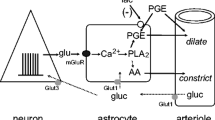
Abstract
Brain glycogen is a specialized energy buffer, rather than a conventional reserve. In the rodent optic nerve, a central white matter tract, it is located in astrocytes, where it is converted to lactate, which is then shuttled intercellularly from the astrocyte to the axon. This basic pathway was elucidated from non-physiological experiments in which the nerve was deprived of exogenous glucose. However, this shuttling also occurs under physiological conditions, when tissue energy demand is increased above baseline levels in the presence of normoglycemic concentrations of glucose. The signaling mechanism by which axons alert astrocytes to their increased energy requirement is likely to be elevated interstitial K+, the inevitable consequence of increased neuronal activity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Andersen B, Rassov A, Westergaard N, Lundgren K (1999) Inhibition of glycogenolysis in primary rat hepatocytes by 1, 4-dideoxy-1,4-imino-D-arabinitol. Biochem J 342:545–550
Andersen B, Mortensen E, Winther GM, Westergaard N (2001) Characterization of isofagomine’s inhibitory effect on glycogen phosphorylase and glycogenolysis in primary rat hepatocytes. Diabetologia 44(Suppl 1):Poster 0618
Attwell D, Laughlin SB (2001) An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab 21:1133–1145
Aubert A, Costalat R, Magistretti PJ, Pellerin L (2005) Brain lactate kinetics: modeling evidence for neuronal lactate uptake upon activation. Proc Natl Acad Sci U S A 102:16448–16453
Aubert A, Pellerin L, Magistretti PJ, Costalat R (2007) A coherent neurobiological framework for functional neuroimaging provided by a model integrating compartmentalized energy metabolism. Proc Natl Acad Sci U S A 104:4188–4193
Baltan Tekkök S, Brown AM, Ransom BR (2003) Axon function persists during anoxia in mammalian white matter. J Cereb Blood Flow Metab 23:1340–1348
Bolton S, Greenwood K, Hamilton N, Butt AM (2006) Regulation of the astrocyte resting membrane potential by cyclic AMP and protein kinase a. Glia 54:316–328
Boron WF, Boulpaep EL (2009) Medical physiology. Elsevier
Brown AM, Tekkok SB, Ransom BR (2003) Glycogen regulation and functional role in mouse white matter. J Physiol 549:501–512
Brown AM et al (2005) Astrocyte glycogen metabolism is required for neural activity during aglycemia or intense stimulation in mouse white matter. J Neurosci Res 79:74–80
Brunet JF, Allaman I, Magistretti PJ, Pellerin L (2010) Glycogen metabolism as a marker of astrocyte differentiation. J Cereb Blood Flow Metab 30:51–55
Cataldo AM, Broadwell RD (1986) Cytochemical identification of cerebral glycogen and glucose-6-phosphatase activity under normal and experimental conditions. I. Neurons and glia. J Elec Micro Tech 3:413–437
Champe PC, Harvey RA (2008) Biochemistry, 4th edn. Lippincott Williams & Wilkins, Baltimore, MD
Chia-Ching JL et al (2017) Identification of diverse astrocyte populations and their malignant analogs. Nat Neurosci 20:396–405
Choi HB et al (2012) Metabolic communication between astrocytes and neurons via bicarbonate-responsive soluble adenylyl cyclase. Neuron 75:1094–1104
Chuquet J, Quilichini P, Nimchinsky EA, Buzsaki G (2010) Predominant enhancement of glucose uptake in astrocytes versus neurons during activation of the somatosensory cortex. J Neurosci 30:15298–15303
Cummins KL, Perkel DH, Dorfman LJ (1979) Nerve fiber conduction-velocity distributions. I. Estimation based on the single-fiber and compound action potentials. Electroencephalogr Clin Neurophysiol 46:634–646
Dalsgaard MK (2006) Fuelling cerebral activity in exercising man. J Cereb Blood Flow Metab 26:731–750
Dalsgaard MK et al (2004) A reduced cerebral metabolic ratio in exercise reflects metabolism and not accumulation of lactate within the human brain. J Physiol 554:571–578
