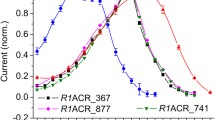
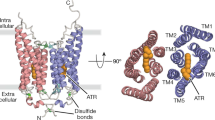
Abstract
Cl−-pump rhodopsin is the second discovered microbial rhodopsin. Although its physiological role has not been fully clarified, its functional mechanism has been studied as a model for anion transporters. After the success of neural activation by channel rhodopsin, the first Cl−-pump halorhodopsin (HR) had become widely used as a neural silencer. The emergence of artificial and natural anion channel rhodopsins lowered the importance of HRs. However, the longer absorption maxima of approximately 585–600 nm for HRs are still advantageous for applications in mammalian brains and collaborations with neural activators possessing shorter absorption maxima. In this chapter, the variation and functional mechanisms of Cl− pumps are summarized. After the discovery of HR, Cl−-pump rhodopsins were confined to only extremely halophilic haloarchaea. However, after 2014, two Cl−-pump groups were newly discovered in marine and terrestrial bacteria. These Cl− pumps are phylogenetically distinct from HRs and have unique characteristics. In particular, the most recently identified Cl− pump has close similarity with the H+ pump bacteriorhodopsin and was converted into the H+ pump by a single amino acid replacement.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- BR:
-
Bacteriorhodopsin
- Brub:
-
Bacterioruberin
- CP:
-
Cytoplasmic
- DDM:
-
n-dodecyl-β-d-maltoside
- EC:
-
Extracellular
- FR:
-
Cl− pump from Fulvimarina pelagi
- HR:
-
Halorhodopsin
- HsHR:
-
HR from Halobacterium salinarum
- Htr:
-
Halobacterial transducer
- MrHR:
-
Cl− pump from Mastigocladopsis repens
- NaR:
-
Na+-pump rhodopsin
- NM-R3:
-
Cl− pump from Nonlabens marinus S1-08T
- NpHR:
-
HR from Natronomonas pharaonis
- PRC:
-
H+-release complex
- PSB:
-
Protonated Schiff base
- SyHR:
-
Cl− pump from Synechocystis sp. PCC 7509
References
Balashov SP (2000) Protonation reactions and their coupling in bacteriorhodopsin. Biochim Biophys Acta 1460(1):75–94
Balashov SP, Govindjee R, Kono M, Imasheva E, Lukashev E, Ebrey TG, Crouch RK, Menick DR, Feng Y (1993) Effect of the arginine-82 to alanine mutation in bacteriorhodopsin on dark adaptation, proton release, and the photochemical cycle. Biochemistry 32(39):10331–10343
Bamberg E, Hegemann P, Oesteriielt D (1984) Reconstitution of the light-driven electrogenic ion pump halorhodopsin in black lipid membranes. Biochim Biophys Acta 773(1):53–60
Bieszke JA, Braun EL, Bean LE, Kang S, Natvig DO, Borkovich KA (1999) The nop-1 gene of Neurospora crassa encodes a seven transmembrane helix retinal-binding protein homologous to archaeal rhodopsins. Proc Natl Acad Sci U S A 96(14):8034
Blatz PE, Mohler JH, Navangul HV (1972) Anion-induced wavelength regulation of absorption maxima of Schiff bases of retinal. Biochemistry 11(5):848–855. https://doi.org/10.1021/bi00755a026
Chen XR, Huang YC, Yi HP, Yang CS (2016) A unique light-driven proton transportation signal in Halorhodopsin from Natronomonas pharaonis. Biophys J 111(12):2600–2607. https://doi.org/10.1016/j.bpj.2016.11.003
