Abstract
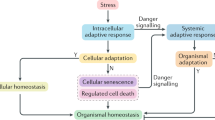
Since the evolution of the cellular phenotype, prokaryotic and eukaryotic cells have had to cope with adverse changes in their environment. Although cells have evolved many distinct stress responses, this chapter will focus on three major, highly conserved, response systems, i.e., the genotoxic response system, which is activated by DNA damage; the oxidative stress response system, which is activated by excess reactive oxygen species (ROS) and imbalances in the oxidant/antioxidant status within cells; and the heat shock response, which is activated by exposure to heat and other agents that adversely affect protein folding (Fig. 1). The sections dealing with each of the stress response systems begin with a description of the prokaryotic stress response because, in most instances, the prokaryotic systems are the best understood. This is followed by a discussion of the eukaryotic stress response systems, focusing on yeast and mammals. Finally, each section concludes with a discussion about what is known concerning the induction of these stress response systems in mammalian embryos, particularly postimplantation mammalian embryos. Normal embryonic development requires a precisely orchestrated chain of temporal and spatial events, and any alterations in this chain could lead to altered development and subsequent pathogenesis. Although the mammalian embryo develops within the protective environment of the uterus, this protection is not absolute and we now know that mammalian development can be perturbed by a wide variety of chemical and physical agents, many of which are known to induce one or more of these stress systems in nonembryonic systems. Thus understanding the embryo’s stress response capabilities is essential to the understanding of how developmental toxicants exert their toxicity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Abbott BD, Perdew GH, Birnbaume LS (1994) Ah receptor in embryonic mouse palate and effects of TCDD on receptor expression. Toxicol Appl Pharmacol 126:16–25
Aggeler J, Murnane JP (1990) Enhanced expression of procollagenase in ataxia-telangiectasia and xeroderma pigmentosum fibroblasts. In vitro Cell Dev Biol 26: 915–922
Anderson CW (1993) DNA damage and the DNA-activated protein kinase. Trends Biochem Sci 18:433-437
Anderson RL, Van-Kersen I, Kraft PE, Hahn GM (1989) Biochemical analysis of heat resistant mouse tumor cell strains: a new member of the HSP70 family. Mol Cell Biol 9: 3509–3516
Angelidis CE, Lazaridis I, Pagoulatos GN (1991) Constitutive expression of heat shock protein 70 in mammalian cells confers thermoresistance. Eur J Biochem 199: 35–39
Anson JF, Hinson WG, Pikin JL, Kwarta RF, Hansen DK, Young JF, Burns ER, Casciano DA (1987) Retinoic acid induction of stress proteins in fetal mouse limb buds. Dev Biol 121: 542–547
Anson JF, Laborde JB, Pipkin JL, Hinson WJ, Hansen DK, Sheehan DM, Young JF (1991) Target tissue specificity of retinoic acid-induced stress proteins and malformations in mice (see comments). Teratology 44: 19–28
Applegate LA, Luscher P, Tyrrell RM (1991) Induction of heme oxygenase: a general response to oxidant stress in cultured mammalian cells. Cancer Res 51: 974–978
Bauman JW, Liu J, Liu YP, Klaassen CD (1991) Increase in metallothionein produced by chemicals that induce oxidative stress. Tox Appl Pharmacol 110: 347–354
Bennett GD, Mohl VK, Finnell RH (1990) Embryonic and maternal heat shock responses to a teratogenic hyperthermic insult. Reprod Toxicol 4: 113–119
Bensaude O, Babinet C, Morange M, Jacob F (1983) Heat shock proteins, first major products of zygotic gene activity in mouse embryo. Nature 305: 331–333
Bevilacqua A, Mangia P (1993) Activity of a microinjected inducible murine hsp68 gene promoter depends on plasmid configuration and the presence of heat shock elements in mouse dictyate oocytes but not in two-cell embryos. Dev Genet 14: 92–102
Brach MA, Hass R, Sherman ML, Gunji H, Weichselbaum R, Kufe D (1991) Ionizing radiation induces expression and binding activity of the nuclear factor kappa B. J Clin Invest 88: 691–695
Carper SW, Duffy JJ, Gerner EW (1987) Heat shock proteins in thermotolerance and other cellular processes. Cancer Res 47: 5249–5255
Chrétien P, Landry J (1988) Enhanced heat constitutive expression of the 27 Kda heat shock proteins in heat resistant variants from Chinese hamster cells. J Cell Physiol 137: 157–166
Christians E, Campion E, Thompson EM, Renard JP (1995) Expression of the HSP 70.1 gene, a landmark of early zygotic activity in the mouse embryo, is restricted to the first burst of transcription. Development 121: 113–122
Christman MF, Morgan RW, Jacobson FS, Ames BN (1985) Positive control of a regulon for defense against oxidative stress and some heat shock proteins in Salmonella typhimurium. Cell 41: 753–762
Christman MF, Storz G, Ames BN (1989) OxyR, a positive regulator of hydrogen peroxide-inducible genes in Escherichia coli and Salmonella typhimurium, is homologous to a family of bacterial regulatory proteins. Proc Natl Acad Sci USA 86: 3484–3488
de Murcia G, Menissier-de-Murcia J, Schreiber V (1994) Poly(ADP-ribose) polymerase: a molecular nick-sensor [published erratum appears in Trends Biochem Sci 19: 250]. Trends Biochem Sci 19: 172–176
el Hage S, Singh SM (1990) Temporal expression of genes encoding free radical-metabolizing enzymes is associated with higher mRNA levels during in utero development in mice. Dev Genet 11: 149–159
Elledge SJ, Davis RW (1987) Identification and isolation of the gene encoding the small subunit of ribonucleotide reductase from Saccharomyces cerevisiae: DNA da-mage-inducible gene required for mitotic viability. Mol Cell Biol 7: 2783–2793
Erickson UJ, Borg LA (1993) Diabetes and embryonic malformations. Role of substrate-induced free-oxygen radical production for dysmorphogenesis in cultured rat embryos. Diabetes 42: 411–19
Evensen G, Seeberg E (1982) Adaptation to alkylation resistance involves the induction of a DNA glycosylase. Nature 296: 773–775
Finnell RH, Van-Waes M, Bennett GD, Eberwine JH (1993) Lack of concordance between heat shock proteins and the development of tolerance to teratogen-in-duced neural tube defects. Dev Genet 14: 137–147
Fisher BR, Kimmel GL, Kimmel CA, Heredia DJ (1991) The association of heat-induced alterations in protein synthesis with somite defects in rat embryos. Teratology 43: 465
Fornace AJ Jr (1992) Mammalian genes induced by radiation; activation of genes associated with growth control. Annu Rev Genet 26: 507–526
Fornace AJ Jr, Nebert DW, Hollander MC, Luethy JD, Papathanasiou M, Fargnoli J, Holbrook NJ (1989) Mammalian genes coordinately regulated by growth arrest signals and DNA-damaging agents. Mol Cell Biol 9: 4196–4203
Fornace AJ Jr, Jackman J, Hollander MC, Hoffman-Liebermann B, Liebermann DA (1992) Genotoxic-stress-response genes and growth-arrest genes, gadd, MyD, and other genes induced by treatments eliciting growth arrest. Ann NY Acad Sci 663: 139–153
Franklin WA, Haseltine WA (1984) Removal of UV light-induced pyrimidine-pyr-imidone (6–4) products from Escherichia coli DNA requires the uvrA, uvrB, and uvrC gene products. Proc Natl Acad Sci USA 81:3821–3824
Friedberg EC (1988) Deoxyribonucleic acid repair in the yeast Saccharomyces cerevisiae. Microbiol Rev 52:70–102
