Abstract
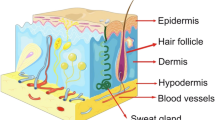
Stem cells are unspecialized cells potentially used to repair and restore lost or defective function of tissues, and are increasingly being recognized as a futuristic therapy. Their differentiation potential ranges from pluripotent to unipotent. Skin has a diversified pool of stem cells that regularly repair or regenerate skin. The pathophysiology of skin repair and regeneration has immensely increased the interest of clinicians and researchers in skin stem cells. Dermal mesenchymal stem cells have been recognized as the source for replenishing dermal fibroblasts. Skin injury response stimulates the epidermal stem cells and follicular stem cells for re-epithelization and healing. Melanocyte stem cells are crucial in maintaining skin and hair color, while sebaceous gland stem cells contribute towards maintaining the barrier function of the skin. Other than this, skin is also known as the homing site of hematopoietic stem cells, which essentially maintains the skin lymphocyte subtypes and contributes to skin immunity. The generation of induced pluripotent stem cells has created new hopes for patients with genetic diseases. Correcting the genetic variations that result in disease and restoration of the natural protein expression is an exciting part of stem cell research. Several promising clinical trials have been done to correct skin diseases using melanocytes, keratinocytes, and mesenchymal stem cells. Still, there is a need to understand the efficacy and safety of stem cell therapies based on racial or ethnic differences. At present, we lack long follow-up clinical trials for skin diseases. Integration with bioengineering has improved the working of stem cells by providing a tissue-specific base to grow. The use of such biomaterials has succeeded in develo** stem cell scaffolds. The biocompatibility of such materials however must be increased. The future of bioengineering is directed at the construction of various tissues including the skin, by using stem cells. Potential treatments are still some years away from clinical use, but results so far are promising.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Badiavas EV, Falanga V (2003) Treatment of chronic wounds with bone marrow-derived cells. Arch Dermatol 139(4):510–516. https://doi.org/10.1001/archderm.139.4.510
Barcelos LS, Duplaa C, Krankel N, Graiani G, Invernici G, Katare R, Siragusa M, Meloni M, Campesi I, Monica M, Simm A, Campagnolo P, Mangialardi G, Stevanato L, Alessandri G, Emanueli C, Madeddu P (2009) Human CD133+ progenitor cells promote the healing of diabetic ischemic ulcers by paracrine stimulation of angiogenesis and activation of Wnt signaling. Circ Res 104(9):1095–1102. https://doi.org/10.1161/CIRCRESAHA.108.192138
Bartsch G, Yoo JJ, De Coppi P, Siddiqui MM, Schuch G, Pohl HG, Fuhr J, Perin L, Soker S, Atala A (2005) Propagation, expansion, and multilineage differentiation of human somatic stem cells from dermal progenitors. Stem Cells Dev 14(3):337–348. https://doi.org/10.1089/scd.2005.14.337
Blanpain C, Fuchs E (2006) Epidermal stem cells of the skin. Annu Rev Cell Dev Biol 22:339–373. https://doi.org/10.1146/annurev.cellbio.22.010305.104357
