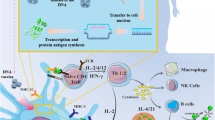
Abstract
Block copolymers are segments of two or more polymers connected through covalent bonds. These polymers enable various features such as wetting, thickening, emulsifying, stabilizing, and dispersing agents. Using these unique physicochemical properties to deliver drugs, genes, and proteins through vaccines is a challenging and exciting approach in nanobiotechnology. The development and delivery of safe and effective polymeric vaccine delivery require an in-depth knowledge of the relationship between the structure and function of the polymer carriers and how they interact with antigen-presenting cells. This chapter is about how vaccines made with block copolymers can change or boost immune responses to existing vaccines or vaccines that are still being made.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Akash MSH, Rehman K, Chen S (2014) Pluronic F127-based thermosensitive gels for delivery of therapeutic proteins and peptides. Polym Rev 54:573–597. https://doi.org/10.1080/15583724.2014.927885
Andima M, Costabile G, Isert L, Ndakala A, Derese S, Merkel O (2018) Evaluation of β-Sitosterol loaded PLGA and PEG-PLA nanoparticles for effective treatment of breast cancer: preparation, physicochemical characterization, and antitumor activity. Pharmaceutics 10:232. https://doi.org/10.3390/pharmaceutics10040232
Barnier Quer C, Robson Marsden H, Romeijn S, Zope H, Kros A, Jiskoot W (2011) Polymersomes enhance the immunogenicity of influenza subunit vaccine. Polym Chem 2:1482. https://doi.org/10.1039/c1py00010a
Biernat L, Grattan VT, Hixon MS, Prensky Z, Vaino AR (2022) A randomized, double-blind, placebo controlled, phase 1 study of the safety, tolerability, pharmacokinetics, and pharmacodynamics of LB-102, a selective dopamine D(2/3)/5-HT(7) inhibitor. Psychopharmacology 239:3009–3018. https://doi.org/10.1007/s00213-022-06185-7
Bobbala S, Tamboli V, McDowell A, Mitra AK, Hook S (2016) Novel injectable pentablock copolymer based thermoresponsive hydrogels for sustained release vaccines. AAPS J 18:261–269. https://doi.org/10.1208/s12248-015-9843-4
Bradford EB, McKeever LD (1971) Block copolymers. Prog Polym Sci 3:109–143. https://doi.org/10.1016/0079-6700(71)90003-7
Brig K (2021) Michael Bennett, War against smallpox: Edward Jenner and the global spread of vaccination. J Hist Med Allied Sci 76:224–225. https://doi.org/10.1093/jhmas/jrab010
Cao W, He L, Cao W, Huang X, Jia K, Dai J (2020) Recent progress of graphene oxide as a potential vaccine carrier and adjuvant. Acta Biomater 112:14–28. https://doi.org/10.1016/j.actbio.2020.06.009
Cho WS, Dart K, Nowakowska DJ, Zheng X, Donaldson K, Howie SEM (2012) Adjuvanticity and toxicity of cobalt oxide nanoparticles as an alternative vaccine adjuvant. Nanomedicine 7:1495–1505. https://doi.org/10.2217/nnm.12.35
Christian NA, Milone MC, Ranka SS, Li G, Frail PR, Davis KP, Bates FS, Therien MJ, Ghoroghchian PP, June CH, Hammer DA (2007) Tat-functionalized near-infrared emissive polymersomes for dendritic cell labeling. Bioconjug Chem 18:31–40. https://doi.org/10.1021/bc0601267
ClinicalTrials.gov. Study of ThermoDox with standardized Radiofrequency Ablation (RFA) for Treatment of Hepatocellular Carcinoma (HCC). Tabular View. n.d.
