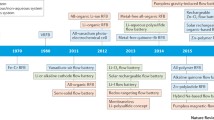
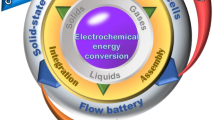
Abstract
This chapter presents a redox flow batteries review that has been investigated and developed over the past few decades. Redox flow batteries (RFBs) can be used as stationary energy storage systems from small to large scale. Flow batteries are interesting energy storage devices that can be designed flexibly due to the possibility of decoupling of power and energy. The design process allows a battery to evolve as the user needs change. Unfortunately, conventional batteries do not provide such a possibility. Therefore, flow batteries can be used as high energy and high power energy storage devices which could work together with grid-connected renewable energy sources (RES). The main aspects of this chapter correspond to the characterization of redox flow electrodes and electrolytes chemistries. Usually, carbon materials can be used to construct electrode materials of RFBs. Carbon materials are characterized by relatively high electrical conductivity, chemical resistance, and stability in a wide electrochemical window. This review provides a summary of current research and development of RFBs technology including cell design, electrode improvements, separators, and electrolytes.
Similar content being viewed by others
References
A. Abbas, S. Abbas, A. Bhattarai, et al., Effect of electrode porosity on the charge transfer in vanadium redox flow battery. J. Power Sources 488, 229411 (2021). https://doi.org/10.1016/j.jpowsour.2020.229411
P. Alotto, M. Guarnieri, F. Moro, Redox flow batteries for the storage of renewable energy: A review. Renew. Sust. Energ. Rev. 29, 325–335 (2014). https://doi.org/10.1016/j.rser.2013.08.001
K. Amini, M.D. Pritzker, Life-cycle analysis of zinc-cerium redox flow batteries. Electrochim. Acta 356, 1–14 (2020). https://doi.org/10.1016/j.electacta.2020.136785
J. Andrews, S. Seif Mohammadi, Towards a “proton flow battery”: Investigation of a reversible PEM fuel cell with integrated metal-hydride hydrogen storage. Int. J. Hydrog. Energy 39, 1740–1751 (2014). https://doi.org/10.1016/j.ijhydene.2013.11.010
R. Banerjee, N. Bevilacqua, A. Mohseninia, et al., Carbon felt electrodes for redox flow battery: Impact of compression on transport properties. J Energy Storage 26 (2019). https://doi.org/10.1016/j.est.2019.100997
A. Bhattarai, N. Wai, R. Schweiss, et al., Advanced porous electrodes with flow channels for vanadium redox flow battery. J. Power Sources 341, 83–90 (2017). https://doi.org/10.1016/j.jpowsour.2016.11.113
M. Burgess, J.S. Moore, J. Rodríguez-López, Redox active polymers as soluble nanomaterials for energy storage. Acc. Chem. Res. 49, 2649–2657 (2016). https://doi.org/10.1021/acs.accounts.6b00341
L. Cao, M. Skyllas-Kazacos, C. Menictas, J. Noack, A review of electrolyte additives and impurities in vanadium redox flow batteries. J Energy Chem 27, 1269–1291 (2018). https://doi.org/10.1016/j.jechem.2018.04.007
M.H. Chakrabarti, N.P. Brandon, S.A. Hajimolana, et al., Application of carbon materials in redox flow batteries. J. Power Sources 253, 150–166 (2014). https://doi.org/10.1016/j.jpowsour.2013.12.038
A. Clemente, R. Costa-Castelló, Redox flow batteries: A literature review oriented to automatic control. Energies 13, 1–31 (2020). https://doi.org/10.3390/en13174514
A. Cunha, J. Martins, N. Rodrigues, F.P. Brito, Vanadium redox flow batteries: A technology review. Int. J. Energy Res. 39, 889–918 (2015). https://doi.org/10.1002/er.3260
