implicated in the autolysis of its cell wall. FEMS Microbiol Lett 60:119–124
Reyes F, Calatayud J, Vazquez C, Martínez MJ (1989) β-N-Acetylglucosaminidase from Aspergillus nidulans which degrades chitin oligomers during autolysis. FEMS Microbiol Lett 65:83–88
Ride JP, Drysdale RB (1972) A rapid method for the chemical estimation of filamentous fungi in plant tissue. Physiol Plant Pathol 2:7–15
Ruiz-Herrera J (1982) Synthesis of chitin microfibrils in vitro. In: Brown RM (ed) Cellulose and other natural polymer systems. New York: Plenum, pp 207–223
Rupérez P, Leal JA (1979) Utilization of nitrogen sources by Aspergillus nidulans for extracellular polysaccharide production. Trans Br Mycol Soc 72:291–297
Rupley JA (1964) The hydrolysis of chitin by concentrated hydrochloric acid and the preparation of low-molecular-weight substrates for lysozymes. Biochim Biophys Acta 83:245–255
Shibata Y, Nisizawa K (1965) Microheterogeneity of β-glucosidases in apricot emulsin. Arch Biochem Biophys 109:516–521
Siegrist J, Kauss H (1990) Chitin deacetylase in cucumber leaves infected by Colletotrichum lagenarium. Physiol Mol Plant Pathol 36:267–275
To-Anun Ch, Toyoda H, Nishiguchi T, Chan SH, Fukamizo T, Ouchi S (1992) Isolation and regeneration of Fusarium oxysporum f. sp. lycopersici protoplats. Nippon Shokobutsu Byori Gakkaiko 57:609–642
Tracey MV (1955) Chitin. In: Paech K, Tracey MV (eds) Modern methods of plant analysis Vol. 2. Berlin: Springer Verlag, pp 264–274
Trevelyan WE, Harrison JS (1952) Studies on yeast metabolism. 1. Fractionation and microdetermination of cell carbohydrates. Biochem J 50:298–303
Trudel J, Asselin A (1990) Detection of chitin deacetylase activity after polyacrylamide gel electrophoresis. Anal Biochem 189:249–253
Trujillo R (1968) Preparation of carboxymethylchitin. Carbohydr Res 7:483–485
Zacharius RM, Zell TE, Morrison JH, Woodlock JJ (1969) Glycoprotein staining following electrophoresis on acrylamide gels. Anal Biochem 30:148–162
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alfonso, C., Nuero, O.M., Santamaría, F. et al. Purification of a heat-stable chitin deacetylase from Aspergillus nidulans and its role in cell wall degradation. Current Microbiology 30, 49–54 (1995). https://doi.org/10.1007/BF00294524
Issue Date:
DOI: https://doi.org/10.1007/BF00294524