Diwan M (2002) Enhancement of immune responses by co-delivery of a CpG oligodeoxynucleotide and tetanus toxoid in biodegradable nanospheres. J Control Release 85:247–262. https://doi.org/10.1016/S0168-3659(02)00275-4
Eetezadi S, Ekdawi SN, Allen C (2015) The challenges facing block copolymer micelles for cancer therapy: in vivo barriers and clinical translation. Adv Drug Deliv Rev 91:7–22. https://doi.org/10.1016/j.addr.2014.10.001
Fawzi Kabil M, Nasr M, El-Sherbiny IM (2021) Conventional and hybrid nanoparticulate systems for the treatment of hepatocellular carcinoma: an updated review. Eur J Pharm Biopharm 167:9–37. https://doi.org/10.1016/j.ejpb.2021.07.003
Ferreira SA, Gama FM, Vilanova M (2013) Polymeric nanogels as vaccine delivery systems. Nanomedicine 9:159–173. https://doi.org/10.1016/j.nano.2012.06.001
Gandhi A, Paul A, Sen SO, Sen KK (2015) Studies on thermoresponsive polymers: phase behaviour, drug delivery and biomedical applications. Asian J Pharm Sci 10:99–107. https://doi.org/10.1016/j.ajps.2014.08.010
Guo S, Fu D, Utupova A, Sun D, Zhou M, ** Z, Zhao K (2019) Applications of polymer-based nanoparticles in the vaccine field. Nanotechnol Rev 8:143–155. https://doi.org/10.1515/ntrev-2019-0014
Gupta NK, Tomar P, Sharma V, Dixit VK (2011) Development and characterization of chitosan coated poly-(ɛ-caprolactone) nanoparticulate system for effective immunization against influenza. Vaccine 29:9026–9037. https://doi.org/10.1016/j.vaccine.2011.09.033
Heath PT, Galiza EP, Baxter DN, Boffito M, Browne D, Burns F, Chadwick DR, Clark R, Cosgrove C, Galloway J, Goodman AL, Heer A, Higham A, Iyengar S, Jamal A, Jeanes C, Kalra PA, Kyriakidou C, McAuley DF, Meyrick A, Minassian AM, Minton J, Moore P, Munsoor I, Nicholls H, Osanlou O, Packham J, Pretswell CH, San Francisco Ramos A, Saralaya D, Sheridan RP, Smith R, Soiza RL, Swift PA, Thomson EC, Turner J, Viljoen ME, Albert G, Cho I, Dubovsky F, Glenn G, Rivers J, Robertson A, Smith K, Toback S (2021) Safety and efficacy of NVX-CoV2373 Covid-19 vaccine. N Engl J Med 385:1172–1183. https://doi.org/10.1056/nejmoa2107659
Illum L, West P, Washington C, Davis S (1989) The effect of stabilising agents on the organ distribution of lipid emulsions. Int J Pharm 54:41–49. https://doi.org/10.1016/0378-5173(89)90163-4
Jain AK, Goyal AK, Mishra N, Vaidya B, Mangal S, Vyas SP (2010) PEG–PLA–PEG block copolymeric nanoparticles for oral immunization against hepatitis B. Int J Pharm 387:253–262. https://doi.org/10.1016/j.ijpharm.2009.12.013
Jenkins C, Hwang JH, Kopp JB, Winkler CA, Cho SK (2022) Review of urate-lowering therapeutics: from the past to the future. Front Pharmacol 13:1–13. https://doi.org/10.3389/fphar.2022.925219
Kalinichev M, Donovan-Rodriguez T, Girard F, Haddouk H, Royer-Urios I, Schneider M, Poli S, Bate ST, Marker C, Pomonis JD (2017) ADX71943 and ADX71441, novel positive allosteric modulators of the GABABreceptor with distinct central/peripheral profiles, show efficacy in the monosodium iodoacetate model of chronic osteoarthritis pain in the rat. Eur J Pharmacol 795:43–49. https://doi.org/10.1016/j.ejphar.2016.11.056
Kojarunchitt T, Baldursdottir S, Dong Y-D, Boyd BJ, Rades T, Hook S (2015) Modified thermoresponsive Poloxamer 407 and chitosan sol-gels as potential sustained-release vaccine delivery systems. Eur J Pharm Biopharm 89:74–81. https://doi.org/10.1016/j.ejpb.2014.11.026
Kudlay D, Svistunov A (2022) COVID-19 vaccines: an overview of different platforms. Bioengineering 9:72. https://doi.org/10.3390/bioengineering9020072
Kumar N, Ravikumar MNV, Domb AJ (2001) Biodegradable block copolymers. Adv Drug Deliv Rev 53:23–44. https://doi.org/10.1016/S0169-409X(01)00219-8
Kumar R, Sahoo GC, Pandey K, Das V, Das P (2015) Study the effects of PLGA-PEG encapsulated Amphotericin B nanoparticle drug delivery system against Leishmania donovani. Drug Deliv 22:383–388. https://doi.org/10.3109/10717544.2014.891271
Lal M, White J, Zhu C (2017) Preparing an adjuvanted thermoresponsive gel formulation for sublingual vaccination. Methods Mol Biol 1494:153–163. https://doi.org/10.1007/978-1-4939-6445-1_11
Lee ALZ, Yang C, Gao S, Wang Y, Hedrick JL, Yang YY (2020) Biodegradable cationic polycarbonates as vaccine adjuvants. ACS Appl Mater Interfaces 12:52285–52297. https://doi.org/10.1021/acsami.0c09649
Li S, Feng X, Wang J, He L, Wang C, Ding J, Chen X (2018) Polymer nanoparticles as adjuvants in cancer immunotherapy. Nano Res 11:5769–5786. https://doi.org/10.1007/s12274-018-2124-7
Liu Z, Yu L, Gu P, Bo R, Xu S, Wusiman A, Liu J, Hu Y, Wang D (2020) Surface-engineered cubosomes serve as a novel vaccine adjuvant to modulate innate immunity and improve adaptive immunity in vivo. Int J Nanomedicine 15:8595–8608. https://doi.org/10.2147/IJN.S266165
Malmsten M. Block copolymers in pharmaceutics. Elsevier; 2000. https://books.google.co.in/books?hl=en&lr=&id=HsUuCbPeiUUC&oi=fnd&pg=PA319&dq=Malmsten+M.+Block+copolymers+in+pharmaceutics.+Amphiphilic+Block+Copolymers:+Self+Assembly+and+Applications.+Amsterdam,+New+York:+Elesvier.+2000+Jan+1:319-46.&ots=Z49N_NKS38&sig.
Mata E, Igartua M, Patarroyo ME, Pedraz JL, Hernández RM (2011) Enhancing immunogenicity to PLGA microparticulate systems by incorporation of alginate and RGD-modified alginate. Eur J Pharm Sci 44:32–40. https://doi.org/10.1016/j.ejps.2011.05.015
Meng C, Zhi X, Li C, Li C, Chen Z, Qiu X, Ding C, Ma L, Lu H, Chen D (2016) Graphene oxides decorated with carnosine as an adjuvant to modulate innate immune and improve adaptive immunity in vivo. ACS Nano 10:2203–2213
Moerkerke R, Meeussen F, Koningsveld R, Berghmans H, Mondelaers W, Schacht E, Dusek K, Solc K (1998) Phase transitions in swollen networks. 3. Swelling behavior of radiation cross-linked poly(vinyl methyl ether) in water. Macromolecules 31:2223–2229. https://doi.org/10.1021/ma971512+
Ndwandwe D, Wiysonge CS (2021) COVID-19 vaccines. Curr Opin Immunol 71:111–116. https://doi.org/10.1016/j.coi.2021.07.003
Nuhn L, Vanparijs N, De Beuckelaer A, Lybaert L, Verstraete G, Deswarte K, Lienenklaus S, Shukla NM, Salyer ACD, Lambrecht BN, Grooten J, David SA, De Koker S, De Geest BG (2016) pH-degradable imidazoquinoline-ligated nanogels for lymph node-focused immune activation. Proc Natl Acad Sci U S A 113:8098–8103. https://doi.org/10.1073/pnas.1600816113