C. DeBruler, B. Hu, J. Moss, et al., Designer two-electron storage viologen anolyte materials for neutral aqueous organic redox flow batteries. Chem 3, 961–978 (2017). https://doi.org/10.1016/j.chempr.2017.11.001
Y. Ding, Y. Li, G. Yu, Exploring bio-inspired quinone-based organic redox flow batteries: A combined experimental and computational study. Chem 1, 790–801 (2016). https://doi.org/10.1016/j.chempr.2016.09.004
Y. Ding, C. Zhang, L. Zhang, et al., Molecular engineering of organic electroactive materials for redox flow batteries. Chem. Soc. Rev. 47, 69–103 (2018). https://doi.org/10.1039/c7cs00569e
A. Fetyan, G.A. El-Nagar, I. Lauermann, et al, Detrimental role of hydrogen evolution and its temperature-dependent impact on the performance of vanadium redox flow batteries. J. Energy Chem. 57–62. (2018). https://doi.org/10.1016/j.jechem.2018.06.010
C. Flox, J. Rubio-garcia, R. Nafria, et al., Active nano-CuPt 3 electrocatalyst supported on graphene for enhancing reactions at the cathode in all-vanadium redox flow batteries. Carbon N Y 50, 2372–2374 (2012). https://doi.org/10.1016/j.carbon.2012.01.060
M.R. Gerhardt, L. Tong, R. Gómez-Bombarelli, et al, Anthraquinone derivatives in aqueous flow batteries. Adv. Energy Mater. 7:1601488 (1–9). (2017). https://doi.org/10.1002/aenm.201601488
P.C. Ghimire, A. Bhattarai, R. Schweiss, et al., A comprehensive study of electrode compression effects in all vanadium redox flow batteries including locally resolved measurements. Appl. Energy 230, 974–982 (2018). https://doi.org/10.1016/j.apenergy.2018.09.049
A. Hazza, D. Pletcher, R. Wills, A novel flow battery – A lead acid battery based on an electrolyte with soluble lead (II) IV. The influence of additives. 149, 103–111 (2005). https://doi.org/10.1016/j.jpowsour.2005.01.049
H. He, S. Tian, B. Tarroja, et al., Flow battery production: Materials selection and environmental impact. J. Clean. Prod. 269, 121740 (2020). https://doi.org/10.1016/j.jclepro.2020.121740
A. Hollas, X. Wei, V. Murugesan, et al., A biomimetic high-capacity phenazine-based anolyte for aqueous organic redox flow batteries. Nat. Energy 3, 508–514 (2018). https://doi.org/10.1038/s41560-018-0167-3
B. Hu, C. DeBruler, Z. Rhodes, T.L. Liu, Long-cycling aqueous organic redox flow battery (AORFB) toward sustainable and safe energy storage. J. Am. Chem. Soc. 139, 1207–1214 (2017). https://doi.org/10.1021/jacs.6b10984
B. Huskinson, M.P. Marshak, M.R. Gerhardt, M.J. Aziz, Cycling of a quinone-bromide flow battery for large-scale electrochemical energy storage. ECS Trans. 61, 27–30 (2014a). https://doi.org/10.1149/06137.0027ecst
B. Huskinson, M.P. Marshak, C. Suh, et al., A metal-free organic-inorganic aqueous flow battery. Nature 505, 195–198 (2014b). https://doi.org/10.1038/nature12909
H.R. Jiang, J. Sun, L. Wei, et al., A high power density and long cycle life vanadium redox flow battery. Energy Storage Mater 24, 529–540 (2020). https://doi.org/10.1016/j.ensm.2019.07.005
D.G. Kwabi, Y. Ji, M.J. Aziz, Electrolyte lifetime in aqueous organic redox flow batteries: A critical review. Chem. Rev. 120, 6467–6489 (2020). https://doi.org/10.1021/acs.chemrev.9b00599
D.G. Kwabi, K. Lin, Y. Ji, et al., Alkaline quinone flow battery with long lifetime at pH 12. Joule 2, 1894–1906 (2018). https://doi.org/10.1016/j.joule.2018.07.005
P. Leung, X. Li, C. Ponce De León, et al., Progress in redox flow batteries, remaining challenges and their applications in energy storage. RSC Adv. 2, 10125–10156 (2012). https://doi.org/10.1039/c2ra21342g