Bruckner-Tuderman L (2019) Newer treatment modalities in epidermolysis bullosa. Indian Dermatol Online J 10(3):244–250. https://doi.org/10.4103/idoj.IDOJ_287_18
Burdon TJ, Paul A, Noiseux N, Prakash S, Shum-Tim D (2011) Bone marrow stem cell derived paracrine factors for regenerative medicine: current perspectives and therapeutic potential. Bone Marrow Res 2011:207326. https://doi.org/10.1155/2011/207326
Chu GY, Chen YF, Chen HY, Chan MH, Gau CS, Weng SM (2018) Stem cell therapy on skin: mechanisms, recent advances and drug reviewing issues. J Food Drug Anal 26(1):14–20. https://doi.org/10.1016/j.jfda.2017.10.004
Ciurea SO, Hansrivijit P, Ciurea AM, Hymes S, Chen J, Rondon G, Hosing C, Popat U, Champlin RE (2019) Curative potential of hematopoietic stem cell transplantation for advanced psoriasis. Am J Hematol 94(6):E176–E180. https://doi.org/10.1002/ajh.25470
ClinicalTrials.gov. https://clinicaltrials.gov/ct2/results?cond=Vitiligo & term=stem+cell & cntry= & state= & city= & dist=
Dash NR, Dash SN, Routray P, Mohapatra S, Mohapatra PC (2009) Targeting nonhealing ulcers of lower extremity in human through autologous bone marrow-derived mesenchymal stem cells. Rejuvenation Res 12(5):359–366. https://doi.org/10.1089/rej.2009.0872
Diez Villanueva P, Sanz-Ruiz R, Nunez Garcia A, Fernandez Santos ME, Sanchez PL, Fernandez-Aviles F (2012) Functional multipotency of stem cells: what do we need from them in the heart? Stem Cells Int 2012:817364. https://doi.org/10.1155/2012/817364
Egger A, Tomic-Canic M, Tosti A (2020) Advances in stem cell-based therapy for hair loss. CellR4 Repair Replace Regen Reprogram 8:e2894. https://www.ncbi.nlm.nih.gov/pubmed/32968692
Elmaadawi IH, Mohamed BM, Ibrahim ZAS, Abdou SM, El Attar YA, Youssef A, Shamloula MM, Taha A, Metwally HG, El Afandy MM, Salem ML (2018) Stem cell therapy as a novel therapeutic intervention for resistant cases of alopecia areata and androgenetic alopecia. J Dermatolog Treat 29(5):431–440. https://doi.org/10.1080/09546634.2016.1227419
Erratum (2015) Expert Opin Emerg Drugs 20(2):347. https://doi.org/10.1517/14728214.2015.1027860
Falanga V, Iwamoto S, Chartier M, Yufit T, Butmarc J, Kouttab N, Shrayer D, Carson P (2007) Autologous bone marrow-derived cultured mesenchymal stem cells delivered in a fibrin spray accelerate healing in murine and human cutaneous wounds. Tissue Eng 13(6):1299–1312. https://doi.org/10.1089/ten.2006.0278
Falto-Aizpurua L, Choudhary S, Tosti A (2014) Emerging treatments in alopecia. Expert Opin Emerg Drugs 19(4):545–556. https://doi.org/10.1517/14728214.2014.974550
Fernandes KJ, McKenzie IA, Mill P, Smith KM, Akhavan M, Barnabe-Heider F, Biernaskie J, Junek A, Kobayashi NR, Toma JG, Kaplan DR, Labosky PA, Rafuse V, Hui CC, Miller FD (2004) A dermal niche for multipotent adult skin-derived precursor cells. Nat Cell Biol 6(11):1082–1093. https://doi.org/10.1038/ncb1181
Fernandes KJ, Toma JG, Miller FD (2008) Multipotent skin-derived precursors: adult neural crest-related precursors with therapeutic potential. Philos Trans R Soc Lond Ser B Biol Sci 363(1489):185–198. https://doi.org/10.1098/rstb.2006.2020
Fu X, Sun X (2009) Can hematopoietic stem cells be an alternative source for skin regeneration? Ageing Res Rev 8(3):244–249. https://doi.org/10.1016/j.arr.2009.02.002
Fuchs E (2008) Skin stem cells: rising to the surface. J Cell Biol 180(2):273–284. https://doi.org/10.1083/jcb.200708185