Georgopoulos C, Welch WJ (1993) Role of the major heat shock proteins as molecular chaperones. Annu Rev Cell Biol 9:601–634
German J (1984) Embryonic stress hypothesis of teratogenesis. Am J Med 76:293–301
German J, Louie E, Banerjee D (1986) The heat shock response in vivo: experimental induction during mammalian organogenesis. Teratog Carcinog Mutagen 6:555–562
Gewirtz DA (1993) DNA damage, gene expression, growth arrest and cell death. Oncol Res 5: 397–408
Goff SA, Goldberg AL (1985) Production of abnormal proteins in E. coli stimulates transcription of Ion and other heat shock genes. Cell 41:587–595
Greenberg JT, Demple B (1989) A global response induced in Escherichia coli by redox cycling agents overlaps with that induced by peroxide stress. J Bacteriol 171:3933–3939
Hahnel AC, Gifford DJ, Heikkila JJ, Schultz GA (1986) Expression of the major heat shock protein (hsp 70) family during early mouse embryo development. Teratog Carcinog Mutagen 6:493–510
Hallahan DE, Spriggs DR, Bockett MA, Kufe DW, Weichselbaum RR (1989) Increased tumor necrosis factor alpha mRNA after cellular exposure to ionizing radiation. Proc Natl Acad Sci USA 86:10104–10107
Hanawalt PC (1994) Transcription-coupled repair and human disease. Science 266:1957–1958
Hansen DK, Anson JF, Hinson WG, Pipkin JL Jr (1988) Phenytoin-induced stress protein synthesis in mouse embryonic tissue. Proc Soc Exp Biol Med 189:136–140
Harris C, Juchau MR, Mirkes PE (1991) Role of glutathione and hsp 70 in the acquisition of thermotolerance in postimplantation rat embryos. Teratology 43:229–239
Heikkila JJ (1993) Heat shock gene expression and development. II. An overview of mammalian and avian developmental systems. Dev Genet 14:87–91
Hendrey J, Kola I (1991) Thermolability of mouse oocytes is due to the lack of expression and/or inducibility of Hsp 70. Mol Reprod Dev 28:1–8
Herrlich P, Angel P, Rahmsdorf HJ, Mallick U, Poting A, Hieber L, Lucke-Huhle C, Schorpp M (1986) The mammalian genetic stress response. Adv Enzyme Regul 25:485–504
Herrlich P, Sachsenmaier C, Radler-Pohl A, Gebel S, Bluttner C, Rahmsdorf HJ (1994) The mammalian UV response: mechanism of DNA damage induced gene expression. Adv Enzyme Regul 34:381–395
Hidalgo E, Demple B (1994) An iron-sulfur center essential for transcriptional activation by the redox-sensing SoxR protein. EMBO J 13:138–146
Hightower LE (1980) Cultured animal cells exposed to amino acid analogues or pur-omycin rapidly synthesize several polypeptides. J Cell Physiol 102:407–427
Higo H, Lee JY, Satow Y, Higo K (1989) Elevated expression of protooncogenes accompany enhanced induction of heat-shock genes after exposure of rat embryos in utero to ionizing radiation. Teratogen Carcinogen Mutagen 9:191–198
Holley JA, Janssen YM, Mossman BT, Taatjes DJ (1992) Increased manganese superoxide dismutase protein in type II epithelial cells of rat lungs after inhalation of crocidolite asbestos or cristobalite silica. Am J Pathol 141: 475–485
Honda K, Hatayama T, Takahashi K, Yukioka M (1991) Heat shock proteins in human and mouse embryonic cells after exposure to heat shock or teratogenic agents. Teratogen Carcinogen Mutagen 11:235–244
Jackman J, Alamo I Jr, Fornace AJ Jr (1994) Genotoxic stress confers preferential and coordinate messenger RNA stability on the five gadd genes. Cancer Res 54:5656–5662
Janssen YM, Marsh JP, Absher MP, Hemenway D, Vacek PM, Leslie KO, Born PJ, Mossman BT (1992) Expression of antioxidant enzymes in rat lungs after inhalation of asbestos or silica. J Biol Chem 267:10625–10630
Janssen YM, van-Houten B, Born PJ, Mossman BT (1993) Cell and tissue responses to oxidative damage. Lab Invest 69:261–274
