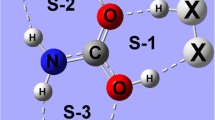
Abstract
The interaction of hydrogen sulfide (H2S) with F, Cl, Br, and OH is investigated using ab initio methods to identify the two-center three-electron hemibond responsible for their complexation. The binding energies are found to be stronger than those in the analogous water complexes, but follow the same trend of increasing strength: F > Cl > Br > OH. The radicals are located nearly perpendicular to the H2S plane forming an angle of about 90˚. Analysis of molecular orbitals and natural bond orbitals are carried out to understand the energetics, structures, and bonding characteristics of these hemibonded complexes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Additional information
Dedicated to Professor Guosen Yan and published as part of the special collection of articles celebrating his 85th birthday.
Rights and permissions
Copyright information
© 2015 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Alday, B., Johnson, R., Li, J., Guo, H. (2015). Hemibond complexes between H2S and free radicals (F, Cl, Br, and OH). In: Guo, H., **e, D., Yang, W. (eds) Guosen Yan. Highlights in Theoretical Chemistry, vol 8. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-47845-5_20
Download citation
DOI: https://doi.org/10.1007/978-3-662-47845-5_20
Received:
Accepted:
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-47844-8
Online ISBN: 978-3-662-47845-5
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)