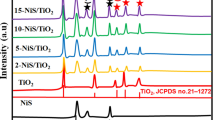
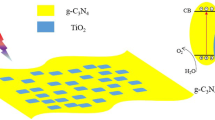
Abstract
The sheet-like NiS-ZnS heterostructured composites were prepared by the microwave hydrothermal. The fabrication of the heterojunctions broadened the range of light response of ZnS. The photoexcited electrons of ZnS migrated to the redox potential of NiS/Ni2S under visible light irradiation, which can facilitate the effective separation of photogenerated electrons and holes in space. The visible photocatalytic activity towards hydrogen production for NiS/ZnS nanosheet heterostructed composites without co-catalyst achieved 0.28 mmol h−1, and the quantum efficiency was up to 18.6% under the wavelength with 420 nm visible light irradiation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
R.D. Cortright, R.R. Davda, and J.A. Dumesic. “Hydrogen from catalytic reforming of biomass-derived hydrocarbons in liquid water,” Nature, 418(6901)(2002), 964–967.
Turner and A. John. “Sustainable hydrogen production,” Science, 305(5686) (2004), 972–974.
Kasuga, Tomoko, et al. “Formation of titanium oxide nanotube,” Langmuir, 14(12) (1998), 3160–3163.
Kudo, Akihiko, and Yugo Miseki. “Heterogeneous photocatalyst materials for water splitting,” Chemical Society Reviews, 38(1) (2009), 253–278.
Kim, Hyun Gyu, Dong Won Hwang, and Jae Sung Lee. “An undoped, single-phase oxide photocatalyst working under visible light,” Journal of the American Chemical Society, 126(29) (2004), 8912–8913.
J.G.. Yu, L.F. Qi, and Mietek Jaroniec. “Hydrogen production by photocatalytic water splitting over Pt/TiO2 nanosheets with exposed (001) facets,” The Journal of Physical Chemistry, 114(30) (2010), 13118–13125.
X.B. Chen, and Samuel S. Mao. “Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications,” Chemical reviews, 107(7) (2007), 2891–2959.
Linsebigler, L. Amy, G..Q. Lu, and T. John Yates Jr. “Photocatalysis on TiO2 surfaces: principles, mechanisms, and selected results.” Chemical reviews 95.3 (1995): 735–758.
J.G. Yu, J. Zhang and S.W. Liu. “Ion-exchange synthesis and enhanced visible-light photoactivity of CuS/ZnS nanocomposite hollow spheres.” The Journal of Physical Chemistry, 114(32) (2010), 13642–13649.
J.G. Yu, and J. Zhang. “A simple template-free approach to TiO2 hollow spheres with enhanced photocatalytic activity,” Dalton Transactions, 39(25) (2010), 5860–5867.
Matsumura, Michio, et al. “Cadmium sulfide photocatalyzed hydrogen production from aqueous solutions of sulfite: effect of crystal structure and preparation method of the catalyst,” The Journal of Physical Chemistry, 89(8) (1985), 1327–1329.
Reber, Jean Francois, and Milos Rusek. “Photochemical hydrogen production with platinized suspensions of cadmium sulfide and cadmium zinc sulfide modified by silver sulfide,” The Journal of Physical Chemis y C , 90(5) (1986), 824–834.
Frank, Arthur J., and Kenji Honda. “Visible-light-induced water cleavage and stabilization of n-type cadmium sulfide to photocorrosion with surface-attached polypyrrole-catalyst coating,” The Journal of Physical Chemistry C , 86(11)(1982), 1933–1935.
N.Z. Bao, et al. “Facile Cd-thiourea complex thermolysis synthesis of phase-controlled CdS nanocrystals for photocatalytic hydrogen production under visible light,” The Journal of Physical Chemistry C, 111(47) (2007), 17527–17534.
Maeda, Kazuhiko, and Kazunari Domen. “New non-oxide photocatalysts designed for overall water splitting under visible light,” The Journal of Physical Chemistry C, 111(22) (2007), 7851–7861.
Ohno, Tomoyuki, et al. “Photocatalytic water splitting using modified GaN: ZnO solid solution under visible light: long-time operation and regeneration of activity,” Journal of the American Chemical Society, 134(19) (2012), 8254–8259.
Kato, Hideki, Kiyotaka Asakura, and Akihiko Kudo. “Highly efficient water splitting into H2 and O2 over lanthanum-doped NaTaO3 photocatalysts with high crystallinity and surface nanostructure,” Journal of the American Chemical Society 125(10)(2003), 3082–3089.
