Abstract
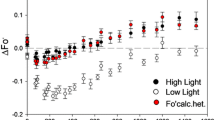
Maximum quantum yields (QY) of photosynthetic electron flows through PSI and PSII were separately assessed in thylakoid membranes isolated from leaves of Cucumis sativus L. (cucumber) that had been chilled in various ways. The QY(PSI) in the thylakoids prepared from the leaves treated at 4° C in moderate light at 220 μmol quanta·m−2·s−1 (400–700 nm) for 5 h, was about 20–30% of that in the thylakoids prepared from untreated leaves, while QY(PSII) decreased, at most, by 20% in response to the same treatment. The decrease in QY(PSI) was observed only when the leaves were chilled at temperatures below 10° C, while such a marked temperature dependency was not observed for the decrease in QY(PSII). In the chilling treatment at 4° C for 5 h, the quantum flux density that was required to induce 50% loss of QY (PSI) was ca. 50 umol quanta·m−2·s−1. When the chilling treatment at 4° C in the light was conducted in an atmosphere of N2, photoinhibition of PSI was largely suppressed, while the damage to PSII was somewhat enhanced. The ferricyanide-oxidised minus ascorbate-reduced difference spectra and the light-induced absorbance changes at 700 nm obtained with the thylakoid suspension, indicated the loss of P700 to extents that corresponded to the decreases in QY(PSI). Accordingly, the decreases in QY(PSI) can largely be attributed to destruction of the PSI reaction centre itself. These results clearly show that, at least in cucumber, a typical chillingsensitive plant, PSI is much more susceptible to aerobic photoinhibition than PSII.
Similar content being viewed by others
Abbreviations
- DCMU:
-
3-(3,4-dichlorophenyl)-1,1-dimethylurea
- P700:
-
primary electron donor of PSI
- PPFD:
-
photosynthetically active photon flux density
- QY:
-
quantum yield
References
Asada, K. (1992) Production and scavenging of active oxygen in chloroplasts. In: Molecular biology of free radical scavenging systems, pp. 173–192, Cold Spring Harbor Laboratory Press, New York
Asada, K., Takahashi, M. (1987) Production and scavenging of active oxygen in photosynthesis. In: Photoinhibition, pp. 227–287, Kyle, D.J., Osmond, C.B., Arntzen, C.J. eds. Elsevier, Amsterdam
Chow, W.S. (1994) Photoprotection and photoinhibitory damage. In: Molecular processes of photosynthesis. Barber, J. ed, JAI Press, Greenwich, Connecticut, in press
Garber, M.P. (1977) Effect of light and chilling temperatures on chilling-sensitive and chilling-resistant plants. Plant Physiol. 59, 981–985
Gerber, D.W., Burris, J.E. (1981) Photoinhibition and P700 in the marine diatom Amphora sp. Plant Physiol. 68, 699–702
Giovanelli, J., San Pietro, A. (1959) Photosynthetic pyridine nucleotide reductase II. Photoinactivation in the presence of chloroplasts. Arch. Biochem. Biophys. 843, 471–485
Hetherington, S.E., He, J., Smillie, R.M. (1989) Photoinhibition at low temperature in chilling-sensitive and -resistant plants. Plant Physiol. 90, 1609–1615
Hodgson, R.A.J., Raison, J.K. (1989) Inhibition of photosynthesis by chilling in moderate light: a comparison of plants sensitive and insensitive to chilling. Planta 178, 545–552
Hodgson, R.A.J., Raison, J.K. (1991) Superoxide production by thylakoids during chilling and its implication in the susceptibility of plants to chilling-induced photoinhibition. Planta 183, 222–228
Hodgson, R.A.J., Orr, G.R., Raison, J.K. (1987) Inhibition of photosynthesis by chilling in the light. Plant Sci. 49, 75–79
