Abstract
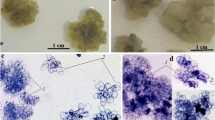
The effects of a range of plant growth regulators on callus production in various Ephedra species were examined. Species examined were E. andina, E. distachya, E. equisitina, E. fragilis var, camplyopoda, E. gerardiana, E. intermedia, E. major ssp procera, E. minima and E. saxatilis. All species produced callus on modified MS medium supplemented with 0.25 μM kinetin and 5.0 μM 2,4-dichlorophenoxyacetic acid or 1-naphthaleneacetic acid. Neither indole-3-acetic acid nor 3-indolebutyric acid induced significant callus formation but the latter maintained growth of established callus cultures in several species. Suspension cultures of several species were established in MS medium supplemented with 0.25 μM kinetin and 5.0 μM 2,4-dichlorophenoxyacetic acid or 1-naphthaleneacetic acid. Sustained fresh weight doubling times of 70±7h were recorded for cell suspension cultures of E. andina grown in a semi-continous air-lift bubble bioreactor and a minimum doubling time of 56 h was recorded for E. andina in batch culture. It also proved possible to immobilise E. andina batch cultures in sodium alginate beads.
Neither parent plants or in vitro cultures of E. distachya, E. fragilis or E. saxatilis produced alkaloids. Trace quantities of 1-ephedrine and trace-0.14% dwt d-pseudoephedrine were produced by in vitro cultures of other species. The ability to produce alkaloid diminished to zero with successive subcultures.
Similar content being viewed by others
Abbreviations
- Eph:
-
1-ephedrine
- Peph:
-
d-pseudoephedrine
- RGR:
-
relative growth rate
- KIN:
-
kinetin
- 2,4-d :
-
2,4-dichlorophenoxyacetic acid
- NAA:
-
1-naphthaleneacetic acid
- IBA:
-
3-in-dolebutyric acid
- IAA:
-
indole-3-acetic acid
References
Barkan S, Weber JD & Smith E (1981) Determination of cross-contamination of the diastereomers ephedrine and pseudoephedrine by high performance liquid chromatography, thin layer chromatography and carbon-13 nuclear magnetic resonance spectroscopy. J. Chromatogr. 219: 81–88
Bhatnagar SP & Singh MN (1984) Organogenesis in the cultured female gametophyte of Ephedra foliata. J. Exp. Bot. 35: 268–278
Darlington CD & Wylie AP (1955) Chromosome Atlas of Flowering Plants (p. 7) (2nd edition). George Allen and Unwin, London
Dixon RA (1985) Plant Cell Culture-a practical approach (p.18) IRL Press, Oxford
Evans WC (1989) Alkaloids. In: Trease and Evan's Pharmacognosy (13th edition, p. 99). Balliere Tindall, London
Feldman DS, Gagnon J, Hofmann R & Simpson J (1987) Statview II. Abacus Concepts Inc., Berkeley, California
Fidgeon C & Wilson G (1987) Growth regulation of Galium mollugo L. cell suspensions by α-naphthaleneacetic acid. J. Exp. Bot. 38: 1491–1500
Khanna P & Uddin A (1976) Production of ephedrine from in vitro tissue cultures of Ephedra foliata Boiss. Proceedings of the Indian Science Congress 62: 93
Konar RN & Singh MN (1979) Production of plantlets from female gametophytes of Ephedra foliata Boiss. Z. Pflanzenphysiol. 95: 87–90
Lee TT (1971) Cytokinin controlled indoleacetic acid oxidase isoenzymes in tobacco callus cultures. Plant Physiol. (Bethesda) 47: 181–185
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissues cultures. Physiol. Plant. 15: 473–497
Nissen SJ & Sutter EG (1990) Stability of IAA and IBA in nutrient medium to several tissue culture procedures. HortScience. 25: 800–802
O'Dowd N (1991) In vitro culture, micropropagation and secondary metabolite production of Ephedra. PhD thesis. Trinity College, University of Dublin
Owens JN & Hardev V (1990) Sex expression in gymnosperms. Crit. Rev. Plant Sci. 9: 281–294
Ramawat KG & Arya HC (1976) Growth and morphogenesis in callus cultures of Ephedra gerardiana, Phytomorphology 26: 395–403
Ramawat KG & Arya HC (1977) Carbohydrate nutrition of Ephedra tissues grown in culture. Indian J. Exp. Bot. 15: 524–527
Ramawat KG & Arya HC (1979a) Alkaloid content of Ephedra in vivo and in vitro. Indian J. Exp. Bot. 17: 106–107
Ramawat KG & Arya HC (1979b) Effect of some growth regulators on ephedrine production in Ephedra gerardiana callus cultures. Indian J. Exp. Bot. 17: 227–228
Ramawat KG & Arya HC (1980) Nitrogen nutrition of Ephedra tissues grown in culture. Phytomorphology 29: 15–26
Sankhla N, Sankhla D & Chatterji UN (1967a) In vitro induction of proliferation in female gametophytic tissue of E. foliata Boiss. Naturwissenschaften 54: 203
Shukla RM (1980) Effect of light quality on ephedrine production in Ephedra foliata callus culture. Indian Drugs 17: 392–393
Singer SR (1986) Analysing growth in cell cultures I: calculating growth rates. Can. J. Bot. 64: 233–237
Straus J & Gerding RK (1963) Auxin oxidase and growth control in tissue cultures of Ephedra. Plant Physiol. (Bethesda) 38: 621–627
Syono K & Furuya T (1972) Effects of cytokinins on the auxin requirement and auxin content of tobacco calluses. Plant Cell Physiol. 13: 843–856
Teasdale RD, Dawson PA & Woolhouse HW (1986) Mineral nutrient requirements of a loblolly pine (Pinus taeda) cell suspension culture. Plant Physiol. (Bethesda) 82: 942–945
Uddin A (1977) Production of amino acids in Ephedra foliata suspension cultures. Curr. Sci. (Bangalore). 46: 825–826
Wilson G (1990) Screeing and selection of cultured plant cells for increased yields of secondary metabolites. In: Dix P (Ed) Plant Cell Line Selection. V.C.H. Weinheim
Yeomann MM (1987) Bypassing the plant. Ann. Bot. (London) 4: 157–174
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
O'Dowd, N.A., McCauley, P.G., Richardson, D.H.S. et al. Callus production, suspension culture and in vitro alkaloid yields of Ephedra . Plant Cell Tiss Organ Cult 34, 149–155 (1993). https://doi.org/10.1007/BF00036095
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00036095