Chizhov I, Chernavskii DS, Engelhard M, Mueller KH, Zubov BV, Hess B (1996) Spectrally silent transitions in the bacteriorhodopsin photocycle. Biophys J 71(5):2329–2345
Duschl A, Lanyi JK, Zimányi L (1990) Properties and photochemistry of a halorhodopsin from the haloalkalophile, Natronobacterium pharaonis. J Biol Chem 265(3):1261–1267
Ernst OP, Lodowski DT, Elstner M, Hegemann P, Brown LS, Kandori H (2014) Microbial and animal rhodopsins: structures, functions, and molecular mechanisms. Chem Rev 114(1):126–163. https://doi.org/10.1021/cr4003769
Facciotti MT, Cheung VS, Nguyen D, Rouhani S, Glaeser RM (2003) Crystal structure of the bromide-bound D85S mutant of bacteriorhodopsin: principles of ion pum**. Biophys J 85(1):451–458. https://doi.org/10.1016/s0006-3495(03)74490-7
Feroz H, Ferlez B, Lefoulon C, Ren T, Baker CS, Gajewski JP, Lugar DJ, Gaudana SB, Butler PJ, Huhn J, Lam** M, Parak WJ, Hibberd JM, Kerfeld CA, Smirnoff N, Blatt MR, Golbeck JH, Kumar M (2018) Light-driven chloride transport kinetics of Halorhodopsin. Biophys J 115(2):353–360. https://doi.org/10.1016/j.bpj.2018.06.009
Govorunova EG, Sineshchekov OA, Li H, Spudich JL (2017) Microbial rhodopsins: diversity, mechanisms, and optogenetic applications. Annu Rev Biochem 86:845–872. https://doi.org/10.1146/annurev-biochem-101910-144233
Guijarro J, Engelhard M, Siebert F (2006) Anion uptake in halorhodopsin from Natromonas pharaonis studied by FTIR spectroscopy: consequences for the anion transport mechanism. Biochemistry 45(38):11578–11588
Harris A, Saita M, Resler T, Hughes-Visentin A, Maia R, Pranga-Sellnau F, Bondar AN, Heberle J, Brown LS (2017) Molecular details of the unique mechanism of chloride transport by a cyanobacterial rhodopsin. Phys Chem Chem Phys 20:3184–3199. https://doi.org/10.1039/c7cp06068h
Hasegawa C, Kikukawa T, Miyauchi S, Seki A, Sudo Y, Kubo M, Demura M, Kamo N (2007) Interaction of the halobacterial transducer to a halorhodopsin mutant engineered so as to bind the transducer: Cl− circulation within the extracellular channel. Photochem Photobiol 83(2):293–302. https://doi.org/10.1562/2006-06-09-RA-916
Hasemi T, Kikukawa T, Kamo N, Demura M (2016) Characterization of a cyanobacterial chloride-pum** rhodopsin and its conversion into a proton pump. J Biol Chem 291(1):355–362. https://doi.org/10.1074/jbc.M115.688614
Hasemi T, Kikukawa T, Watanabe Y, Aizawa T, Miyauchi S, Kamo N, Demura M (2019) Photochemical study of a cyanobacterial chloride-ion pum** rhodopsin. Biochim Biophys Acta Bioenerg 1860(2):136–146. https://doi.org/10.1016/j.bbabio.2018.12.001
Havelka WA, Henderson R, Oesterhelt D (1995) Three-dimensional structure of halorhodopsin at 7 Å resolution. J Mol Biol 247(4):726–738
Hazemoto N, Kamo N, Kobatake Y, Tsuda M, Terayama Y (1984) Effect of salt on photocycle and ion-pum** of halorhodopsin and third rhodopsinlike pigment of Halobacterium halobium. Biophys J 45(6):1073–1077. https://doi.org/10.1016/s0006-3495(84)84254-x
Hosaka T, Yoshizawa S, Nakajima Y, Ohsawa N, Hato M, DeLong EF, Kogure K, Yokoyama S, Kimura-Someya T, Iwasaki W, Shirouzu M (2016) Structural mechanism for light-driven transport by a new type of chloride ion pump, Nonlabens marinus Rhodopsin-3. J Biol Chem 291(34):17488–17495. https://doi.org/10.1074/jbc.M116.728220