P.K. Leung, C. Ponce-De-León, C.T.J. Low, et al., Characterization of a zinc-cerium flow battery. J. Power Sources 196, 5174–5185 (2011). https://doi.org/10.1016/j.jpowsour.2011.01.095
B. Li, Z. Nie, M. Vijayakumar, et al., Ambipolar zinc-polyiodide electrolyte for a high-energy density aqueous redox flow battery. Nat. Commun. 6, 1–8 (2015). https://doi.org/10.1038/ncomms7303
W. Li, J. Liu, C. Yan, Multi-walled carbon nanotubes used as an electrode reaction catalyst for VO 2 + / VO 2 + for a vanadium redox flow battery. Carbon N Y 49, 3463–3470 (2011a). https://doi.org/10.1016/j.carbon.2011.04.045
Y. Li, J. Parrondo, S. Sankarasubramanian, V. Ramani, Impact of surface carbonyl- and hydroxyl-group concentrations on electrode kinetics in an all-vanadium redox flow battery 2 (2019). https://doi.org/10.1021/acs.jpcc.8b11874
Z. Li, S. Li, S. Liu, et al., Electrochemical properties of an all-organic redox flow battery using 2,2,6,6-tetramethyl-1-piperidinyloxy and N-Methylphthalimide. Electrochem. Solid-State Lett. 14, A171–A173 (2011b). https://doi.org/10.1149/2.012112esl
Z. Li, M.S. Pan, L. Su, et al., Air-breathing aqueous sulfur flow battery for ultralow-cost long-duration electrical storage. Joule 1, 306–327 (2017). https://doi.org/10.1016/j.joule.2017.08.007
K. Lin, Q. Chen, M.R. Gerhardt, et al., Alkaline quinone flow battery. Science (80-) 349, 1529–1532 (2015). https://doi.org/10.1126/science.aab3033
K. Lin, R. Gómez-Bombarelli, E.S. Beh, et al., A redox-flow battery with an alloxazine-based organic electrolyte. Nat. Energy 1, 1–8 (2016). https://doi.org/10.1038/nenergy.2016.102
T. Liu, X. Li, C. Xu, H. Zhang, Activated carbon fiber paper based electrodes with high electrocatalytic activity for vanadium flow batteries with improved power density activated carbon fiber paper based electrodes with high electrocatalytic activity for vanadium flow batteries with im. ACS Appl. Mater. Interfaces 9, 4626–4633 (2017). https://doi.org/10.1021/acsami.6b14478
T. Liu, X. Wei, Z. Nie, et al., A total organic aqueous redox flow battery employing a low cost and sustainable methyl viologen anolyte and 4-HO-TEMPO catholyte. Adv. Energy Mater. 6(1–8), 1501449 (2016). https://doi.org/10.1002/aenm.201501449
D. Lloyd, E. Magdalena, L. Sanz, et al., Preparation of a cost-effective, scalable and energy efficient all-copper redox flow battery. J. Power Sources 292, 87–94 (2015). https://doi.org/10.1016/j.jpowsour.2015.04.176
D. Lloyd, T. Vainikka, K. Kontturi, The development of an all copper hybrid redox flow battery using deep eutectic solvents. Electrochim. Acta 100, 18–23 (2013). https://doi.org/10.1016/j.electacta.2013.03.130
J.D. Milshtein, J.L. Barton, R.M. Darling, F.R. Brushett, 4-Acetamido-2,2,6,6-tetramethylpiperidine-1-oxyl as a model organic redox active compound for nonaqueous flow batteries. J. Power Sources 327, 151–159 (2016). https://doi.org/10.1016/j.jpowsour.2016.06.125
E.C. Montoto, G. Nagarjuna, J.S. Moore, J. Rodríguez-López, Redox active for non-aqueous redox flow batteries: Validation of the size-exclusion approach. J. Electrochem. Soc. 164, A1688–A1694 (2017). https://doi.org/10.1149/2.1511707jes
G. Nagarjuna, J. Hui, K.J. Cheng, et al., Impact of redox-active polymer molecular weight on the electrochemical properties and transport across porous separators in nonaqueous solvents. J. Am. Chem. Soc. 136, 16309–16316 (2014). https://doi.org/10.1021/ja508482e