Pacifici N, Bolandparvaz A, Lewis JS (2020) Stimuli-responsive biomaterials for vaccines and immunotherapeutic applications. Adv Ther 3:2000129. https://doi.org/10.1002/adtp.202000129
Pérez O, Romeu B, Cabrera O, González E, Batista-Duharte A, Labrada A, Pérez R, Reyes LM, Ramírez W, Sifontes S, Fernández N, Lastre M (2013) Adjuvants are key factors for the development of future vaccines: lessons from the Finlay adjuvant platform. Front Immunol 4:407. https://doi.org/10.3389/fimmu.2013.00407
Pippa N, Gazouli M, Pispas S (2021) Recent advances and future perspectives in polymer-based nanovaccines. Vaccine 9:558. https://doi.org/10.3390/vaccines9060558
Reddy ST, Rehor A, Schmoekel HG, Hubbell JA, Swartz MA (2006) In vivo targeting of dendritic cells in lymph nodes with poly(propylene sulfide) nanoparticles. J Control Release 112:26–34. https://doi.org/10.1016/j.jconrel.2006.01.006
Safari F, Tamaddon AM, Zarghami N, Abolmali S, Akbarzadeh A (2016) Polyelectrolyte complexes of hTERT siRNA and polyethyleneimine: effect of degree of PEG grafting on biological and cellular activity. Artif Cells Nanomed Biotechnol 44:1561–1568. https://doi.org/10.3109/21691401.2015.1064936
Schmidt KT, Karzai F, Bilusic M, Cordes LM, Chau CH, Peer CJ, Wroblewski S, Huitema ADR, Schellens JHM, Gulley JL, Dahut WL, Figg WD, Madan RA (2022) A single-arm phase II study combining NLG207, a nanoparticle camptothecin, with enzalutamide in advanced metastatic castration-resistant prostate cancer post-enzalutamide. Oncologist 27:718–e694. https://doi.org/10.1093/oncolo/oyac100
Sellaturay P, Nasser S, Islam S, Gurugama P, Ewan PW (2021) Polyethylene glycol (PEG) is a cause of anaphylaxis to the Pfizer/BioNTech mRNA COVID-19 vaccine. Clin Exp Allergy 51:861–863. https://doi.org/10.1111/cea.13874
Shae D, Postma A, Wilson JT (2016) Vaccine delivery: where polymer chemistry meets immunology. Ther Deliv 7:193–196. https://doi.org/10.4155/tde-2016-0008
Shah RR, Hassett KJ, Brito LA (2017) Overview of vaccine adjuvants: introduction, history, and current status. Methods Mol Biol 1494:1–13. https://doi.org/10.1007/978-1-4939-6445-1_1
Shakya AK, Nandakumar KS (2013) Applications of polymeric adjuvants in studying autoimmune responses and vaccination against infectious diseases. J R Soc Interface 10:20120536. https://doi.org/10.1098/rsif.2012.0536
Shakya AK, Kumar A, Nandakumar KS (2011) Adjuvant properties of a biocompatible thermo-responsive polymer of N-isopropylacrylamide in autoimmunity and arthritis. J R Soc Interface 8:1748–1759. https://doi.org/10.1098/rsif.2011.0114
Siu L, Brody J, Gupta S, Marabelle A, Jimeno A, Munster P, Grilley-Olson J, Rook AH, Hollebecque A, Wong RKS, Welsh JW, Wu Y, Morehouse C, Hamid O, Walcott F, Cooper ZA, Kumar R, Ferté C, Hong DS (2020) Safety and clinical activity of intratumoral MEDI9197 alone and in combination with durvalumab and/or palliative radiation therapy in patients with advanced solid tumors. J Immunother Cancer 8:1–8. https://doi.org/10.1136/jitc-2020-001095
Soni V, Pandey V, Asati S, Gour V, Tekade RK (2019) Biodegradable block copolymers and their applications for drug delivery. In: Basic Fundam Drug Deliv. Elsevier, pp 401–447. https://doi.org/10.1016/B978-0-12-817909-3.00011-X