Gauthier Y, Surleve-Bazeille JE (1992) Autologous grafting with noncultured melanocytes: a simplified method for treatment of depigmented lesions. J Am Acad Dermatol 26(2 Pt 1):191–194. https://doi.org/10.1016/0190-9622(92)70024-a
Gill BS, Brar MS, Chaudhary N, Randhawa A (2019) Non-cultured melanocyte transfer in the management of stable vitiligo. J Family Med Prim Care 8(9):2912–2916. https://doi.org/10.4103/jfmpc.jfmpc_546_19
Gurdon JB (1962) The developmental capacity of nuclei taken from intestinal epithelium cells of feeding tadpoles. J Embryol Exp Morphol 10:622–640. https://www.ncbi.nlm.nih.gov/pubmed/13951335
Hammersen J, Has C, Naumann-Bartsch N, Stachel D, Kiritsi D, Soder S, Tardieu M, Metzler M, Bruckner-Tuderman L, Schneider H (2016) Genotype, clinical course, and therapeutic decision making in 76 infants with severe generalized junctional epidermolysis bullosa. J Invest Dermatol 136(11):2150–2157. https://doi.org/10.1016/j.jid.2016.06.609
Hamza AM, Hussein TM, Shakshouk HAR (2019) Noncultured extracted hair follicle outer root sheath cell suspension versus noncultured epidermal cell suspension in the treatment of stable vitiligo. J Cutan Aesthet Surg 12(2):105–111. https://doi.org/10.4103/JCAS.JCAS_136_18
Harris ML, Buac K, Shakhova O, Hakami RM, Wegner M, Sommer L, Pavan WJ (2013) A dual role for SOX10 in the maintenance of the postnatal melanocyte lineage and the differentiation of melanocyte stem cell progenitors. PLoS Genet 9(7):e1003644. https://doi.org/10.1371/journal.pgen.1003644
Has C, Bauer JW, Bodemer C, Bolling MC, Bruckner-Tuderman L, Diem A, Fine JD, Heagerty A, Hovnanian A, Marinkovich MP, Martinez AE, McGrath JA, Moss C, Murrell DF, Palisson F, Schwieger-Briel A, Sprecher E, Tamai K, Uitto J, Woodley DT, Zambruno G, Mellerio JE (2020) Consensus reclassification of inherited epidermolysis bullosa and other disorders with skin fragility. Br J Dermatol 183(4):614–627. https://doi.org/10.1111/bjd.18921
Horsley V, O’Carroll D, Tooze R, Ohinata Y, Saitou M, Obukhanych T, Nussenzweig M, Tarakhovsky A, Fuchs E (2006) Blimp1 defines a progenitor population that governs cellular input to the sebaceous gland. Cell 126(3):597–609. https://doi.org/10.1016/j.cell.2006.06.048
Hsu YC, Li L, Fuchs E (2014) Emerging interactions between skin stem cells and their niches. Nat Med 20(8):847–856. https://doi.org/10.1038/nm.3643
Ito M, Liu Y, Yang Z, Nguyen J, Liang F, Morris RJ, Cotsarelis G (2005) Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat Med 11(12):1351–1354. https://doi.org/10.1038/nm1328
Jackow J, Titeux M, Portier S, Charbonnier S, Ganier C, Gaucher S, Hovnanian A (2016) Gene-corrected fibroblast therapy for recessive dystrophic epidermolysis bullosa using a self-inactivating COL7A1 retroviral vector. J Invest Dermatol 136(7):1346–1354. https://doi.org/10.1016/j.jid.2016.02.811
Jaks V, Barker N, Kasper M, van Es JH, Snippert HJ, Clevers H, Toftgard R (2008) Lgr5 marks cycling, yet long-lived, hair follicle stem cells. Nat Genet 40(11):1291–1299. https://doi.org/10.1038/ng.239
Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, Reyes M, Lenvik T, Lund T, Blackstad M, Du J, Aldrich S, Lisberg A, Low WC, Largaespada DA, Verfaillie CM (2002) Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418(6893):41–49. https://doi.org/10.1038/nature00870