Johnson AL, Barker DG, Johnston LH (1986) Induction of yeast DNA ligase genes in exponential and stationary phase cultures in response to DNA damaging agents. Curr Genet 11:107–112
Johnston LH, White JH, Johnson AL, Lucchini G, Plevani P (1987) The yeast DNA polymerase I transcript is regulated in both the mitotic cell cycle and in meiosis and is also induced after DNA damage. Nucleic Acids Res 15:5017–5030
Johnston RN, Kucey BL (1988) Competitive inhibition of hsp 70 gene expression causes thermosensitivity. Science 242:1551–1554
Jornot L, Junod AF (1992) Response for human endothelial cell antioxidant enzymes to hyperoxia. Am J Respir Cell Mol Biol 6:107–115
Kam**a HH, Brunsting JF, Stege GJ, Konings AW, Landry J (1994) Cells over-expressing Hsp 27 show accelerated recovery from heat induced nuclear protein aggregation. Biochem Biophys Res Commun 204:1170–1177
Kapron-Brás CM, Hales BF (1991) Heat-shock induced tolerance to the embryotoxic effects of hyperthermia and cadmium in mouse embryos in vitro. Teratology 43:83–94
Kapron-Brás CM, Hales BF (1992) Genetic differences in heat-induced tolerance to cadmium in cultured mouse embryos are not correlated with changes in a 68-kD heat shock protein. Teratology 46:191–200
Karran P, Hjelmgren T, Lindahl T (1982) Induction of a DNA glycosylase for N-methylated purines is part of the adaptive response to alkylating agents. Nature 296:770–773
Kartasova T, van de Putte P (1988) Isolation, characterization, and UV-stimulated expression of two families of genes encoding polypeptides of related structure in human epidermal keratinocytes. Mol Cell Biol 8:2195–2203
Kataoka H, Sekiguchi M (1985) Molecular cloning and characterization of the alkB gene of Escherichia coli. Mol Gen Genet 198:263–269
Kataoka H, Yamamoto Y, Sekiguchi M (1983) A new gene (alkB) of Escherichia coli that controls sensitivity to methyl methane sulfonate. J Bacteriol 153:1301–1307
Kelley PM, Schlesinger M J (1978) The effect of amino acid analogues and heat shock on gene expression in chicken embryo fibroblasts. Cell 15:1277–1286
Kenyon CJ, Walker GC (1980) DNA-damaging agents stimulate gene expression at specific loci in Escherichia coli. Proc Natl Acad Sci USA 77:2819–2823
Key se SM (1990) Oxidant stress lead to transcriptional activation of the human heme oxygenase gene in cultured cells. Mol Cell Biol 10:4967–4969
Keyse SM (1993) The induction of gene expression in mammalian cells by radiation. Cancer Biol 4:119–128
Kimmel CA, Kimmel GL, Lu C, Heredia DJ, Fisher BR, Brown NT (1991) Stress protein synthesis as a potential biomarker for heat-induced developmental toxicity. Teratology 43: 465
Kimmel CA, Cuff JM, Kimmel GL, Heredia DJ, Tudor N, Silverman PM (1993) Skeletal development following heat exposure in the rat. Teratology 47:229–242
Kimmel CA, Claggett TW, Kimmel GL, Tudor N, Hogan KA (1994) Segmentation anomalies and stress proteins induced by heat on gestation days (GD) 10, 11 or 12 in the rat. Teratology 49: 409
Kimmel GL, Cuff JM, Kimmel CA, Heredia DJ, Tudor N, Silverman PM (1993) Embryonic development in vitro following short-duration exposure to heat. Teratology 47:243–251
Kimmel GL, Claggett TW, Kimmel CA, Bolou B, Todor N (1995) The progression of effects of heat stress on somitogenesis in the rat. Teratology 51:157–158
Landini P, Hajec LI, Volkert MR (1994) Structure and transcriptional regulation of the Escherichia coli adaptive response gene aidB. J Bacteriol 176:6583–6589
Landry J, Chrétien P, Lambert H, Hickey E, Weber LA (1989) Heat shock resistance conferred by expression of the human HSP27 gene in rodent cells. J Cell Biol 109:7–15
Laszlo A, Li GC (1985) Heat resistant variants of Chinese hamster fibroblasts altered in expression of heat shock protein. Proc Natl Acad Sci USA 82:8029–8033
Lavoie JN, Gingras-Breton G, Tanguay RM, Landry J (1993) Induction of Chinese hamster HSP27 gene expression in mouse cells confers resistance to heat shock. HSP27 stabilization of the microfilament organization. J Biol Chem 268:3420–3429
Lavoie JN, Lambert H, Hickey E, Weber LA, Landry J (1995) Modulation of cellular thermoresistance and actin filament stability accompanies phosphorylation-in-duced changes in the oligomeric structure of heat shock protein 27. Mol Cell Biol 15:505–516
Lemotte PK, Walker GC (1985) Induction and autoregulation of ada, a positively acting element regulating the response of Escherichia coli K-12 to methylating agents. J Bacteriol 161:888–895
Li GC, Laszlo A (1985) Thermotolerance in mammalian cells: a possible role for heat shock proteins. In: Atkinson BG, Waiden DB (eds) Changes in eukaryotic gene expression in response to environmental stress. Academic, Orlando, p 227
Li GC, Werb Z (1982) Correlation between synthesis of heat shock proteins and development of thermotolerance in Chinese hamster fibroblasts. Proc Natl Acad Sci USA 79:3218–3222
Li Z, Demple B (1994) SoxS, an activator of superoxide stress genes in Escherichia coli. Purification and interaction with DNA. J Biol Chem 269:18371–18377
Li GC, Li LG, Liu YK et al. (1991) Thermal response of rat fibroblasts stably trans-fected with the human 70-kDa heat shock protein-encoding gene. Proc Natl Acad Sci USA 88:1681–1685
Little JW, Mount DW (1982) The SOS regulatory system of Escherichia coli. Cell 29:11–22
Little SA, Mirkes PE (1994) Induction of the growth arrest-DNA damage induced gene (gadd 153) in rat embryos in response to genotoxic agents. Teratology 49: 400
Lloyd RG, Benson FE, Shurvinton CE (1984) Effect of ruv mutations on recombination and DNA repair in Escherichia coli Kl2. Mol Gen Genet 194:303–309
Lord KA, Hoffman-Liebermann B, Liebermann DA (1990) Complexity of the immediate early response of myeloid cells to terminal differentiation and growth arrest includes ICAM-1, Jun-B and histone variants. Oncogene 5:387–396
Maines MD (1984) New Developments in the regulation of heme metabolism and their implications. Crit Rev Toxicol 12:241–314
Marquez CM, Sneed PK, Li GC, Mak JY, Phillips TL (1994) HSP 70 synthesis in clinical hyperthermia patients: preliminary results of a new technique. Int J Radiat Oncol Biol Phys 28: 425–30
Matts RL, Xu Z, Pal JK, Chen J J (1992) Interactions of the heme-regulated eIF-2 alpha kinase with heat shock proteins in rabbit reticulocyte lysates. J Biol Chem 267:18160–18167
McCarthy TV, Karran P, Lindahl T (1984) Inducible repair of O-alkylated DNA pyrimidines in Escherichia coli. EMBO J 3:545–550
McClanahan T, McEntee K (1984) Specific transcripts are elevated in Saccharomyces cerevisiae in response to DNA damage. Mol Cell Biol 4:2356–2363
Mirkes PE (1985) Hyperthermia-induced heat shock response and thermotolerance in postimplantation rat embryos. Dev Biol 119:115–122
Mirkes PE, Cornel L (1992) A comparison of sodium arsenite-and hyperthermia-induced stress responses and abnormal development in cultured postimplantation rat embryos. Teratology 46:251–259
Mirkes PE, Doggett B (1992) Accumulation of heat shock protein 72 (hsp 72) in postimplantation rat embryos after exposure to various periods of hyperthermia (40–43 °C) in vitro: evidence that heat shock protein 72 is a biomarker of heat-induced embryo toxicity. Teratology 46:301–309
Mirkes PE, Grace RH, Little SA (1991) Developmental regulation of heat shock protein synthesis and HSP 70 RNA accumulation during postimplantation rat embryogenesis. Teratology 44:77–89