Inoue, K., Sakurai, H., Hiyama, T. (1986) Photoinactivation sites of photosystem I in isolated chloroplasts. Plant Cell Physiol. 27, 961–968
Kaniuga, Z., Sochanowicz, B., Zabek, J., Krzystyniak, K. (1978 a) Photosynthetic apparatus in chilling-sensitive plants. I. Reactivation of Hill reaction activity inhibited on the cold and dark storage of detached leaves and intact plants. Planta 140, 121–128
Kaniuga, Z., Zabek, J., Sochanowicz, B. (1978 b) Photosynthetic apparatus in chilling-sensitive plants. III. Contribution of loosely bound manganese to the mechanism of reversible inactivation of Hill reaction activity following cold and dark storage and illumination of leaves. Planta 144, 49–56
Kislyuk, I.M., Vas'kovskii, M.D. (1972) Effect of cooling cucumber leaves on photosynthesis and photochemical reaction. Soviet Plant Physiol. 191, 688–692 (Fiziol. Rast. 19, 813–818)
Krause, G.H., Köster, S., Wong, S.C. (1985) Photoinhibition of photosynthesis under anaerobic conditions studied with leaves and chloroplasts of Spinada oleracea L. Planta 165, 430–438
Margulies, M.M. (1972) Effect of cold-storage of bean leaves on photosynthetic reactions of isolated chloroplasts. Inability to donate electrons to photosystem II and relation to manganese content. Biochim. Biophys. Acta 267, 96–103
Margulies, M.M., Jagendorf, A.T. (1960) Effect of bean leaves on photosynthetic reaction of isolated chloroplasts. Arch. Biochem. Biophys. 90, 176–183
Ooms, J.J.J., Vredenberg, W.J., Buurneijer, W.F. (1989) Evidence for an electrogenic and a non-electrogenic component in the slow phase of the P515 response in chloroplasts. Photosynth. Res. 20, 119–128
Öquist, G., Greer, D.H., Ögren, E. (1987) Light stress at low temperature. In: Photoinhibition, pp. 67–87, Kyle, D.J., Osmond, C.B., Arntzen, C.J. eds. Elsevier, Amsterdam
Porra, R.J., Thompson, W.A., Kriedemann, P.E. (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta 975, 384–394
Peeler, T.C., Naylor, A.W. (1988) A comparison of the effects of chilling on thylakoid electron transfer in pea (Pisum sativum L.) and cucumber (Cucumis sativus L.). Plant Physiol. 86, 147–151
Petering, D., Fee, J.A., Palmer, G. (1971) The oxygen sensitivity of spinach ferredoxin and other iron-sulfur proteins. The formation of protein-bound sulfur-zero. J. Biol. Chem. 246, 643–653
Sakihama, N., Ohmori, H., Sugimoto, N., Yamasaki, Y., Ohsino, R., Shin, M. (1983) Toyopearl HW-65: ammonium sulfate as a new column Chromatographic adsorbent for enzyme purifications. J. Biochem. 93, 129–134
Scheibe, R. (1990) Light/dark modulation: Regulation of chloroplast metabolism in a new light. Bot. Acta 103, 327–334
Sassenrath, G.F., Ort, D.R., Portis, A.R. Jr. (1990) Impaired reductive activation of stromal bisphosphatases in tomato leaves following low-temperature exposure at high light. Arch. Biochem. Biophys. 282, 302–308
Satoh, Ka., Fork, D.C. (1982) Photoinhibition of reaction centers of photosystems I and II in intact Bryopsis chloroplasts under anaerobic conditions. Plant Physiol. 70, 1004–1008
Satoh, Ki. (1970 a) Mechanism of photoinactivation in photosynthetic systems. I. The dark reaction in photoinactivation. Plant Cell Physiol. 11, 15–27
Satoh, Ki. (1970 b) Mechanism of photoinactivation in photosynthetic systems. II. The occurrence and properties of two different types of photoinactivation. Plant Cell Physiol. 11, 29–38