Davies P (1998) The fifth miracle. Penguin Press, London
Dienel GA (2012) Brain lactate metabolism: the discoveries and the controversies. J Cereb Blood Flow Metab 32:1107–1138
Dienel GA, Byrne JH, Roberts JL (eds) (2009) From Molecules to Networks: An Introduction to cellular and molecular neuroscience. Chapter 3. Academic Press, pp 49–110
DiNuzzo M, Mangia S, Maraviglia B, Giove F (2010) Glycogenolysis in astrocytes supports blood-borne glucose channeling not glycogen-derived lactate shuttling to neurons: evidence from mathematical modeling. J Cereb Blood Flow Metab 30:1895–1904
Dringen R, Gebhardt R, Hamprecht B (1993) Glycogen in astrocytes: possible function as lactate supply for neighboring cells. Brain Res 623:208–214
Dringen R, Peters H, Wiesinger H, Hamprecht B (1995) Lactate transport in cultured glial cells. Dev Neurosci 17:63–69
Farmer WT, Murai K (2017) Resolving astrocyte heterogeneity in the CNS. Front Cell Neurosci 11:300
Ferguson JM (2001) SSRI antidepressant medications: adverse effects and tolerability. Prim Care Companion J Clin Psychiatry 3:22–27
Fosgerau K et al (2000) Kinetic and functional characterization of 1,4-dideoxy-1, 4-imino-d- arabinitol: a potent inhibitor of glycogen phosphorylase with anti- hyperglyceamic effect in ob/ob mice. Arch Biochem Biophys 380:274–284
Frier BM, Fisher BM (2007) Hypoglycaemia in clinical diabetes, 2nd edn. John Wiley and Sons, Ltd., New York, pp 1–346
Frier BM, Heller SR, McCrimmon RJ (2014) Hypoglycaemia in clinical diabetes, 3rd edn. Wiley and Sons, Chichester
Gale J (2009) Astrobiology of Earth. Oxford University Press, Oxford
Hille B (2001) Ion channels of excitable membranes. Sinauer Associates Inc., Sunderland, MA
Hodgkin AL, Huxley AF (1952) A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol 117:500–544
Hodgkin AL, Huxley AF (1953) Movement of radioactive potassium and membrane current in a giant axon. J Physiol 121:403–414
Hu Y, Wilson GS (1997a) Rapid changes in local extracellular rat brain glucose observed with an in vivo glucose sensor. J Neurochem 68:1745–1752
Hu Y, Wilson GS (1997b) A temporary local energy pool coupled to neuronal activity: fluctuations of extracellular lactate levels in rat brain monitored with rapid-response enzyme-based sensor. J Neurochem 69:1484–1490
E. R. Kandel, J. H. Schwartz, T. M. Jessell, S. A. Siegelbaum, A. J. Hudspeth, in Principles of neural science, E. R. Kandel, J. H. Schwartz, T. M. Jessell, S. A. Siegelbaum, A. J. Hudspeth, Eds. (McGraw Hill, New York, 2013), pp. 126–147
Karbowski J (2007) Global and regional brain metabolic scaling and its functional consequences. BMC Biol 5:1–11
Kilic K et al (2018) Inadequate brain glycogen or sleep increases spreading depression susceptibility. Ann Neurol 83:61–73
Koizumi J (1974) Glycogen in the central nervous system. Prog Histochem Cytochem 6:1–37
Koizumi J, Shiraishi H (1970a) Ultrastructural appearance of glycogen in the hypothalamus of the rabbit following chlorpromazine administration. Exp Brain Res 10:276–282
Koizumi J, Shiraishi H (1970b) Glycogen accumulation in dendrites of the rabbit pallidum following trifluoperazine administration. Exp Brain Res 11:387–391
Korf J (2006) Is brain lactate metabolized immediately after neuronal activity through the oxidative pathway? J Cereb Blood Flow Metab 26:1584–1586
Lane N (2015) The vital question. Profile Publishing, London
Magistretti PJ, Allaman I (2018) Lactate in the brain: from metabolic end-product to signalling molecule. Nat Rev Neurosci 19:235–249
Mangia S et al (2009a) Metabolic and hemodynamic events after changes in neuronal activity: current hypotheses, theoretical predictions and in vivo NMR experimental findings. J Cereb Blood Flow Metab 29:441–463
Mangia S, Simpson IA, Vannucci SJ, Carruthers A (2009b) The in vivo neuron-to-astrocyte lactate shuttle in human brain: evidence from modeling of measured lactate levels during visual stimulation. J Neurochem 109(Suppl 1):55–62
McKenna MC, Gruetter R, Sonnewald U, Waagepetersen HS, Schousboe A (2006) In: Siegel GJ, Albers RW, Brady ST, Price DL (eds) Basic neurochemistry. Academic, San Diego, CA, pp 531–558
McKhann GM 2nd, D’Ambrosio R, Janigro D (1997) Heterogeneity of astrocyte resting membrane potentials and intercellular coupling revealed by whole-cell and gramicidin-perforated patch recordings from cultured neocortical and hippocampal slice astrocytes. J Neurosci 17:6850–6863
Meakin PJ et al (2007) Fructose metabolism in the adult mouse optic nerve, a central white matter tract. J Cereb Blood Flow Metab 27:86–99
Nehlig A, Wittendorp-Rechenmann E, Lam CD (2004) Selective uptake of [14C]2-deoxyglucose by neurons and astrocytes: high-resolution microautoradiographic imaging by cellular 14C-trajectography combined with immunohistochemistry. J Cereb Blood Flow Metab 24:1004–1014
Nelson SR, Schulz DW, Passonneau JV, Lowry OH (1968) Control of glycogen levels in brain. J Neurochem 15:1271–1279
Orkand RK, Nicholls JG, Kuffler SW (1966) Effect of nerve impulses on the membrane potential of glial cells in the central nervous system of amphibia. J Neurophysiol 29:788–806
Passonneau JV, Gatfield PD, Schulz DW, Lowry OH (1967) An enzymic method for measurement of glycogen. Anal Biochem 19:315–326
Pellerin L, Magistretti PJ (1994) Glutamate uptake into astrocytes stimulates aerobic glycolysis: a mechanism coupling neuronal activity to glucose utilization. Proc Natl Acad Sci U S A 91:10625–10629
Pellerin L, Magistretti PJ (2012) Sweet sixteen for ANLS. J Cereb Blood Flow Metab 32:1152–1166
Pellerin L, Bonvento G, Chatton JY, Pierre K, Magistretti PJ (2002) Role of neuron-glia interaction in the regulation of brain glucose utilization. Diabetes Nutr Metab 15:268–273; discussion 273
Pfeiffer-Guglielmi B et al (2006) Immunocytochemical analysis of rat vagus nerve by antibodies against glycogen phosphorylase isozymes. Brain Res 1110:23–29
Pfeiffer-Guglielmi B, Francke M, Reichenbach A, Hamprecht B (2007) Glycogen phosphorylase isozymes and energy metabolism in the rat peripheral nervous system—an immunocytochemical study. Brain Res 1136:20–27
Phelps CH (1972) Barbiturate-induced glycogen accumulation in brain. An electron microscopic study. Brain Res 39:225–234
Phelps CH (1975) An ultrastructural study of methionine sulphoximine-induced glycogen accumulation in astrocytes of the mouse cerebral cortex. J Neurocytol 4:479–490
Pocock G, Richards CD (2006) Human physiology: the basis of medicine, 3rd edn. OUP, Oxford
Ransom B (2009) In: Boron WF, Boulpaep EL (eds) Medical physiology. Saunders Elsevier, Philadelphia, PA, pp 289–309
Ransom BR, Fern R (1997) Does astrocytic glycogen benefit axon function and survival in CNS white matter during glucose deprivation? Glia 21:134–141
Ransom BR, Goldring S (1973) Slow depolarization in cells presumed to be glia in cerebral cortex of cat. J Neurophysiol 36:869–878
Ransom BR, Orkand RK (1996) Glial-neuronal interactions in non-synaptic areas of the brain: studies in the optic nerve. TINS 19:352–358
Schousboe A et al (1997) Trafficking between glia and neurons of TCA cycle intermediates and related metabolites. Glia 21:99–105
Schurr A, Payne RS (2007) Lactate, not pyruvate, is neuronal aerobic glycolysis end product: an in vitro electrophysiological study. Neuroscience 147:613–619
Siegel GJ, Albers RW, Brady ST, Price DL (2006) Brain neurochemistry, 7th edn. Elsevier, London
Silverthorn DU (2007) Human physiology. Chapter 15. Pearson, San Francisco, CA, pp 500–533