Jones PH, Watt FM (1993) Separation of human epidermal stem cells from transit amplifying cells on the basis of differences in integrin function and expression. Cell 73(4):713–724. https://doi.org/10.1016/0092-8674(93)90251-k
Kim HS, Lee JH, Roh KH, Jun HJ, Kang KS, Kim TY (2017) Clinical trial of human umbilical cord blood-derived stem cells for the treatment of moderate-to-severe atopic dermatitis: phase I/IIa studies. Stem Cells 35(1):248–255. https://doi.org/10.1002/stem.2401
Kloepper JE, Tiede S, Brinckmann J, Reinhardt DP, Meyer W, Faessler R, Paus R (2008) Immunophenoty** of the human bulge region: the quest to define useful in situ markers for human epithelial hair follicle stem cells and their niche. Exp Dermatol 17(7):592–609. https://doi.org/10.1111/j.1600-0625.2008.00720.x
Lang D, Lu MM, Huang L, Engleka KA, Zhang M, Chu EY, Lipner S, Skoultchi A, Millar SE, Epstein JA (2005) Pax3 functions at a nodal point in melanocyte stem cell differentiation. Nature 433(7028):884–887. https://doi.org/10.1038/nature03292
Langton AK, Herrick SE, Headon DJ (2008) An extended epidermal response heals cutaneous wounds in the absence of a hair follicle stem cell contribution. J Invest Dermatol 128(5):1311–1318. https://doi.org/10.1038/sj.jid.5701178
Li B, Hu W, Ma K, Zhang C, Fu X (2019) Are hair follicle stem cells promising candidates for wound healing? Expert Opin Biol Ther 19(2):119–128. https://doi.org/10.1080/14712598.2019.1559290
Nishimura EK, Jordan SA, Oshima H, Yoshida H, Osawa M, Moriyama M, Jackson IJ, Barrandon Y, Miyachi Y, Nishikawa S (2002) Dominant role of the niche in melanocyte stem-cell fate determination. Nature 416(6883):854–860. https://doi.org/10.1038/416854a
Nowak JA, Polak L, Pasolli HA, Fuchs E (2008) Hair follicle stem cells are specified and function in early skin morphogenesis. Cell Stem Cell 3(1):33–43. https://doi.org/10.1016/j.stem.2008.05.009
Osawa M, Egawa G, Mak SS, Moriyama M, Freter R, Yonetani S, Beermann F, Nishikawa S (2005) Molecular characterization of melanocyte stem cells in their niche. Development 132(24):5589–5599. https://doi.org/10.1242/dev.02161
Ouji Y, Yoshikawa M, Moriya K, Nishiofuku M, Matsuda R, Ishizaka S (2008) Wnt-10b, uniquely among Wnts, promotes epithelial differentiation and shaft growth. Biochem Biophys Res Commun 367(2):299–304. https://doi.org/10.1016/j.bbrc.2007.12.091
Ozgul Ozdemir RB, Ozdemir AT, Kirmaz C, Ovali E, Olmez E, Kerem H, Evrenos MK, Deniz G (2020) Mesenchymal stem cells: a potential treatment approach for refractory chronic spontaneous urticaria. Stem Cell Rev Rep 17(3):911–922. https://doi.org/10.1007/s12015-020-10059-w
Pandya V, Parmar KS, Shah BJ, Bilimoria FE (2005) A study of autologous melanocyte transfer in treatment of stable vitiligo. Indian J Dermatol Venereol Leprol 71(6):393–397. https://doi.org/10.4103/0378-6323.18942
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284(5411):143–147. https://doi.org/10.1126/science.284.5411.143
Ramalho-Santos M, Willenbring H (2007) On the origin of the term “stem cell”. Cell Stem Cell 1(1):35–38. https://doi.org/10.1016/j.stem.2007.05.013
Rashidghamat E, McGrath JA (2017) Novel and emerging therapies in the treatment of recessive dystrophic epidermolysis bullosa. Intractable Rare Dis Res 6(1):6–20. https://doi.org/10.5582/irdr.2017.01005