Mirkes PE, Doggett B, Cornel L (1994) Induction of heat shock response (HSP 72) in rat embryos exposed to selected chemical teratogens. Teratology 49:135–142
Miskin R, Ben-Ishai R (1981) Induction of plasminogen activator by UV light in normal and xeroderma pigmentosum fibroblasts. Proc Natl Acad Sci USA 78:6236–6240
Modrich P (1994) Mismatch repair, genetic stability and cancer. Science 266:1959–1960
Morange M, Diu A, Bensaude O, Babinet C (1984) Altered expression of heat shock proteins in embryonal carcinoma and mouse early embryonic cells. Mol Cell Biol 4:730–735
Morimoto RI (1993) Cells in stress: transcriptional activation of heat shock genes. Science 259:1409–1410
Nakai A, Morimoto RI (1993) Characterization of a novel chicken heat shock transcription factor, heat shock factor 3, suggests a new regulatory pathway. Mol Cell Biol 13:1983–1997
Nebert DW (1994) Drug-metabolizing enzymes in ligand-modulated transcription. Biochem Pharmacol 47:25–37
Nebert DW, Petersen DD, Fornace AJ Jr (1990) Cellular responses to oxidative stress: the [Ah] gene battery as a paradigm. Environ Health Perspect 88:13–25
Nover L (1991) Heat shock response. CRC Press, Boca Raton
Papathanasiou MA, Fornace AJ Jr (1991) DNA-damage inducible genes. Cancer Treat Res 57:13–36
Pongratz I, Mason GG, Poellinger L (1992) Dual roles of the 90-kDa heat shock protein hsp90 in modulating functional activities of the dioxin receptor. Evidence that the dioxin receptor functionally belongs to a subclass of nuclear receptors which require hsp 90 both for ligand binding activity and repression of intrinsic DNA binding activity. J Biol Chem 267:13728–13734
Reimer DL, Singh SM (1990) In situ hybridization studies on murine catalase mRNA expression during embryonic development. Dev Genet 11:318–325
Riabowol KT, Mizzen LA, Welch WJ (1988) Heat shock is lethal to fibroblasts mi-croinjected with antibodies against hsp 70. Science 242: 433–36
Ritossa F (1962) A new puffing pattern induced by temperature shock and DNP in Drosophila. Experientia 18:571–573
Ron D, Habener JF (1992) CHOP, a novel developmentally regulated nuclear protein that dimerizes with transcription factors C/EBP and LAP and functions as a dominant negative inhibitor of gene transcription. Genes Dev 6: 439–453
Ruby SW, Szostak JW (1985) Specific Saccharomyces cerevisiae genes are expressed in response to DNA-damaging agents. Mol Cell Biol 5:75–84
Rushmore TH, Morton MR, Pickett CB (1991) The antioxidant responsive element. Activation by oxidative stress and identification of the DNA consensus sequence required for functional activity. J Biol Chem 266:11632–11639
Samson L, Cairns J (1977) A new pathway for DNA repair in Escherichia coli. Nature 267:281–283
Sancar A (1994) Mechanisms of DNA excision repair. Science 266:1954–1956
Sancar A, Rupp WD (1983) A novel repair enzyme: UVRABC excision nuclease of Escherichia coli cuts a DNA strand on both sides of the damaged region. Cell 33:249–260
Sanchez Y, Lindquist SL (1990) HSP 104 required for induced thermotolerance. Science 248:1112–1115
Sanchez Y, Taulien J, Borkovich KA, Lindquist S (1992) HSP104 is required for tolerance to many forms of stress. EMBO J 11:2357–2364
Sedgwick B (1983) Molecular cloning of a gene which regulates the adaptive response to alkylating agents in Escherichia coli. Mol Gen Genet 191:466–472
Shaknovich R, Shue G, Kohtz DS (1992) Conformational activation of a basic helix-loop-helix protein (MyoDl) by the C-terminal region of murine HSP90 (HSP84). Mol Cell Biol 12:5059–5068
Sistonen L, Sarge KD, Morimoto RI (1994) Human heat shock factors 1 and 2 are differentially activated and can synergistically induce hsp 70 gene transcription. Mol Cell Biol 14:2087–2099