J. Noack, L. Wietschel, N. Roznyatovskaya, et al., Techno-economic modeling and analysis of redox flow battery systems. Energies 9 (2016). https://doi.org/10.3390/en9080627
D.O. Opar, R. Nankya, C.J. Raj, H. Jung, In-situ functionalization of binder-free three-dimensional boron-doped mesoporous graphene electrocatalyst as a high-performance electrode material for all-vanadium redox flow batteries. Appl. Mater. Today 22 (2021). https://doi.org/10.1016/j.apmt.2021.100950
F. Pan, Q. Wang, Redox species of redox flow batteries: A review. Molecules 20, 20499–20517 (2015). https://doi.org/10.3390/molecules201119711
A. Parasuraman, T.M. Lim, C. Menictas, M. Skyllas-Kazacos, Review of material research and development for vanadium redox flow battery applications. Electrochim. Acta 101, 27–40 (2013). https://doi.org/10.1016/j.electacta.2012.09.067
S.E. Park, S.Y. Yang, K.J. Kim, Boron-functionalized carbon felt electrode for enhancing the electrochemical performance of vanadium redox flow batteries. Appl. Surf. Sci. 546, 148941 (2021). https://doi.org/10.1016/j.apsusc.2021.148941
J. Piwek, C.R. Dennison, E. Frackowiak, et al., Vanadium-oxygen cell for positive electrolyte discharge in dual-circuit vanadium redox flow battery. J. Power Sources 439, 227075 (2019). https://doi.org/10.1016/j.jpowsour.2019.227075
C. Ponce de León, A. Frías-Ferrer, J. González-García, et al., Redox flow cells for energy conversion. J. Power Sources 160, 716–732 (2006). https://doi.org/10.1016/j.jpowsour.2006.02.095
G.J.W. Radford, J. Cox, R.G.A. Wills, F.C. Walsh, Electrochemical characterisation of activated carbon particles used in redox flow battery electrodes. 185, 1499–1504 (2008). https://doi.org/10.1016/j.jpowsour.2008.08.020
F. Rahman, M. Skyllas-Kazacos, Solubility of vanadyl sulfate in concentrated sulfuric acid solutions. J. Power Sources 72, 105–110 (1998). https://doi.org/10.1016/S0378-7753(97)02692-X
G.W. Reade, F.C. Walsh, Reticulated vitreous carbon as an electrode material n. 561, 203–217 (2004). https://doi.org/10.1016/j.jelechem.2003.07.019
D. Reynard, H. Vrubel, C.R. Dennison, et al., On-site purification of copper-contaminated vanadium electrolytes by using a vanadium redox flow battery. ChemSusChem 12, 1222–1228 (2019). https://doi.org/10.1002/cssc.201802895
N. Roznyatovskaya, J. Noack, H. Mild, et al., Vanadium electrolyte for all-vanadium redox-flow batteries: The effect of the counter ion. Batteries 5, 1–16 (2019). https://doi.org/10.3390/batteries5010013
E. Ruban, A. Stepashkin, N. Gvozdik, et al., Carbonized elastomer composite filled with hybrid carbon fillers for vanadium redox flow battery bipolar plates. Mater Today Commun 26, 101967 (2021). https://doi.org/10.1016/j.mtcomm.2020.101967
L. Sanz, D. Lloyd, E. Magdalena, et al., Study and characterization of positive electrolytes for application in the aqueous all-copper redox flow battery. J. Power Sources 278, 175–182 (2015). https://doi.org/10.1016/j.jpowsour.2014.12.034
C.S. Sevov, S.L. Fisher, L.T. Thompson, M.S. Sanford, Mechanism-based development of a low-potential, soluble, and cyclable multielectron anolyte for nonaqueous redox flow batteries. J. Am. Chem. Soc. 138, 15378–15384 (2016). https://doi.org/10.1021/jacs.6b07638
M. Skyllas-Kazacos, L. Cao, M. Kazacos, et al., Vanadium electrolyte studies for the vanadium redox battery—A review. ChemSusChem 9, 1521–1543 (2016). https://doi.org/10.1002/cssc.201600102
C.Y. Sun, H. Zhang, Investigation of Nafion series membranes on the performance of iron-chromium redox flow battery. Int. J. Energy Res. 43, 8739–8752 (2019). https://doi.org/10.1002/er.4875
