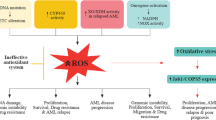
Abstract
Leukemia disorders have showed resistance to conventional treatment. Therefore, the idea of treating them with drugs that either directly or indirectly generates oxidative stress (OS) and hydrogen peroxide (H2O2) as effective therapeutic strategy for selectively pushing those cells over reactive oxygen species (ROS) threshold and induce apoptosis has ultimately gained scientific momentum. Our research group has focused on unrevealing the molecular mechanism of OS/H2O2 -induced apoptosis in leukemia cells by metabolic alterations (e.g., glucose starvation, fructose as only energetic source) and some selected natural or chemical agents including vitamin C, vitamin K3, vitamin E D-α-tocopheryl polyethylene glycol succinate, snake venom metalloproteinase nasulysin-1, and l-amino oxidase MipLAAO, avocado extracts, mitochondrial complex I inhibitor rotenone, metal chelator TPEN, antibiotic doxorubicin, and minocycline. Original published work by the authors and the PubMed database was consulted to compile this review. Therefore, this review aims to provide hematologists and physicians with an overview of results from in vitro models illustrating the complex interactions between metabolisms, natural and chemical molecules, OS, and apoptosis as therapeutic strategy against leukemia.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- ABL1 :
-
Abelson murine leukemia viral oncogene homolog 1
- ALL :
-
acute lymphoblastic leukemia
- AML :
-
acute myelogenous leukemia
- BCR :
-
breakpoint cluster region
- BAX :
-
Bcl-2-associated X protein
- BAD :
-
Bcl-2-associated death promoter
- BID :
-
BH3-interacting domain death agonist
- BOK :
-
Bcl-2-related ovarian killer
- BIK :
-
Bcl-2-interacting killer
- BAK :
-
Bcl-2 homologous antagonist killer
- CLL :
-
chronic lymphoblastic leukemia
- CML :
-
chronic myelogenous leukemia
- DOX :
-
doxorubicin
- GS :
-
glucose starvation
- H 2 O 2 :
-
hydrogen peroxide
- MipLAAO :
-
Microrus mipartitus l-amino oxidase
- MC :
-
minocyclin
- OS :
-
oxidative stress
- PUMA :
-
p53 upregulated modulator of apoptosis
- ROS :
-
reactive oxygen species
- ROT :
-
rotenone
- RCD :
-
regulated cell death
- TPEN :
-
N,N,N′,N′-tetrakis(2-pyridinylmethyl)-1,2-ethanediamine
- TPGS :
-
D-α-tocopheryl polyethylene glycol succinate
- VC :
-
vitamin C
- VK3 :
-
vitamin K3
References
Bedoya-Medina J, Mendivil-Perez M, Rey-Suarez P, Jimenez-Del-Rio M, Núñez V, Velez-Pardo C (2019) L-amino acid oxidase isolated from Micrurus mipartitus snake venom (MipLAAO) specifically induces apoptosis in acute lymphoblastic leukemia cells mostly via oxidative stress-dependent signaling mechanism. Int J Biol Macromol 134:1052–1062
Birkinshaw RW, Gong JN, Luo CS, Lio D, White CA, Anderson MA, Blombery P, Lessene G, Majewski IJ, Thijssen R, Roberts AW, Huang DCS, Colman PM, Czabotar PE (2019) Structures of BCL-2 in complex with venetoclax reveal the molecular basis of resistance mutations. Nat Commun 10:2385
Blaszczak W, Barczak W, Masternak J, Kopczyński P, Zhitkovich A, Rubiś B (2019) Vitamin C as a modulator of the response to cancer therapy. Molecules 24(3)
Bonilla-Porras AR, Jimenez-Del-Rio M, Velez-Pardo C (2011) Vitamin K3 and vitamin C alone or in combination induced apoptosis in leukemia cells by a similar oxidative stress signalling mechanism. Cancer Cell Int 11:19
Bonilla-Porras AR, Salazar-Ospina A, Jimenez-Del-Rio M, Pereañez-Jimenez A, Velez-Pardo C (2014) Pro-apoptotic effect of Persea americana var. Hass (avocado) on Jurkat lymphoblastic leukemia cells. Pharm Biol 52:458–465
Bonilla-Porras AR, Vargas LJ, Jimenez-Del-Rio M, Nuñez V, Velez-Pardo C (2016) Purification of nasulysin-1: a new toxin from Porthidium nasutum snake venom that specifically induces apoptosis in leukemia cell model through caspase-3 and apoptosis-inducing factor activation. Toxicon 120:166–174
Bonilla-Porras AR, Arevalo-Arbelaez A, Alzate-Restrepo JF, Velez-Pardo C, Jimenez-Del-Rio M (2018) PARKIN overexpression in human mesenchymal stromal cells from Wharton’s jelly suppresses 6-hydroxydopamine-induced apoptosis: potential therapeutic strategy in Parkinson’s disease. Cytotherapy 20:45–61
Chandrasekhar C, Kumar PS, Sarma PVGK (2019) Novel mutations in the kinase domain of BCR-ABL gene causing imatinib resistance in chronic myeloid leukemia patients. Sci Rep 9:2412
Chio IIC, Tuveson DA (2017) ROS in cancer: the burning question. Trends Mol Med 23(5):411–429
Cross N (2017) BCR-ABL Negative CML-Like Disorder. Clin Lymphoma Myeloma Leuk 17:S107–S108
Di Marzo N, Chisci E, Giovannoni R (2018) The role of hydrogen peroxide in redox-dependent Signaling: homeostatic and pathological responses in mammalian cells. Cells 7(10). pii: E156
Diaz-Aguirre V, Velez-Pardo C, Jimenez-Del-Rio M (2016) Fructose sensitizes Jurkat cells oxidative stress-induced apoptosis via caspase-dependent and caspase-independent mechanisms. Cell Biol Int 40:1162–1173
Dieck CL, Ferrando A (2019) Genetics and mechanisms of NT5C2-driven chemotherapy resistance in relapsed ALL. Blood 133:2263–2268
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM, Lydon NB, Kantarjian H, Capdeville R, Ohno-Jones S, Sawyers CL (2001) Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 344:1031–1037
Foster MN, Carr AC, Antony A, Peng S, Fitzpatrick MG (2018) Intravenous vitamin C administration improved blood cell counts and health-related quality of life of patient with history of relapsed acute myeloid leukaemia. Antioxidants (Basel) 7. pii: E92
Galadari S, Rahman A, Pallichankandy S, Thayyullathil F (2017) Reactive oxygen species and cancer paradox: to promote or to suppress? Free Radic Biol Med 104:144–164
Galluzzi L, Vitale I, Abrams JM, Alnemri ES, Baehrecke EH, Blagosklonny MV, Dawson TM, Dawson VL, El-Deiry WS, Fulda S, Gottlieb E, Green DR, Hengartner MO, Kepp O, Knight RA, Kumar S, Lipton SA, Lu X, Madeo F, Malorni W, Mehlen P, Nuñez G, Peter ME, Piacentini M, Rubinsztein DC, Shi Y, Simon HU, Vandenabeele P, White E, Yuan J, Zhivotovsky B, Melino G, Kroemer G (2012) Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ 19:107–120
Galluzzi L, Vitale I, Aaronson SA, 165 authors (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death. Cell Death Differ 25:486–541
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Harris IS, Treloar AE, Inoue S, Sasaki M, Gorrini C, Lee KC, Yung KY, Brenner D, Knobbe-Thomsen CB, Cox MA, Elia A, Berger T, Cescon DW, Adeoye A, Brüstle A, Molyneux SD, Mason JM, Li WY, Yamamoto K, Wakeham A, Berman HK, Khokha R, Done SJ, Kavanagh TJ, Lam CW, Mak TW (2015) Glutathione and thioredoxin antioxidant pathways synergize to drive cancer initiation and progression. Cancer Cell 27:211–222
Heisterkamp N, Stephenson JR, Groffen J, Hansen PF, de Klein A, Bartram CR, Grosveld G (1983) Localization of the c-abl oncogene adjacent to a translocation break point in chronic myelocytic leukaemia. Nature 306(5940):239–242
Kampen KR (2012) The discovery and early understanding of leukemia. Leuk Res 36:6–13
Kinumi T, Kimata J, Taira T, Ariga H, Niki E (2004) Cysteine-106 of DJ-1 is the most sensitive cysteine residue to hydrogen peroxide-mediated oxidation in vivo in human umbilical vein endothelial cells. Biochem Biophys Res Commun 317:722–728
Kitada T, Hikita R, Hirose H (2016) Parkin, Parkinson disease gene product, directly reduces hydrogen peroxide (mitochondrial oxidant), and forms dimerization reversibly. Int J Latest res Sci Technol 5:1–3
Kroemer G, Galluzzi L, Vandenabeele P, Abrams J, Alnemri ES, Baehrecke EH, Blagosklonny MV, El-Deiry WS, Golstein P, Green DR, Hengartner M, Knight RA, Kumar S, Lipton SA, Malorni W, Nuñez G, Peter ME, Tschopp J, Yuan J, Piacentini M, Zhivotovsky B, Melino G; Nomenclature Committee on Cell Death 2009 (2009) Classification of cell death: recommendations of the nomenclature committee on cell death 2009. Cell Death Differ 16:3–11
Lin X, Qureshi MZ, Attar R, Khalid S, Tahir F, Yaqub A, Aslam A, Yaylim I, De Carlos Back LK, Farooqi AA, Ismail M (2016) Targeting of BCR-ABL: lessons learned from BCR-ABL inhibition. Cell Mol Biol (Noisy-le-Grand) 62:129–137
Mansoori B, Mohammadi A, Davudian S, Shirjang S, Baradaran B (2017) The different mechanisms of Cancer drug resistance: a brief review. Adv Pharm Bull 7:339–348
Marinho HS, Real C, Cyrne L, Soares H, Antunes F (2014) Hydrogen peroxide sensing, signaling and regulation of transcription factors. Redox Biol 2:535–562
McBride A, Houtmann S, Wilde L, Vigil C, Eischen CM, Kasner M, Palmisiano N (2019) The role of inhibition of apoptosis in acute Leukemias and myelodysplastic syndrome. Front Oncol 9:192
Mendivil-Perez M, Velez-Pardo C, Jimenez-Del-Rio M (2012) TPEN induces apoptosis independently of zinc chelator activity in a model of acute lymphoblastic leukemia and ex vivo acute leukemia cells through oxidative stress and mitochondria caspase-3- and AIF-dependent pathways. Oxidative Med Cell Longev 2012:313275
Mendivil-Perez M, Jimenez-Del-Rio M, Velez-Pardo C (2013) Glucose starvation induces apoptosis in a model of acute T leukemia dependent on caspase-3 and apoptosis-inducing factor: a therapeutic strategy. Nutr Cancer 65:99–109
Mendivil-Perez M, Jimenez-Del-Rio M, Velez-Pardo C (2014) Response to rotenone is glucose-sensitive in a model of human acute lymphoblastic leukemia: involvement of oxidative stress mechanism, DJ-1, Parkin, and PINK-1 proteins. Oxidative Med Cell Longev 2014:457154
Mendivil-Perez M, Velez-Pardo C, Jimenez-Del-Rio M (2015) Doxorubicin induces apoptosis in Jurkat cells by mitochondria-dependent and mitochondria-independent mechanisms under normoxic and hypoxic conditions. Anti-Cancer Drugs 26:583–598
Mukherjee S (2010) The emperor of all maladies: a biography of cancer. Scribner, New York; Toronto
Nowell PC (2007) Discovery of the Philadelphia chromosome: a personal perspective. J Clin Invest 117:2033–2035
Oh SE, Maral Mouradian M (2018) Cytoprotective mechanisms of DJ-1 against oxidative stress through modulating ERK1/2 and ASK1 signal transduction. Redox Biol 14:211–217
Poole LB (2015) The basics of thiols and cysteines in redox biology and chemistry. Free Radic Biol Med 80:148–157
Prokocimer M, Molchadsky A, Rotter V (2017) Dysfunctional diversity of p53 proteins in adult acute myeloid leukemia: projections on diagnostic workup and therapy. Blood 130:699–712
Reczek CR, Chandel NS (2017) The two faces of reactive oxygen species in cancer. Annu Rev Cancer Biol 1:4.1–4.20
Rojas-Valencia L, Velez-Pardo C, Jimenez-Del-Rio M (2017) Metal chelator TPEN selectively induces apoptosis in K562 cells through reactive oxygen species signaling mechanism: implications for chronic myeloid leukemia. Biometals 30:405–421
Ruiz-Moreno C, Jimenez-Del-Rio M, Sierra-Garcia L, Lopez-Osorio B, Velez-Pardo C (2016) Vitamin E synthetic derivate-TPGS-selectively induces apoptosis in jurkat t cells via oxidative stress signaling pathways: implications for acute lymphoblastic leukemia. Apoptosis 21:1019–1032
Ruiz-Moreno C, Velez-Pardo C, Jimenez-Del-Rio M (2018) Minocycline induces apoptosis in acute lymphoblastic leukemia Jurkat cells. Toxicol In Vitro 50:336–346
Samimi A, Kalantari H, Lorestani MZ, Shirzad R, Saki N (2018) Oxidative stress in normal hematopoietic stem cells and leukemia. APMIS 126:284–294
Sies H (2015) Oxidative stress: a concept in redox biology and medicine. Redox Biol 4:180–183
Sies H (2018) On the history of oxidative stress: concept and some aspects of current development. Curr Opinion Toxicol 7:122–126
Sung KW, Choi J, Hwang YK, Lee SJ, Kim HJ, Kim JY, Cho EJ, Yoo KH, Koo HH (2009) Overexpression of X-linked inhibitor of apoptosis protein (XIAP) is an independent unfavorable prognostic factor in childhood de novo acute myeloid leukemia. J Korean Med Sci 24:605–613
Tasian SK, Loh ML, Hunger SP (2017) Philadelphia chromosome-like acute lymphoblastic leukemia. Blood 130:2064–2072
Wapner J (2013) The Philadelphia chromosome: a genetic mystery, a lethal cancer, and the improbable invention of a lifesaving treatment. The Experiment, New York
Zhang J, Lei W, Chen X, Wang S, Qian W (2018) Oxidative stress response induced by chemotherapy in leukemia treatment. Mol Clin Oncol 8:391–399
Zou Z, Chang H, Li H, Wang S (2017) Induction of reactive oxygen species: an emerging approach for cancer therapy. Apoptosis 22:1321–1335
Acknowledgements
This work was supported by the University of Antioquia, UdeA, and “Fundación Alfonso Moreno Jaramillo” grants #2017-16748 and #2018-20454 to CV-P and MJ-Del-R. The funders had no role, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Velez-Pardo, C., Jimenez-Del-Rio, M. (2022). Hel** Leukemia Cells to Die with Natural or Chemical Compounds Through H2O2 Signaling. In: Chakraborti, S., Ray, B.K., Roychoudhury, S. (eds) Handbook of Oxidative Stress in Cancer: Mechanistic Aspects. Springer, Singapore. https://doi.org/10.1007/978-981-15-9411-3_45
Download citation
DOI: https://doi.org/10.1007/978-981-15-9411-3_45
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-9410-6
Online ISBN: 978-981-15-9411-3
eBook Packages: Biomedical and Life SciencesReference Module Biomedical and Life Sciences