Simpson IA, Carruthers A, Vannucci SJ (2007) Supply and demand in cerebral energy metabolism: the role of nutrient transporters. J Cereb Blood Flow Metab 27:1766–1791
Sonnewald U, Westergaard N, Schousboe A (1997) Glutamate transport and metabolism in astrocytes. Glia 21:56–63
Sontheimer H (2008) A role for glutamate in growth and invasion of primary brain tumors. J Neurochem 105:287–295
Sorg O, Magistretti PJ (1992) Vasoactive intestinal peptide and noradrenaline exert long-term control on glycogen levels in astrocytes: blockade by protein synthesis inhibition. J Neurosci 12:4923–4931
Sotelo-Hitschfeld T et al (2015) Channel-mediated lactate release by K+-stimulated astrocytes. J Neurosci 35:4168–4178
Stryer L (1995) Biochemistry, 4th edn. W.H. Freeman & Co., New York
Stys PK, Lopachin RM Jr (1996) Elemental composition and water content of rat optic nerve myelinated axons during in vitro post-anoxia reoxygenation. Neuroscience 73:1081–1890
Stys PK, Ransom BR, Waxman SG (1991) Compound action potential of nerve recorded by suction electrode: a theoretical and experimental analysis. Brain Res 546:18–32
Stys PK, Waxman SG, Ransom BR (1992) Ionic mechanisms of anoxic injury in mammalian CNS white matter: role of Na+ channels and Na+-Ca2+ exchanger. J Neurosci 12:430–439
Swanson RA, Choi DW (1993) Glial glycogen stores affect neuronal survival during glucose deprivation in vitro. J Cereb Blood Flow Met 13:162–169
Tekkok SB, Brown AM, Westenbroek R, Pellerin L, Ransom BR (2005) Transfer of glycogen-derived lactate from astrocytes to axons via specific monocarboxylate transporters supports mouse optic nerve activity. J Neurosci Res 81:644–652
Thorens B, Mueckler MM (2010) Glucose transporters in the 21st century. Am J Physiol Endocrinol Metab 298:E141–E145
Tymoczko JL, Berg JM, Stryer L (2015) Biochemistry: a short course, 3rd edn. W.H. Freeman and Co., New York
Vannucci SJ, Maher F, Simpson IA (1997) Glucose transporter proteins in brain: delivery of glucose to neurons and glia. Glia 21:2–21
Vilchez D et al (2007) Mechanism suppressing glycogen synthesis in neurons and its demise in progressive myoclonus epilepsy. Nat Neurosci 10:1407–1413
Waagepetersen HS, Westergaard N, Schousboe A (2000) The effects of isofagomine, a potent glycogen phosphorylase inhibitor, on glycogen metabolism in cultured mouse cortical astrocytes. Neurochem Int 36:435–440
Walls AB et al (2008) Characterization of 1,4-dideoxy-1,4-imino-d-arabinitol (DAB) as an inhibitor of brain glycogen shunt activity. J Neurochem 105:1462–1470
Wang SS et al (2008) Sha** of white matter composition by biophysical scaling constraints. J Neurosci 28:4047–4056
Wender R et al (2000) Astrocytic glycogen influences axon function and survival during glucose deprivation in central white matter. J Neurosci 20:6804–6810
Whatley SA, Hall C, Lim L (1981) Hypothalamic neurons in dissociated cell culture: the mechanism of increased survival times in the presence of non-neuronal cells. J Neurochem 36:2052–2056
Yang X et al (2014) Novel hypoglycemic injury mechanism: N-methyl-D-aspartate receptor-mediated white matter damage. Ann Neurol 75:492–507
Zhao Y et al (2017) Decreased glycogen content might contribute to chronic stress-induced atrophy of hippocampal astrocyte volume and depression-like behavior in rats. Sci Rep 7:43192
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Brown, A.M., Rich, L.R., Ransom, B.R. (2019). Metabolism of Glycogen in Brain White Matter. In: DiNuzzo, M., Schousboe, A. (eds) Brain Glycogen Metabolism. Advances in Neurobiology, vol 23. Springer, Cham. https://doi.org/10.1007/978-3-030-27480-1_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-27480-1_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-27479-5
Online ISBN: 978-3-030-27480-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)