Inoue K, Kubo M, Demura M, Kamo N, Terazima M (2009) Reaction dynamics of halorhodopsin studied by time-resolved diffusion. Biophys J 96(9):3724–3734
Inoue K, Ono H, Abe-Yoshizumi R, Yoshizawa S, Ito H, Kogure K, Kandori H (2013) A light-driven sodium ion pump in marine bacteria. Nat Commun 4:1678. https://doi.org/10.1038/ncomms2689
Inoue K, Kato Y, Kandori H (2014a) Light-driven ion-translocating rhodopsins in marine bacteria. Trends Microbiol 23:91–98. https://doi.org/10.1016/j.tim.2014.10.009
Inoue K, Koua FH, Kato Y, Abe-Yoshizumi R, Kandori H (2014b) Spectroscopic study of a light-driven chloride ion pump from marine bacteria. J Phys Chem B 118(38):11190–11199. https://doi.org/10.1021/jp507219q
Inoue K, Nomura Y, Kandori H (2016) Asymmetric functional conversion of eubacterial light-driven ion pumps. J Biol Chem 291(19):9883–9893. https://doi.org/10.1074/jbc.M116.716498
Kanada S, Takeguchi Y, Murakami M, Ihara K, Kouyama T (2011) Crystal structures of an O-like blue form and an anion-free yellow form of pharaonis halorhodopsin. J Mol Biol 413(1):162–176
Kandori H (2015) Ion-pum** microbial rhodopsins. Front Mol Biosci 2:52. https://doi.org/10.3389/fmolb.2015.00052
Kikukawa T, Kusakabe C, Kokubo A, Tsukamoto T, Kamiya M, Aizawa T, Ihara K, Kamo N, Demura M (2015) Probing the Cl−-pum** photocycle of pharaonis halorhodopsin: examinations with bacterioruberin, an intrinsic dye, and membrane potential-induced modulation of the photocycle. Biochim Biophys Acta Bioenerg 1847(8):748–758. https://doi.org/10.1016/j.bbabio.2015.05.002
Kim K, Kwon SK, Jun SH, Cha JS, Kim H, Lee W, Kim JF, Cho HS (2016) Crystal structure and functional characterization of a light-driven chloride pump having an NTQ motif. Nat Commun 7:12677. https://doi.org/10.1038/ncomms12677
Kouyama T, Kawaguchi H, Nakanishi T, Kubo H, Murakami M (2015) Crystal structures of the L1, L2, N, and O states of pharaonis halorhodopsin. Biophys J 108(11):2680–2690. https://doi.org/10.1016/j.bpj.2015.04.027
Kouyama T, Ihara K, Maki K, Chan SK (2018) Three-step isomerization of the retinal chromophore during the anion pum** cycle of Halorhodopsin. Biochemistry 57(41):6013–6026. https://doi.org/10.1021/acs.biochem.8b00631
Kubo M, Kikukawa T, Miyauchi S, Seki A, Kamiya M, Aizawa T, Kawano K, Kamo N, Demura M (2009) Role of Arg123 in light-driven anion pump mechanisms of pharaonis halorhodopsin. Photochem Photobiol 85(2):547–555
Lanyi JK (2004) Bacteriorhodopsin. Annu Rev Physiol 66:665–688
Ludmann K, Ibron G, Lanyi JK, Váró G (2000) Charge motions during the photocycle of pharaonis halorhodopsin. Biophys J 78(2):959–966
Matsuno-Yagi A, Mukohata Y (1977) Two possible roles of bacteriorhodopsin; a comparative study of strains of Halobacterium halobium differing in pigmentation. Biochem Biophys Res Commun 78(1):237–243
Matsuno-Yagi A, Mukohata Y (1980) ATP synthesis linked to light-dependent proton uptake in a red mutant strain of Halobacterium lacking bacteriorhodopsin. Arch Biochem Biophys 199(1):297–303
Mevorat-Kaplan K, Brumfeld V, Engelhard M, Sheves M (2006) The protonated Schiff base of halorhodopsin from Natronobacterium pharaonis is hydrolyzed at elevated temperatures. Photochem Photobiol 82(6):1414–1421
Mukohata Y, Kaji Y (1981) Light-induced membrane-potential increase, ATP synthesis, and proton uptake in Halobacterium halobium R1mR catalyzed by halorhodopsin: effects of N, N'-dicyclohexylcarbodiimide, triphenyltin chloride, and 3, 5-di-tert-butyl-4-hydroxybenzylidenemalononitrile (SF6847). Arch Biochem Biophys 206(1):72–76
Muneyuki E, Shibazaki C, Ohtani H, Okuno D, Asaumi M, Mogi T (1999) Time-resolved measurements of photovoltage generation by bacteriorhodopsin and halorhodopsin adsorbed on a thin polymer film. J Biochem 125(2):270–276
Muroda K, Nakashima K, Shibata M, Demura M, Kandori H (2012) Protein-bound water as the determinant of asymmetric functional conversion between light-driven proton and chloride pumps. Biochemistry 51:4677–4684
Niho A, Yoshizawa S, Tsukamoto T, Kurihara M, Tahara S, Nakajima Y, Mizuno M, Kuramochi H, Tahara T, Mizutani Y, Sudo Y (2017) Demonstration of a light-driven SO4 2− transporter and its spectroscopic characteristics. J Am Chem Soc 139:4376–4389. https://doi.org/10.1021/jacs.6b12139
Oesterhelt D (1995) Structure and function of halorhodopsin. Isr J Chem 35(3–4):475–494
Rüdiger M, Oesterhelt D (1997) Specific arginine and threonine residues control anion binding and transport in the light-driven chloride pump halorhodopsin. EMBO J 16(13):3813–3821
Sasaki J, Brown LS, Chon YS, Kandori H, Maeda A, Needleman R, Lanyi JK (1995) Conversion of bacteriorhodopsin into a chloride ion pump. Science 269(5220):73–75. https://doi.org/10.1126/science.7604281
Sato M, Kanamori T, Kamo N, Demura M, Nitta K (2002) Stopped-flow analysis on anion binding to blue-form halorhodopsin from Natronobacterium pharaonis: comparison with the anion-uptake process during the photocycle. Biochemistry 41(7):2452–2458
Sato M, Kikukawa T, Araiso T, Okita H, Shimono K, Kamo N, Demura M, Nitta K (2003) Roles of Ser130 and Thr126 in chloride binding and photocycle of pharaonis halorhodopsin. J Biochem 134(1):151–158
Sato M, Kubo M, Aizawa T, Kamo N, Kikukawa T, Nitta K, Demura M (2005) Role of putative anion-binding sites in cytoplasmic and extracellular channels of Natronomonas pharaonis halorhodopsin. Biochemistry 44(12):4775–4784
Scharf B, Engelhard M (1994) Blue halorhodopsin from Natronobacterium pharaonis: wavelength regulation by anions. Biochemistry 33(21):6387–6393
Schobert B, Lanyi JK (1982) Halorhodopsin is a light-driven chloride pump. J Biol Chem 257(17):10306–10313
Seki A, Miyauchi S, Hayashi S, Kikukawa T, Kubo M, Demura M, Ganapathy V, Kamo N (2007) Heterologous expression of pharaonis halorhodopsin in Xenopus laevis oocytes and electrophysiological characterization of its light-driven Cl− pump activity. Biophys J 92(7):2559–2569. https://doi.org/10.1529/biophysj.106.093153
Shibasaki K, Shigemura H, Kikukawa T, Kamiya M, Aizawa T, Kawano K, Kamo N, Demura M (2013) Role of Thr218 in the light-driven anion pump halorhodopsin from Natronomonas pharaonis. Biochemistry 52(51):9257–9268. https://doi.org/10.1021/bi401295e