K. Takechi, Y. Kato, Y. Hase, A highly concentrated catholyte based on a solvate ionic liquid for rechargeable flow batteries. Adv. Mater. 27, 2501–2506 (2015). https://doi.org/10.1002/adma.201405840
X. Teng, J. Dai, J. Su, G. Yin, Modification of Nafion membrane using fluorocarbon surfactant for all vanadium redox flow battery. J Memb Sci 476, 20–29 (2015). https://doi.org/10.1016/j.memsci.2014.11.014
X. Teng, C. Sun, J. Dai, et al., Solution casting Nafion/polytetrafluoroethylene membrane for vanadium redox flow battery application. Electrochim. Acta 88, 725–734 (2013). https://doi.org/10.1016/j.electacta.2012.10.093
H.M. Tsai, S.J. Yang, C.C.M. Ma, X. **e, Preparation and electrochemical activities of iridium-decorated graphene as the electrode for all-vanadium redox flow batteries. Electrochim. Acta 77, 232–236 (2012). https://doi.org/10.1016/j.electacta.2012.05.099
M.C. Tucker, A. Phillips, A.Z. Weber, All-iron redox flow battery tailored for off-grid portable applications. ChemSusChem 8, 3996–4004 (2015). https://doi.org/10.1002/cssc.201500845
W.H. Wang, X.D. Wang, Investigation of Ir-modified carbon felt as the positive electrode of an all-vanadium redox flow battery. Electrochim. Acta 52, 6755–6762 (2007). https://doi.org/10.1016/j.electacta.2007.04.121
A.Z. Weber, M.M. Mench, J.P. Meyers, et al., Redox flow batteries: A review. J. Appl. Electrochem. 41, 1137–1164 (2011). https://doi.org/10.1007/s10800-011-0348-2
L. Wei, T.S. Zhao, Q. Xu, et al., In-situ investigation of hydrogen evolution behavior in vanadium redox flow batteries. Appl. Energy 190, 1112–1118 (2017). https://doi.org/10.1016/j.apenergy.2017.01.039
R.G.A. Wills, J. Collins, D. Stratton-Campbell, et al., Developments in the soluble lead-acid flow battery. J. Appl. Electrochem. 40, 955–965 (2010). https://doi.org/10.1007/s10800-009-9815-4
Z. Yang, J. Zhang, M.C.W. Kintner-Meyer, et al., Electrochemical energy storage for green grid. Chem. Rev. 111, 3577–3613 (2011). https://doi.org/10.1021/cr100290v
X.Z. Yuan, C. Song, A. Platt, et al., A review of all-vanadium redox flow battery durability: Degradation mechanisms and mitigation strategies. Int. J. Energy Res. 43, 6599–6638 (2019). https://doi.org/10.1002/er.4607
Y. Zhai, Y. Dou, D. Zhao, et al., Carbon materials for chemical capacitive energy storage. Adv. Mater. 23, 4828–4850 (2011). https://doi.org/10.1002/adma.201100984
F. Zhong, M. Yang, M. Ding, C. Jia, Organic electroactive molecule-based electrolytes for redox flow batteries: Status and challenges of molecular design. Front. Chem. 8(1–14), 451 (2020). https://doi.org/10.3389/fchem.2020.00451
H. Zhou, H. Zhang, P. Zhao, B. Yi, A comparative study of carbon felt and activated carbon based electrodes for sodium polysulfide / bromine redox flow battery 51, 6304–6312 (2006). https://doi.org/10.1016/j.electacta.2006.03.106
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Lota, G., Graś-Ligocka, M., Kolanowski, Ł., Lota, K. (2022). Flow Batteries: Recent Advancement and Challenges. In: Gupta, R. (eds) Handbook of Energy Materials. Springer, Singapore. https://doi.org/10.1007/978-981-16-4480-1_50-1
Download citation
DOI: https://doi.org/10.1007/978-981-16-4480-1_50-1
Received:
Accepted:
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-4480-1
Online ISBN: 978-981-16-4480-1
eBook Packages: Springer Reference Chemistry and Mat. ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics