Summary
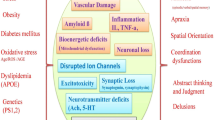
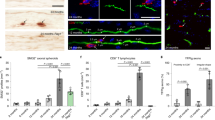
Recent evidence indicates that programmed cell death (apoptosis) may contribute to neuronal death in Alzheimer’s disease (AD). In situ data derived from post mortem brain tissue indicate that DNA fragmentation which represents an important and typical apoptotic feature is markedly increased in brain cells of AD patients compared to controls. Furthermore, in vitro studies demonstrate that the peptide β-amyloid (Aβ) and its fragments induce apoptosis in neuronal cell cultures. One possible mechanism initiating apoptosis could be free radical generation by the peptide leading to oxidative stress. In a wide range of cell types common morphological and molecular events occur during apoptosis and several genes appear to be involved. Particularly in lymphocytes, apoptosis plays an important physiological role. Our data demonstrate that similar oxidative stressors induce apoptosis in mature human lymphoctes as in neurons. In addition, first evidence indicates that susceptibility to apoptosis is altered in lymphocytes from AD patients compared to non-demented controls. Our preliminary findings suggest that changes of the individual sensitivity to undergo cellular apoptosis are already detectable in lymphocytes from AD patients, probably as a consequence of genetic as well as other risk factors. Therefore, this biochemical marker might have the potential for identifying individuals at risk of the diseases.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Barbieri D, Grassilli E, Salvioli S, Franceschini MG, Franchini A, Bellesia E, Salomoni P, Negro P, Capri M, Tojano L, Franceschi C (1994) D-ribose and deoxy-D-ribose induce apoptosis in human quiescent peripheral blood mononuclear cells. Biochem Biophys Res Commun 201: 1109–1116
Behl C, Davis JB, Lesley R, Schubert D (1994) Hydrogen peroxide mediates amyloid β protein toxicity. Cell 77: 817–827
Butterfield DA, Hensley K, Harris M, Mattson M, Carney J (1994) β-Amyloid peptide free radical fragments initiate synaptosomal lipoperoxidation in a sequence-specific fashion: implications to Alzheimer’s disease. Biochem Biophys Res Commun 200: 710–715
Cotman CW, Anderson A (1995) A potential role for apoptosis in neurodegeneration and Alzheimer’s disease. Mol Neurobiol 10: 19–45
Cotman CW, Whittemoore ER, Watt JA, Anderson AJ, Loo DT (1994) Possible role of apoptosis in Alzheimer’s disease. Ann NY Acad Sci 747: 36–49
Davis-Salinas J, Saporito-Irwin SM, Cotman CW, van Norstrand WE (1995) Amyloid β protein induces its own production in cultured degenerating cerebrovascular smooth muscle cells. J Neurochem 65: 931–934
Eckert A, Hartmann H, Muller WE (1993) β-Amyloid protein enhances the mitogen-induced calcium response in circulating human lymphocytes. FEBS Lett 330: 49–52
Eckert A, Forstl H, Hartmann H, Czech C, Mönning U, Beyreuther K, Müller WE (1995) The amplifying effect of β-amyloid on cellular calcium signalling is reduced in Alzheimer’s disease. Neuroreport 6: 1199–1202
Eckert A, Cotman CW, Zerfass R, Hennerici M, Müiler WE (1996) Vulnerability of lymphocytes against programmed cell death in Alzheimer’s disease. Soc Neurosci 22: 827
Eckert A, Cotman CW, Zerfass R, Hennerici M, Müller WE (1998) Enhanced vulnerability to apoptotic cell death in sporadic Alzheimer’s disease. Neuroreport (in press)
Forloni G, Chiesa R, Smiroldo S, Verga L, Salmona M, Tagliavini F, Angeretti N (1993) Apoptosis mediated neurotoxicity induced by chronic application of β amyloid fragment 25–35. Neuroreport 4: 523–526
Hensley K, Carney JM, Mattson MP, Aksenova M, Harris M, Wu JF, Floyd RA, Butterfield DA (1994) A model for β-amyloid aggregation and neurotoxicity based on free radical generation by the peptide: relevance to Alzheimer disease. Proc Natl Acad Sci USA 9: 3270–3274
Lassmann H, Bancher C, Breitschopf H, Wegiel J, Bobinski M, Jellinger K, Wisniewski HM (1995) Cell death in Alzheimer’s disease evaluated by DNA fragmentation in situ. Acta Neuropathol 89: 35–41
Leonardo MJ (1991) Interleukin-2 programs mouse αβ T lymphocytes for apoptosis. Nature 353: 858–861
Loo DT, Copani A, Pike CJ, Whittemoore ER, Walencewicz AJ, Cotman CW (1993) Apoptosis is induced by β-amyloid in cultured central nervous system neurons. Proc Natl Acad Sci USA 90: 7951–7955
Mark RJ, Blanc EM, Mattson MP (1996) Amyloid β-peptide and oxidative cellular injury in Alzheimer’s disease. Mol Neurobiol 12: 211–224
Nicoletti I, Migliorati G, Pagliacci MC, Grignani F, Riccardi C (1991) A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J Immunol Meth 139: 271–279
Pike CJ, Burdick D, Walencewicz A, Glabe CG, Cotman CW (1993) Neurodegeneration induced by β-amyloid peptides in vitro: the role of peptide assembly state. J Neurosci 13: 1676–1687
Su JH, Anderson AJ, Cummings BJ, Cotman CW (1994) Immunohistochemical evidence for apoptosis in Alzheimer’s disease. Neuroreport 5: 2529–2533
Watt JA, Pike CJ, Wassermann AJ, Cotman CW (1994) Ultrastructural analysis of β-amyloid-induced apoptosis in cultured hippocampal neurons. Brain Res 661:147–156
Whittemore ER, Loo DT, Cotman CW (1994) Exposure to hydrogen peroxide induces cell death via apoptosis in cultured rat cortical neurons. Neuroreport 5: 1485–1488
Zheng L, Fisher G, Miller RE, Peschon J, Lynch DH, Leonardo MJ (1995) Induction of apoptosis in mature T cells by tumor necrosis factor. Nature 377: 348–351
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1998 Springer-Verlag/Wien
About this paper
Cite this paper
Eckert, A., Cotman, C.W., Zerfass, R., Hennerici, M., Müller, W.E. (1998). Lymphocytes as cell model to study apoptosis in Alzheimer’s disease: vulnerability to programmed cell death appears to be altered. In: Gertz, HJ., Arendt, T. (eds) Alzheimer’s Disease — From Basic Research to Clinical Applications. Journal of Neural Transmission. Supplementa, vol 54. Springer, Vienna. https://doi.org/10.1007/978-3-7091-7508-8_25
Download citation
DOI: https://doi.org/10.1007/978-3-7091-7508-8_25
Publisher Name: Springer, Vienna
Print ISBN: 978-3-211-83112-0
Online ISBN: 978-3-7091-7508-8
eBook Packages: Springer Book Archive