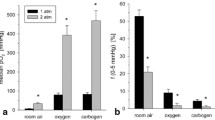
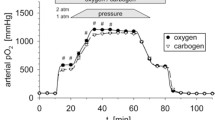
Abstract
Tumor hypoxia is an important factor limiting the efficiency of sparsely ionizing ra-diation and O2-dependent chemotherapy. Since the tumor pO2 is the result of a dynamic steady state between oxygen supply and O2 consumption of the tumor tissue, hypoxia could be reduced either by increasing the O2-supply or by reducing the O2 demand of the tumor cells. The O2 supply can be improved for instance by (i) increasing the arterial oxy-gen partial pressure, (ii) improving (and homogenizing) the tumor perfusion, or (iii) en-hancing the O2 release from blood into the tissue by right-shifting the HbO2 dissociation curve. Theoretically, it should also be possible to improve tumor oxygenation by a rela-tively small decrease in O2 consumption rate of the tumor cells. However, at present in-creasing the arterial pO2 by breathing hyperoxic gas mixtures seems to be the most effective method to improve tumor oxygenation and, thus, to enhance the efficiency of standard radio- and chemotherapy in experimental malignancies [1,7,10,11,15,24] as well as in human tumors [3,9,12,14,22,29,30]. However, since in some tumor entities oxygena-tion is inadequate and anisotropic [27], normobaric hyperoxia is often not sufficient to completely eradicate tumor hypoxia [6,16,18,26]. In these cases only breathing of hyper-oxic gases under hyperbaric conditions may be sufficient to lead to therapeutic results. On the other hand, studies on experimental tumors in animals as well as clinical trials in pa-tients showed non-uniform results concerning the therapeutic benefit of inspiratory hyper-oxia ranging from clear improvement of radiosensitivity [3,4,20,30] to no effect on therapeutic outcome [3,4]. Finally, enhancement of tumor growth by hyperbaric oxygenation has been discussed [13,23].
Supported by the Deutsche Krebshilfe (Grant M 40/91 Va 1) and the Dr.med.h.c. Erwin Braun Foundation, Basel (Grant 5.5)
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Alagoz T, Buller RE, Anderson B, Terrell KL, Squatrito RC, Niemann TH, Tatman DJ, Jebson P. Evalu-ation of hyperbaric oxygcn as a chemoscnsitizer in the treatment of epithelial ovarian cancer in xenografts in mice. Cancer 75: 2313–2322 (1995).
Brizel DM, Lin S, Johnson JL, Brooks J, Dewhirst MW, Piantadosi CA. The mechanisms by which hyper-baric oxygen and carbogen improve tumour oxygenation. Brit. J. Cancer 72: 1120–1124 (1995).
Denham JW, Yeoh EK, Wittwer G, Ward GG, Ahmad AS, Harvey ND. Radiation therapy in hyperbaric oxygen for head and neck cancer at Royal Adelaide Hospital-1964 to 1980. Int. J. Radiat. Oncol. Biol.Phys. 73:201–208(1987).
Dishe S. What have we learnt from hyperbaric oxygen? Radiother. Oncol. 20 (Suppl.): 71–74 (1991).
Dische S, Anderson PJ, Sealy R, Watson ER. Carcinoma of the cervix-anaemia, radiotherapy and hyper-baric oxygen. Br. J. Radiol. 56: 251–255 (1983).
Falk SJ, Ward R, Bleehen NM. The influence of carbogen breathing on tumour tissue oxygenation in man evaluated by computerised pO2 histography. Br. J. Cancer 66: 919–924 (1992).
Gerweck LE, Hetzel FW. pO2 in irradiated versus nonirradiated tumors of mice breathing oxygen at normal and elevated pressure. Int. J. Radiat. Oncol. Biol. Phys. 32: 695–701 (1995).
Grau C, Horsman MR, Overgaard J. Improving the radiation response in a C3H mouse mammary carci-noma by normobaric oxygen or carbogen breathing. Int. J. Radiat. Oncol. Biol. Phys. 22: 415–419 (1992).
Henk JM. Late results of a trial of hyperbaric oxygen and radiotherapy in head and neck cancer: a rationale for hypoxic cell sensitizers? Int. J. Radiat. Oncol. Biol. Phys. 12: 1339–1341 (1986).
Horsman MR, Nordsmark M, Khalil AA, Hill SA, Chaplin DJ, Siemann DW, Overgaard J. Reducing acute and chronic hypoxia in tumors by combining nicotinamide with carbogen breathing. Acta Oncol. 33: 371–376(1994).
Kjellen E, Joiner MC, Collier JM, Johns H, Rojas A. A therapeutic benefit from combining normobaric car-bogen or oxygen with nicotinamide in fractionated X-ray treatments.Radiother. Oncol. 22: 81–91 (1991).
Laurence VM, Ward R, Dennis IF, Bleehen NM. Carbogen breathing with nicotinamide improves the oxy-gen status of tumours in patients. Br. J. Cancer 72: 198–205 (1995).
Margaretten NC, Witschi HP. Effects of hyperoxia on growth characteristics of metastatic murine tumors in the lung. Cancer Res. 48: 2779–2783 (1988).
Martin L, Lartigau E, Weeger P, Lambin P, Le Ridnat AM, Lusinchi A, Wibault P, Eschwege F, Luboinski B, Guichard M. Changes in the oxygenation of head and neck tumors during carbogen breathing. Ra-diother. Oncol. 27: 123–130(1993).
Martin LM, Thomas CD, Guichard M. Nicotinamide and carbogen: relationship between pO-2 and radiosen-sitivity in three tumour lines. Int. J. Radiat. Biol. 65: 379–386 (1994).
Mueller-Klieser W, Vaupel P. Tumor oxygenation under normobaric and hyperbaric conditions. Br. J. Ra-diol. 56:559–564(1983).
Overgaard J. Sensitization of hypoxic tumour cells—clinical experience. Int. J. Radiat. Biol. 56: 801–811 (1989).
Perry PM, Nias AH. The optimum pressure of oxygen for radiotherapy of a mouse tumour. Br. J. Radiol. 65:784–786 (1992).
Rockwell S, Irvin CG, Kelley M, Hughes CS, Yabuki H, Porter E, Fischer JJ. Effects of hyperbaric oxygen and a perfluorooctylbromide emulsion on the radiation responses of tumors and normal tissues in rodents. Int. J. Radiat. Oncol. Biol. Phys. 22: 87–93 (1992).
Rojas A, Joiner MC, Hodgkiss RJ, Carl U, Kjellen E, Wilson GD. Enhancement of tumor radiosensitivity and reduced hypoxia-dependent binding of a 2-nitroimidazole with normobaric oxygen and carbogen: a therapeutic comparison with skin and kidneys. Int. J. Radiat. Oncol. Biol. Phys. 23: 361–366 (1992).
Rojas A, Stewart FA, Smith KA, Soranson JA, Randhawa VS, Stratford MR, Denekamp J. Effect of ane-mia on tumor radiosensitivity under normo-and hyperbaric conditions. Int. J. Radiat. Oncol. Biol. Phys. 13: 1681–1689(1987).
Sealy R, Cridland S, Barry L, Norris R. Irradiation with misonidazole and hyperbaric oxygen: final report on a randomized trial in advanced head and neck cancer. Int. J. Radiat. Oncol. Biol. Phys. 12: 1343–1346 (1986).
Sklizovic D, Sanger JR, Kindwall EP, Fink JG, Grunert BK, Campbell BH. Hyperbaric oxygen therapy and squamous cell carcinoma cell line growth. Head Neck 15: 236–240 (1993).
Teicher BA, Sotomayor EA, Robinson MF, Dupuis NP, Schwartz GN, Frei III E. Tumor oxygenation and radiosensitization by pentoxifylline and perflubron emulsion/carbogen breathing. Int. J. Oncol. 2: 13–21 (1993).
Teicher BA, Holden SA, Cathcart KN, Herman TS. Effect of various oxygenation conditions and fluosol-DA on cytotoxicity and antitumor activity of bleomycin in mice. J. Natl. Cancer Inst. 80: 599–603 (1988).
Thews O, Kelleher DK, Vaupel PW. Modulation of spatial O2 tension distribution in experimental tumors by increasing arterial O2 supply. Acta Oncol. 34: 291–295 (1995).
Thews O, Kelleher DK, Vaupel PW. pO2-map** of experimental rat tumors: visualization and statistical analysis. In: PW Vaupel, DK Kelleher, M Günderoth (eds.). Tumor oxygenation. Gustav Fischer, Stuttgart, 27–38(1995).
Vaupel P, Schienger K, Knoop C, Hückel M. Oxygenation of human tumors: Evaluation of tissue oxygen distribution in breast cancers by computerized O2 tension measurements. Cancer Res. 51: 3116–3122 (1991).
Voute PA, van der Kleij AJ, De Kraker J, Hoefnagel CA, Tiel van Buul MM, Van Gennip H. Clinical expe-rience with radiation enhancement by hyperbaric oxygen in children with recurrent neuroblastoma stage IV Eur. J. Cancer 31A: 596–600(1995).
Whittle RJ, Fuller AP, Foley RR. Glottic cancer: results of treatment with radiotherapy in air and hyper-baric oxygen. Clin. Oncol. Roy. Coll. Radiol. 2: 214–219 (1990).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1997 Springer Science+Business Media New York
About this chapter
Cite this chapter
Thews, O., Kelleher, D.K., Vaupel, P. (1997). Tumor Oxygenation Under Normobaric and Hyperbaric Hyperoxia. In: Harrison, D.K., Delpy, D.T. (eds) Oxygen Transport to Tissue XIX. Advances in Experimental Medicine and Biology, vol 428. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-5399-1_12
Download citation
DOI: https://doi.org/10.1007/978-1-4615-5399-1_12
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-7465-7
Online ISBN: 978-1-4615-5399-1
eBook Packages: Springer Book Archive