Shibata M, Muneda N, Sasaki T, Shimono K, Kamo N, Demura M, Kandori H (2005) Hydrogen-bonding alterations of the protonated Schiff base and water molecule in the chloride pump of Natronobacterium pharaonis. Biochemistry 44(37):12279–12286. https://doi.org/10.1021/bi050726d
Tamogami J, Kikukawa T, Miyauchi S, Muneyuki E, Kamo N (2009) A tin oxide transparent electrode provides the means for rapid time-resolved pH measurements: application to photoinduced proton transfer of bacteriorhodopsin and proteorhodopsin. Photochem Photobiol 85(2):578–589. https://doi.org/10.1111/j.1751-1097.2008.00520.x
Tittor J, Haupts U, Haupts C, Oesterhelt D, Becker A, Bamberg E (1997) Chloride and proton transport in bacteriorhodopsin mutant D85T: different modes of ion translocation in a retinal protein. J Mol Biol 271(3):405–416. https://doi.org/10.1006/jmbi.1997.1204
Tsukamoto T, Yoshizawa S, Kikukawa T, Demura M, Sudo Y (2017) Implications for the light-driven chloride ion transport mechanism of Nonlabens marinus rhodopsin 3 by its photochemical characteristics. J Phys Chem B 121(9):2027–2038. https://doi.org/10.1021/acs.jpcb.6b11101
Váró G, Brown LS, Sasaki J, Kandori H, Maeda A, Needleman R, Lanyi JK (1995a) Light-driven chloride ion transport by halorhodopsin from Natronobacterium pharaonis. I. The photochemical cycle. Biochemistry 34(44):14490–14499
Váró G, Needleman R, Lanyi JK (1995b) Light-driven chloride ion transport by halorhodopsin from Natronobacterium pharaonis. II. Chloride release and uptake, protein conformation change, and thermodynamics. Biochemistry 34(44):14500–14507
Váró G, Zimányi L, Fan X, Sun L, Needleman R, Lanyi JK (1995c) Photocycle of halorhodopsin from Halobacterium salinarium. Biophys J 68(5):2062–2072
Váró G, Brown LS, Needleman R, Lanyi JK (1996) Proton transport by halorhodopsin. Biochemistry 35(21):6604–6611. https://doi.org/10.1021/bi9601159
Walter TJ, Braiman MS (1994) Anion-protein interactions during halorhodopsin pum**: halide binding at the protonated Schiff base. Biochemistry 33(7):1724–1733. https://doi.org/10.1021/bi00173a015
Yoshizawa S, Kumagai Y, Kim H, Ogura Y, Hayashi T, Iwasaki W, DeLong EF, Kogure K (2014) Functional characterization of flavobacteria rhodopsins reveals a unique class of light-driven chloride pump in bacteria. Proc Natl Acad Sci U S A 111(18):6732–6737. https://doi.org/10.1073/pnas.1403051111
Acknowledgments
The author thanks the collaborators and the financial supports from JSPS KAKENHI (17K07326, 26440042, 23510251, 19614001) and the Global Station for Soft Matter, a project of the Global Institution for Collaborative Research and Education at Hokkaido University.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kikukawa, T. (2021). Functional Mechanism of Cl−-Pump Rhodopsin and Its Conversion into H+ Pump. In: Yawo, H., Kandori, H., Koizumi, A., Kageyama, R. (eds) Optogenetics. Advances in Experimental Medicine and Biology, vol 1293. Springer, Singapore. https://doi.org/10.1007/978-981-15-8763-4_4
Download citation
DOI: https://doi.org/10.1007/978-981-15-8763-4_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-8762-7
Online ISBN: 978-981-15-8763-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)