Summary
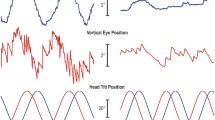
Off-vertical rotation (OVAR) in darkness induced continuous horizontal nystagmus in humans at small tilts of the rotation axis (5 to 30 degrees). The horizontal slow eye velocity had two components: a mean velocity in the direction opposite to head rotation and a sinusoidal modulation around the mean. Mean velocity generally did not exceed 10 deg/s, and was less than or equal to the maximum velocity of optokinetic after-nystagmus (OKAN). Both the mean and modulation components of horizontal nystagmus increased with tilt angle and rotational velocity. Vertical slow eye velocity was also modulated sinusoidally, generally around zero. The amplitude of the vertical modulation increased with tilt angle, but not with rotational velocity. In addition to modulations in eye velocity, there were also modulations in horizontal and vertical eye positions. These would partially compensate for head position changes in the yaw and pitch planes during each cycle of OVAR. Modulations in vertical eye position were regular, increased with increases in tilt angle and were separated from eye velocity by 90 deg. These results are compatible with the interpretation that, during OVAR, mean slow velocity of horizontal nystagmus is produced by the velocity storage mechanism in the vestibular system. In addition, they indicate that the otolith organs induce compensatory eye position changes with regard to gravity for tilts in the pitch, yaw and probably also the roll planes. Such compensatory changes could be utilized to study the function of the otolith organs. A functional interpretation of these results is that nystagmus attempts to stabilize the image on the retina of one point of the surrounding world. Mean horizontal velocity would then be opposite to the estimate of head rotational velocity provided by the output of the velocity storage mechanism, as charged by an otolithic input during OVAR. In spite of the lack of actual translation, an estimate of head translational velocity could, in this condition, be constructed from the otolithic signal. The modulation in horizontal eye position would then be compensatory for the perceived head translation. Modulation of vertical eye velocity would compensate for actual changes in head orientation with respect to gravity.
Similar content being viewed by others
References
Baloh RW, Lyerly K, Yee RD, Honrubia V (1984) Voluntary control of the human vestibulo-ocular reflex. Acta Otolaryngol (Stockh) 97: 1–6
Barr CC, Schulteis LW, Robinson DA (1976) Voluntary nonvisual control of the human vestibulo-ocular reflex. Acta Otolaryngol (Stockh) 81: 365–375
Benson AJ (1974) Modification of the response to angular accelerations by linear accelerations. In: Kornhuber H (ed) Handbook of sensory physiology, Vol VI/2. Springer, Berlin Heidelberg New York, pp 281–320
Benson AJ, Barnes GR (1973) Responses to rotating linear acceleration vectors considered in relation to a model of the otolith organs. In: 5th symposium on the role of the vestibular organs in space exploration. NASA SP 314
Benson AJ, Bodin MA (1966) Interaction of linear and angular accelerations on vestibular receptors in man. Aerosp Med 37: 144–154
Buizza A, Leger A, Droulez J, Berthoz A, Schmid R (1980) Influence of otolithic stimulation by horizontal linear acceleration on optokinetic nystagmus and visual motion perception. Exp Brain Res 39: 509–522
Chun K-S, Robinson DA (1978) A model of quick phase generation in the vestibulo-oculr reflex. Biol Cybern 28: 209–231
Cohen B, Raphan T, Henn V, Dennett D (1981) Velocity storage nystagmus and visual vestibular interactions in humans. Ann NY Acad Sci 374: 421–433
Cohen B, Suzuki J, Raphan T (1983) Role of the otolith organs in generation of horizontal nystagmus; effects of selective labyrinthine lesions. Brain Res 276: 159–164
Correia MJ, Guedry FE (1966) Modification of vestibular responses as a function of rate of rotation about an earthhorizontal axis. Acta Otolaryngol (Stockh) 62: 297–308
Correia MJ, Money KE (1970) The effect of blockage of all six semi-circular canal ducts on nystagmus produced by linear acceleration in the cat. Acta Otolaryngol (Stockh) 69: 7–16
Darlot C, Lopez-Barneo J, Tracey D (1981) Asymmetry of vertical vestibular nystagmus in the cat. Exp Brain Res 41: 420–426
Fernández C, Goldberg JM (1976a) Physiology of peripheral neurons innervating otolith organs of the squirrel monkey. I. Response to the static tilt and to long-duration centrifugal force. J Neurophysiol 39: 970–984
Fernández C, Goldberg JM (1976b) Physiology of peripheral neurons innervating otolith organs of the squirrel monkey. II. Directional selectivity and force-response relations. J Neurophysiol 39: 985–995
Fernández C, Goldberg JM (1976c) Physiology of peripheral neurons innervating otolith organs of the squirrel monkey. III. Response dynamics. J Neurophysiol 39: 996–1008
Fernández C, Goldberg JM, Abend WK (1972) Response to static tilt of peripheral neurons innervating otolith organs of the squirrel monkey. J Neurophysiol 35: 978–997
Goldberg JM, Fernández C (1981) Physiological mechanisms of the nystagmus produced by rotations about an earth-horizontal axis. Ann NY Acad Sci 374: 40–43