Roncalli JG, Tongers J, Renault MA, Losordo DW (2008) Endothelial progenitor cells in regenerative medicine and cancer: a decade of research. Trends Biotechnol 26(5):276–283. https://doi.org/10.1016/j.tibtech.2008.01.005
Rosell A, Morancho A, Navarro-Sobrino M, Martinez-Saez E, Hernandez-Guillamon M, Lope-Piedrafita S, Barcelo V, Borras F, Penalba A, Garcia-Bonilla L, Montaner J (2013) Factors secreted by endothelial progenitor cells enhance neurorepair responses after cerebral ischemia in mice. PLoS One 8(9):e73244. https://doi.org/10.1371/journal.pone.0073244
Sackstein R (2004) The bone marrow is akin to skin: HCELL and the biology of hematopoietic stem cell homing. J Investig Dermatol Symp Proc 9(3):215–223. https://doi.org/10.1111/j.0022-202X.2004.09301.x
Schneider MR, Schmidt-Ullrich R, Paus R (2009) The hair follicle as a dynamic miniorgan. Curr Biol 19(3):R132–R142. https://doi.org/10.1016/j.cub.2008.12.005
Shpichka A, Butnaru D, Bezrukov EA, Sukhanov RB, Atala A, Burdukovskii V, Zhang Y, Timashev P (2019) Skin tissue regeneration for burn injury. Stem Cell Res Ther 10(1):94. https://doi.org/10.1186/s13287-019-1203-3
Stenn KS, Paus R (2001) Controls of hair follicle cycling. Physiol Rev 81(1):449–494. https://doi.org/10.1152/physrev.2001.81.1.449
Suh W, Kim KL, Kim JM, Shin IS, Lee YS, Lee JY, Jang HS, Lee JS, Byun J, Choi JH, Jeon ES, Kim DK (2005) Transplantation of endothelial progenitor cells accelerates dermal wound healing with increased recruitment of monocytes/macrophages and neovascularization. Stem Cells 23(10):1571–1578. https://doi.org/10.1634/stemcells.2004-0340
Sullivan KM, Goldmuntz EA, Keyes-Elstein L, McSweeney PA, Pinckney A, Welch B, Mayes MD, Nash RA, Crofford LJ, Eggleston B, Castina S, Griffith LM, Goldstein JS, Wallace D, Craciunescu O, Khanna D, Folz RJ, Goldin J, St Clair EW, Seibold JR, Phillips K, Mineishi S, Simms RW, Ballen K, Wener MH, Georges GE, Heimfeld S, Hosing C, Forman S, Kafaja S, Silver RM, Griffing L, Storek J, LeClercq S, Brasington R, Csuka ME, Bredeson C, Keever-Taylor C, Domsic RT, Kahaleh MB, Medsger T, Furst DE, Investigators SS (2018) Myeloablative autologous stem-cell transplantation for severe scleroderma. N Engl J Med 378(1):35–47. https://doi.org/10.1056/nejmoa1703327
Takahashi K, Yamanaka S (2013) Induced pluripotent stem cells in medicine and biology. Development 140(12):2457–2461. https://doi.org/10.1242/dev.092551
Taub AF, Pham K (2018) Stem cells in dermatology and anti-aging care of the skin. Facial Plast Surg Clin North Am 26(4):425–437. https://doi.org/10.1016/j.fsc.2018.06.004
Taylor G, Lehrer MS, Jensen PJ, Sun TT, Lavker RM (2000) Involvement of follicular stem cells in forming not only the follicle but also the epidermis. Cell 102(4):451–461. https://doi.org/10.1016/s0092-8674(00)00050-7
Thakur V, Kumar S, Kumaran MS, Kaushik H, Srivastava N, Parsad D (2019) Efficacy of transplantation of combination of noncultured dermal and epidermal cell suspension vs epidermal cell suspension alone in vitiligo: a randomized clinical trial. JAMA Dermatol 155(2):204–210. https://doi.org/10.1001/jamadermatol.2018.4919