Satoh, Ki. (1970 c) Mechanism of photoinactivation in photosynthetic systems. III. Site and mode of photoinactivation in photosystem I. Plant Cell Physiol. 11, 187–197
Satoh, Ki. (1971) Mechanism of photoinactivation in photosynthetic systems. IV. Light-induced changes in the fluorescence transients. Plant Cell Physiol. 12, 13–27
Shen, J.-R., Terashima, I., Katoh, S. (1990) The cause for dark, chilling-induced inactivation of photosynthetic oxygen-evolving system in cucumber leaves. Plant Physiol. 93, 1354–1357
Smillie, R.M., Nott, R. (1979) Assay of chilling injury in wild and domestic tomatoes based on photosystem activity of the chilled leaves. Plant Physiol. 63, 796–801
Sonoike, K., Katoh, S. (1988) Effects of sodium dodecyl sulfate and methyl viologen on the differential extinction coefficient of P-700 — a band shift of chlorophyll a associated with oxidation of P-700. Biochim. Biophys. Acta 935, 61–71
Takahashi, M., Shimura, S., Hamaguchi, Y., Fujita, Y. (1971) Photoinhibition of phytoplankton photosynthesis as a function of exposure time. J. Oceanogr. Soc. Jpn. 27, 43–50
Terashima, I., Huang, L.-K., Osmond, C.B. (1989 a) Effects of leaf chilling on thylakoid functions, measured at room temperature, in Cucumis sativus L. and Oryza sauva L. Plant Cell Physiol. 30, 841–850
Terashima, I., Shen, J.-R., Katoh, S. (1989 b) Chilling damage in cucumber (Cucumis sativus L.) thylakoids. In: Plant water relations and growth under stress, pp. 470–472, Tazawa, M., Katsumi, M., Masuda, Y., Okamoto, H., eds. Yamada Science Foundation, Osaka and Myu K.K., Tokyo
Terashima, I., Kashino, Y., Katoh, S. (1991 a) Exposure of leaves of Cucumis sativus L. to low temperatures in the light causes uncoupling of thylakoids. I. Studies with isolated thylakoids. Plant Cell Physiol. 32, 1267–1274
Terashima, I., Sonoike, K., Kawazu, T., Katoh, S. (1991 b) Exposure of leaves of Cucumis sativus L. to low temperature in the light causes uncoupling of thylakoids II. Non-destructive measurements in intact leaves. Plant Cell Physiol. 32, 1275–1283
van Hasselt, P.R., van Berlo, H.A.C. (1980) Photooxidative damage to the photosynthetic apparatus during chilling. Physiol. Plant. 50, 52–56
Wise, R.R., Naylor, A.W. (1987 a) Chilling-enhanced photooxidation. The peroxidative destruction of lipids during chilling injury to photosynthesis and ultrastructure. Plant Physiol. 87, 272–277
Wise, R.R., Naylor, A.W. (1987 b) Chilling-enhanced photooxidation. Evidence for the role of singlet oxygen and Superoxide in the breakdown of pigments and endogenous antioxidants. Plant Physiol. 83, 278–282
Wise, R.R., Ort, D.R. (1989) Photophosphorylation after chilling in the light. Plant Physiol. 90, 657–664
Author information
Authors and Affiliations
Additional information
We are grateful to invaluable comments by Prof. S. Katoh, K. Hikosaka and the members of our laboratory. We also thank A. Aoyama for technical assistance. This work was partly supported by the grants from the Ministry of Education, Science, and Culture, Japan, to I. Terashima (#03740342 and #04640621).
Rights and permissions
About this article
Cite this article
Terashima, I., Funayama, S. & Sonoike, K. The site of photoinhibition in leaves of Cucumis sativus L. at low temperatures is photosystem I, not photosystem II. Planta 193, 300–306 (1994). https://doi.org/10.1007/BF00192544
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00192544