Swider E, Koshkina O, Tel J, Cruz LJ, de Vries IJM, Srinivas M (2018) Customizing poly(lactic-co-glycolic acid) particles for biomedical applications. Acta Biomater 73:38–51. https://doi.org/10.1016/j.actbio.2018.04.006
Taluja A, Youn YS, Bae YH (2007) Novel approaches in microparticulate PLGA delivery systems encapsulating proteins. J Mater Chem 17:4002. https://doi.org/10.1039/b706939a
Tan J, Ding B, Teng B, Ma P, Lin J (2022) Understanding structure–function relationships of nanoadjuvants for enhanced cancer vaccine efficacy. Adv Funct Mater 32:1–40. https://doi.org/10.1002/adfm.202111670
Thomas C, Rawat A, Hope-Weeks L, Ahsan F (2011) Aerosolized PLA and PLGA nanoparticles enhance humoral, mucosal and cytokine responses to hepatitis B vaccine. Mol Pharm 8:405–415. https://doi.org/10.1021/mp100255c
Tregoning JS, Brown ES, Cheeseman HM, Flight KE, Higham SL, Lemm N-M, Pierce BF, Stirling DC, Wang Z, Pollock KM (2020) Vaccines for COVID-19. Clin Exp Immunol 202:162–192. https://doi.org/10.1111/cei.13517
Wen PY, Reardon DA, Forst DA, Lee EQ, Haas B, Daoud T, Berthoud T, Diaz-Mitoma F, Anderson DE, Lassman AB, Iwamoto FM (2022) Evaluation of tumor responses and overall survival in patients with recurrent glioblastoma (GBM) from a phase IIa trial of a CMV vaccine immunotherapeutic candidate (VBI-1901). J Clin Oncol 40:2014. https://doi.org/10.1200/JCO.2022.40.16_suppl.2014
Wiggan O, Livengood JA, Silengo SJ, Kinney RM, Osorio JE, Huang CY-H, Stinchcomb DT (2011) Novel formulations enhance the thermal stability of live-attenuated flavivirus vaccines. Vaccine 29:7456–7462. https://doi.org/10.1016/j.vaccine.2011.07.054
Zakeri A, Kouhbanani MAJ, Beheshtkhoo N, Beigi V, Mousavi SM, Hashemi SAR, Karimi Zade A, Amani AM, Savardashtaki A, Mirzaei E, Jahandideh S, Movahedpour A (2018) Polyethylenimine-based nanocarriers in co-delivery of drug and gene: a develo** horizon. Nano Rev Exp 9:1488497. https://doi.org/10.1080/20022727.2018.1488497
Zhang X-Z, Yang Y-Y, Chung T-S (2002) The influence of cold treatment on properties of temperature-sensitive poly(N-isopropylacrylamide) hydrogels. J Colloid Interface Sci 246:105–111. https://doi.org/10.1006/jcis.2001.8063
Zhu Z (2013) Effects of amphiphilic diblock copolymer on drug nanoparticle formation and stability. Biomaterials 34:10238–10248. https://doi.org/10.1016/j.biomaterials.2013.09.015
Funding
Authors are thankful for getting financial support from BRICS, DST/IMRCD/BRICS/PILOTCall3/Nanodrug/2019, and also Indian Council of Medical Research, Division Reproductive Biology, Mental and Child Health (Department of Health Research, Ministry of Health & Family Welfare), Government of India.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Goyal, A.K., Jarande, M.S., Sahu, G., Basak, T., Kumari, P. (2023). Role of Block Copolymers in Vaccines. In: Mishra, N., Pandey, V. (eds) Block Co-polymeric Nanocarriers: Design, Concept, and Therapeutic Applications. Springer, Singapore. https://doi.org/10.1007/978-981-99-6917-3_10
Download citation
DOI: https://doi.org/10.1007/978-981-99-6917-3_10
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-99-6916-6
Online ISBN: 978-981-99-6917-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)