Smith DF, Toft DO (1993) Steroid receptors and their associated proteins. Mol Endocrinol 7:4–11
Somashekaraiah BV, Padmaja K, Prasad AR (1992) Lead-induced lipid peroxidation and antioxidant defense components of develo** chick embryos. Free Radic Biol Med 13:107–114
Stocker R (1990) Induction of haem oxygenase as a defence against oxidative stress. Free Radic Res Commun 9:101–112
Teo I, Sedgwick B, Demple B, Li B, Lindahl T (1984) Induction of resistance to alkylating agents in E. Coli: the ada+ gene product serves both as a regulatory protein and as an enzyme for repair or mutagenic damage. EMBO J 3:2151–2157
Vile GF, Tyrrell RM (1993) Oxidative stress resulting from ultraviolet A irradiation of human skin fibroblasts leads to a heme oxygenase-dependent increase in ferritin. J Biol Chem 268:14678–14681
Walker GC (1984) Mutagenesis and inducible responses to deoxyribonucleic acid damage in Escherichia coli. Microbiol Rev 48:60–93
Walker GC (1985) Inducible DNA repair systems. Annu Rev Biochem 54: 425–157
Walkup LK, Kogoma I (1989) Escherichia coli proteins inducible by oxidative stress mediated by the superoxide radical. J Bacteriol 171:1476–1484
Walsh DA, Klein NW, Hightower LE, Edwards MJ (1987) Heat shock and thermo-tolerance during early rat embryo development. Teratology 36:181–191
Welch WJ (1992) Mammalian stress response: cell physiology, structure/function of stress proteins, and implications for medicine and disease. Physiol Rev 72:1063–1081
Werner-Washburne M, Stone DE, Craig EA (1987) Complex interactions among members of an essential subfamily of hsp 70 genes in Saccharomyces cerevisiae. Mol Cell Biol 7:2568–2577
West SC, Howard-Flanders P (1984) Duplex-duplex interactions catalyzed by RecA protein allow strand exchange to pass double-strand breaks in DNA. Cell 37:683–691
West SC, Cassuto E, Howard-Flanders P (1981) Mechanism of E. coli RecA protein directed strand exchanges in post-replication repair of DNA. Nature 294:659–662
West SC, Cassuto E, Howard-Flanders P (1982) Postreplication repair in E. coli: strand exchange reactions of gapped DNA by RecA protein. Mol Gen Genet 187:209–217
White CN, High to wer LE, Schultz RJ (1994) Variation in heat-shock proteins among species of desert fishes (Poeciliidae, Poeciliopsis). Mol Biol Evol 11:106–119
Williams RS, Thomas JA, Fina M, German Z (1993) Human heat shock protein 70 (hsp 70) protects murine cells from injury during metabolic stress. J Clin Invest 92:503–508
Wittig S, Hensse S, Keitel C, Eisner C, Wittig B (1983) Heat shock gene expression is regulated during teratocarcinoma cell differentiation and early embryonic development. Dev Biol 96:507–514
Woloschak GE, Chang-Liu CM, Jones PS, Jones CA (1990) Modulation of gene expression in Syrian hamster embryo cells following ionizing radiation. Cancer Res 50:339–344
Yamamoto Y, Kataoka H, Nakabeppu Y, Tsuzuki T, Sekiguchi M (1983) The genes involved in the repair of alkylated DNA in Escherichia coli K-12. In: Friedberg EG, Bridges BA (eds) Cellular responses to DNA damage. Liss, New York, pp 271–278
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1997 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Mirkes, P.E. (1997). Cellular Responses to Stress. In: Kavlock, R.J., Daston, G.P. (eds) Drug Toxicity in Embryonic Development I. Handbook of Experimental Pharmacology, vol 124 / 1. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-60445-4_9
Download citation
DOI: https://doi.org/10.1007/978-3-642-60445-4_9
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-64408-5
Online ISBN: 978-3-642-60445-4
eBook Packages: Springer Book Archive