J. Bandara, C. P. K. Udawatta, and C. S. K. Rajapakse. “Highly stable CuO incorporated TiO2 catalyst for photocatalytic hydrogen production from H2O,” Photochemical & Photobiological Sciences, 4(11) (2005), 857–861.
J.G. Yu, J. Zhang, and Mietek Jaroniec. “Preparation and enhanced visible-light photocatalytic H 2-production activity of CdS quantum dots-sensitized Zn 1- x Cd x S solid solution,” Green Chemistry 12(9) (2010), 1611–1614.
Innocenti, M., et al. “Ternary cadmium and zinc sulfides: composition, morphology and photoelectrochemistry,” Electrochimica acta, 49(8) (2004), 1327–1337.
Yoshimura, Joji, et al. “Visible Light Induced Hydrogen Evolution on CdS/K4Nb6O17 Photocatalyst,” Bulletin of the chemical society of Japan, 68(8) (1995), 2439–2445.
H. Zhang, and Y.F. Zhu. “Significant visible photoactivity and antiphotocorrosion performance of CdS photocatalysts after monolayer polyaniline hybridization,” The Journal of Physical Chemistry C, 114(13) (2010), 5822–5826.
Jang, Jum Suk, et al. “Role of platinum-like tungsten carbide as cocatalyst of CdS photocatalyst for hydrogen production under visible light irradiation,” Applied Catalysis A: General, 346(1)(2008), 149–154.
Bataille, Frédéric, et al. “Alkyldibenzothiophenes hydrodesulfurization-promoter effect, reactivity, and reaction mechanism,” Journal of catalysis,191(2)(2000), 409–422.
Chianelli, Russell R., et al. “Catalytic properties of single layers of transition metal sulfide catalytic materials,” Catalysis Reviews , 48(1) (2006), 1–41.
Moses, Poul Georg, et al. “Corrigendum to The hydrogenation and direct desulfurization reaction pathway in thiophene hydrodesulfurization over MoS2 catalysts at realistic conditions: A density functional study,” Journal of Catalysis, 260(1)(2008), 202–203.
Y.P. Bi, et al. “Facile synthesis of rhombic dodecahedral AgX/Ag 3 PO 4 (X= Cl, Br, I) heterocrystals with enhanced photocatalytic properties and stabilities,” Physical Chemistry Chemical Physics, 13(21) (2011), 10071–10075.
Z. Xu, et al. “Photocatalytic H2 evolution on MoS2/CdS catalysts under visible light irradiation,” The Journal of Physical Chemistry C, 114(4) (2010), 1963–1968.
H. J. Yan, J. H. Yang, G. J. Ma, G. P. Wu, X. Zong, Z. B. Lei, J. Y. Shi, C. Li, Visible-lightdriven hydrogen production with extremely high quantum efficiency on Pt–PdS/CdS photocatalyst. Journal of catalysis, 266(2009), 165–168.
Reber, Jean Francois, and Milos Rusek. “Photochemical hydrogen production with platinized suspensions of cadmium sulfide and cadmium zinc sulfide modified by silver sulfide,” The Journal of Physical Chemistry , 90(5) (1986), 824–834.
S.H. Shen, et al. “Effect of Ag2 S on solar-driven photocatalytic hydrogen evolution of nanostructured CdS,” international journal of hydrogen energy, 35(13) (2010), 7110–7115.
H.P. Liu, et al. “SrS/CdS composite powder as a novel photocatalyst for hydrogen production under visible light irradiation,” International Journal of Hydrogen Energy, 35(13) (2010), 7080–7086.
J. Zhang, J. G. Yu, M. Jaroniec. “Noble metal-free reduced graphene oxide-ZnxCd1-xS nanocomposite with enhanced solar photocatalytic H2-production performance,”. Nano Letter., 12 (2012),: 4584–4589.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 TMS (The Minerals, Metals & Materials Society)
About this chapter
Cite this chapter
Li, L., Chen, J., Hwang, JY., Liu, J., Zhou, Y., Lu, L. (2015). Photocatalytic H2 Production under Visible Light Irradiation on Novel Heterostructure Nis/Zns Nanosheet Photocatalyst. In: Carpenter, J.S., et al. Characterization of Minerals, Metals, and Materials 2015. Springer, Cham. https://doi.org/10.1007/978-3-319-48191-3_98
Download citation
DOI: https://doi.org/10.1007/978-3-319-48191-3_98
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48601-7
Online ISBN: 978-3-319-48191-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)