Goldberg JM, Fernández C (1982) Eye movements and vestibular nerve responses produced in the squirrel monkey by rotation about an earth-horizontal axis. Exp Brain Res 46: 393–402
Gresty MA, Bronstein A (1986) Otolith stimulation evokes compensatory reflex eye movements of high velocity when linear motion of the head is combined with concurrent angular motion. Neurosci Lett 65: 149–154
Gresty MA, Bronstein AM, Baratt H (1987) Eye movement responses to combined linear and angular head movement. Exp Brain Res 65: 337–384
Guedry FE (1966) Influence of linear and angular accelerations on nystagmus. In: 2nd symposium on the role of the vestibular organs in space exploration. NASA SP 115: 185–198
Guedry FE (1974) Psychophysics of the vestibular sensation. In: Kornhuber H (ed) Handbook of sensory physiology, Vol VI/ 2. Springer, Berlin Heidelberg New York, pp 3–154
Guedry FE (1965) Orientation of the rotation axis relative to gravity: its influence on nystagmus and the sense of rotation. Acta Otolaryngol (Stockh) 60: 30–48
Guitton D, Volle M (1987) Gaze control in humans: eye-head coordination during orienting movements to targets within and beyond the oculomotor range. J Neurophysiol 58: 427–459
Hain TC (1986) A model of the nystagmus induced by off vertical axis rotation. Biol Cybern 54: 337–350
Harris LR (1987) Vestibular and optokinetic eye movements evoked in the cat by rotation about a tilted axis. Exp Brain Res 66: 522–532
Harris LR, Barnes GB (1986) The orientation of vestibular nystagmus is modified by head tilt. Int. Meeting Barany Soc., Ann Arbor, Michigan
Jell RM, Ireland DJ, Lafortune S (1985) Human optokinetic after-nystagmus. Acta Otolaryngol (Stockh) 99: 95–102
Krejcova H, Highstein S, Cohen B (1971) Labyrinthine and extra-labyrinthine effects on ocular counter-rolling. Acta Otolaryngol (Stockh) 72: 165–171
Markham CH, Diamond SG (1983) Do the eyes counterroll during barbecue rotation? Soc Neurosci Abstr 9: 868
Markham CH, Diamond SG (1985) Dynamic ocular counterrolling. In: Igarashi, Black (eds) Vestibular and visual control on posture and locomotor equilibrium. 7th Int. Symp. Int. Soc. Posturography, Houston, Tex, 1983, pp 246–250
Matsuo V, Cohen B (1984) Vertical optokinetic nystagmus and vestibular nystagmus in the monkey: up-down as asymmetry and effects of gravity. Exp Brain Res 53: 197–216
Mayne R (1974) A system concept of the vestibular organs. In: Kornhuber HH (ed) Handbook of sensory physiology, Vol VI/2. The vestibular system. Springer, New York, pp 493–580
Meiry JL (1965) The vestibular system and human dynamic space orientation. Thesis. M.I.T. Cambridge, Massachusetts
Melvill-Jones G, Berthoz A, Segal B (1984) Adaptive modification of the vestibulo-ocular reflex by mental effort in darkness. Exp Brain Res 56: 149–153
Niven JI, Hixson WC, Correia MJ (1965) Elicitation of horizontal nystagmus by periodic linear acceleration. Acta Otolaryngol (Stockh) 62: 429–441
Raphan T, Cohen B (1985) Velocity storage and the ocular response to multidimensional vestibular stimuli. Rev Oculomotor Res 1: 123–143
Raphan T, Schnalbok C (1988) Modelling slow phase velocity generations during off vertical axis rotation (OVAR). In: Representation of three dimensional space in the vestibular, oculomotor and visual systems. Ann NY Acad Sci (in press)
Raphan T, Cohen B, Henn V (1981a) Effects of gravity on rotatory nystagmus in monkeys. Ann NY Acad Sci 374: 337–346
Raphan T, Matuso V, Cohen B (1979) Velocity storage in the vestibulo-ocular reflex arc. Exp Brain Res 35: 229–248
Raphan T, Waespe W, Cohen B (1981b) Vestibular nerve activity induced by off-vertical axis rotations. Soc Neurosci Abstr 7: 39
Saito I, Wada H, Yagura S, Kato N (1968) On the influences of gravitational acceleration to nystagmus. Jpn J Aerosp Med Psy
Stockwell CW, Turnipseed GT, Guedry FE (1970) Nystagmus responses during rotation about a tilted axis. NAMRL 1129. USAARL 71-15, Pensacola, Fla., Naval
Viirre E, Tweed D, Milner K, Vilis T (1986) A re-examination of the gain of the vestibulo-ocular reflex. J Neurophysiol 56: 439–450
Wall C, Black O (1984) The modulation component of nystagmus during earth-horizontal rotation: relationship with gaze angle. Acta Otolaryngol (Stockh) 97: 193–201
Young LR (1967) Effects of linear acceleration on vestibular nystagmus. 3rd symposium on the role of the vestibular organ in space exploration. NASA SP 152
Young L (1972) Cross coupling between effects of linear and angular acceleration on vestibular nystagmus. Bibl Ophthalmol 82: 116–121
Young L, Henn V (1975) Nystagmus produced by pitch and yaw rotation of monkeys about non-vertical axes. Fortschr Zool 23: 235–246
Young L, Meiry JL (1968) A revised dynamic otolith model. 3rd symposium on the role of the vestibular organ in space exploration. NASA SP 152
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Darlot, C., Denise, P., Droulez, J. et al. Eye movements induced by off-vertical axis rotation (OVAR) at small angles of tilt. Exp Brain Res 73, 91–105 (1988). https://doi.org/10.1007/BF00279664
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00279664