Toma JG, McKenzie IA, Bagli D, Miller FD (2005) Isolation and characterization of multipotent skin-derived precursors from human skin. Stem Cells 23(6):727–737. https://doi.org/10.1634/stemcells.2004-0134
Uddin F, Rudin CM, Sen T (2020) CRISPR gene therapy: applications, limitations, and implications for the future. Front Oncol 10:1387. https://doi.org/10.3389/fonc.2020.01387
Vaculik C, Schuster C, Bauer W, Iram N, Pfisterer K, Kramer G, Reinisch A, Strunk D, Elbe-Burger A (2012) Human dermis harbors distinct mesenchymal stromal cell subsets. J Invest Dermatol 132(3 Pt 1):563–574. https://doi.org/10.1038/jid.2011.355
Venugopal SS, Yan W, Frew JW, Cohn HI, Rhodes LM, Tran K, Melbourne W, Nelson JA, Sturm M, Fogarty J, Marinkovich MP, Igawa S, Ishida-Yamamoto A, Murrell DF (2013) A phase II randomized vehicle-controlled trial of intradermal allogeneic fibroblasts for recessive dystrophic epidermolysis bullosa. J Am Acad Dermatol 69(6):898–908 e897. https://doi.org/10.1016/j.jaad.2013.08.014
Verfaillie CM, Pera MF, Lansdorp PM (2002) Stem cells: hype and reality. Hematology Am Soc Hematol Educ Program 369–391. https://doi.org/10.1182/asheducation-2002.1.369
Wagner JE, Ishida-Yamamoto A, McGrath JA, Hordinsky M, Keene DR, Woodley DT, Chen M, Riddle MJ, Osborn MJ, Lund T, Dolan M, Blazar BR, Tolar J (2010) Bone marrow transplantation for recessive dystrophic epidermolysis bullosa. N Engl J Med 363(7):629–639. https://doi.org/10.1056/NEJMoa0910501
Wettstein R, Savic M, Pierer G, Scheufler O, Haug M, Halter J, Gratwohl A, Baumberger M, Schaefer DJ, Kalbermatten DF (2014) Progenitor cell therapy for sacral pressure sore: a pilot study with a novel human chronic wound model. Stem Cell Res Ther 5(1):18. https://doi.org/10.1186/scrt407
Wong T, Gammon L, Liu L, Mellerio JE, Dop**-Hepenstal PJ, Pacy J, Elia G, Jeffery R, Leigh IM, Navsaria H, McGrath JA (2008) Potential of fibroblast cell therapy for recessive dystrophic epidermolysis bullosa. J Invest Dermatol 128(9):2179–2189. https://doi.org/10.1038/jid.2008.78
Yang R, Wang J, Chen X, Shi Y, **e J (2020) Epidermal stem cells in wound healing and regeneration. Stem Cells Int 2020:9148310. https://doi.org/10.1155/2020/9148310
Zakrzewski W, Dobrzynski M, Szymonowicz M, Rybak Z (2019) Stem cells: past, present, and future. Stem Cell Res Ther 10(1):68. https://doi.org/10.1186/s13287-019-1165-5
Zhang J, Chopp M (2013) Cell-based therapy for ischemic stroke. Expert Opin Biol Ther 13(9):1229–1240. https://doi.org/10.1517/14712598.2013.804507
Zhang CP, Fu XB (2008) Therapeutic potential of stem cells in skin repair and regeneration. Chin J Traumatol 11(4):209–221. https://doi.org/10.1016/s1008-1275(08)60045-0
Zokaei S, Farhud DD, Keykhaei M, Zarif Yeganeh M, Rahimi H, Moravvej H (2019) Cultured epidermal melanocyte transplantation in vitiligo: a review article. Iran J Public Health 48(3):388–399. https://www.ncbi.nlm.nih.gov/pubmed/31223565
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kumar, V., Handa, S. (2022). Stem Cells in Dermatology: What the Future May Hold. In: Sobti, R., Ganju, A.K. (eds) Biomedical Translational Research. Springer, Singapore. https://doi.org/10.1007/978-981-16-8845-4_2
Download citation
DOI: https://doi.org/10.1007/978-981-16-8845-4_2
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-8844-7
Online ISBN: 978-981-16-8845-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)