Abstract
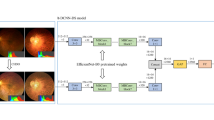
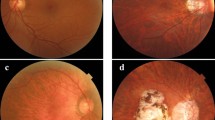
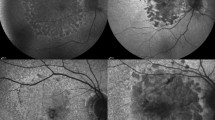
Pathologic myopia (PM) is a sight-threatening disease characterised by abnormal ocular changes due to excessive axial elongation in myopes. One important clinical manifestation of PM is myopic maculopathy (MM), which is categorised into 5 ordinal classes based on the established META-PM classification framework. This paper details a robust deep learning approach to automatically classifying MM from colour fundus photographs as part of the recently held Myopic Maculopathy Analysis Challenge (MMAC). A ResNet-18 model pretrained on ImageNet-1K was trained for the task. Pertinent MM lesions (patchy or macular atrophy) were manually segmented in images from the MMAC dataset and another publicly available dataset (PALM) to create a collection of lesion masks based on which an additional 250 images with severe MM were synthesised to mitigate class imbalance in the original training set. The image synthesis pipeline was guided by clinical domain knowledge: (1) synthesised macular atrophy tended to be circular with a regressed fibrovascular membrane near its centre, while patchy atrophy was more irregular and varied more greatly in size; (2) synthesised images were created using images with diffuse or patchy atrophy as background; and (3) synthesised images included examples that were not easily classifiable (e.g. creating patchy lesions that were in close proximity to the fovea). This, coupled with mix-up augmentation and ensemble prediction via test-time augmentation, enabled the model to rank first in the validation phase and fifth in the test phase. The source code is freely available at https://github.com/fyii200/MyopicMaculopathyClassification.
F. Yii is supported by the Medical Research Council [grant number MR/N013166/1]. The funder had no role in the design and conduct of this work nor the decision to submit this manuscript for publication.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jonas, J.B., Spaide, R.F., Ostrin, L.A., Logan, N.S., Flitcroft, I., Panda-Jonas, S.: IMI-nonpathological human ocular tissue changes with axial myopia. Invest. Ophthalmol. Vis. Sci. 64, 5 (2023)
Ohno-Matsui, K., et al.: IMI pathologic myopia. Invest. Ophthalmol. Vis. Sci. 62, 5 (2021)
Ohno-Matsui, K., et al.: International photographic classification and grading system for myopic maculopathy. Am. J. Ophthalmol. 159, 877-883.e877 (2015)
Li, J., et al.: Automated detection of myopic maculopathy from color fundus photographs using deep convolutional neural networks. Eye Vis. (Lond) 9, 13 (2022)
Rauf, N., Gilani, S.O., Waris, A.: Automatic detection of pathological myopia using machine learning. Sci. Rep. 11, 16570 (2021)
Hemelings, R., Elen, B., Blaschko, M.B., Jacob, J., Stalmans, I., De Boever, P.: Pathological myopia classification with simultaneous lesion segmentation using deep learning. Comput. Methods Programs Biomed. 199, 105920 (2021)
Cui, J., Zhang, X., **ong, F., Chen, C.L.: Pathological myopia image recognition strategy based on data augmentation and model fusion. J. Healthc. Eng. 2021, 5549779 (2021)
Tan, T.E., et al.: Retinal photograph-based deep learning algorithms for myopia and a blockchain platform to facilitate artificial intelligence medical research: a retrospective multicohort study. Lancet Digit. Health 3, e317–e329 (2021)
Himami, Z.R., Bustamam, A., Anki, P.: Deep learning in image classification using dense networks and residual networks for pathologic myopia detection. In: 2021 International Conference on Artificial Intelligence and Big Data Analytics, pp. 1–6. (2021)
Dai, S., Chen, L., Lei, T., Zhou, C., Wen, Y.: Automatic detection of pathological myopia and high myopia on fundus images. In: 2020 IEEE International Conference on Multimedia and Expo (ICME), pp. 1–6. (2020)
Ali, S., Raut, S.: Detection of pathological myopia from fundus images. In: Sharma, H., Shrivastava, V., Bharti, K.K., Wang, L. (eds.) Communication and Intelligent Systems. Lecture Notes in Networks and Systems, vol. 686, pp. 201-208. Springer, Singapore (2023). https://doi.org/10.1007/978-981-99-2100-3_17
Park, S.J., Ko, T., Park, C.K., Kim, Y.C., Choi, I.Y.: Deep learning model based on 3D optical coherence tomography images for the automated detection of pathologic myopia. Diagnostics (Basel) 12, 742 (2022)
Lu, L., et al.: Development of deep learning-based detecting systems for pathologic myopia using retinal fundus images. Commun. Biol. 4, 1225 (2021)
Du, R., et al.: Deep learning approach for automated detection of myopic maculopathy and pathologic myopia in fundus images. Ophthalmol. Retina 5, 1235–1244 (2021)
Lu, L., et al.: AI-model for identifying pathologic myopia based on deep learning algorithms of myopic maculopathy classification and “plus” lesion detection in fundus images. Front. Cell Dev. Biol. 9, 719262 (2021)
Tang, J., et al.: An artificial-intelligence-based automated grading and lesions segmentation system for myopic maculopathy based on color fundus photographs. Transl. Vis. Sci. Technol. 11, 16 (2022)
Fang, H., et al.: PALM: open fundus photograph dataset with pathologic myopia recognition and anatomical structure annotation (2023). ar**v:2305.07816
Itoi, M., Hieda, O., Kusada, N., Miyatani, T., Kojima, K., Sotozono, C.: Progression of myopic maculopathy: a systematic review and meta-analysis. Eye Contact Lens 49, 83–87 (2023)
Zhang, H., Cisse, M., Dauphin, Y.N., Lopez-Paz, D.: mixup: beyond empirical risk minimization (2017). ar**v:1710.09412
Chang, O., Tran, D.N., Koishida, K.: Single-channel speech enhancement using learnable loss mixup. In: Interspeech, pp. 2696–2700 (2021)
Czodrowski, P.: Count on kappa. J. Comput. Aided Mol. Des. 28(11), 1049–1055 (2014). https://doi.org/10.1007/s10822-014-9759-6
Cohen, J.: A coefficient of agreement for nominal scales. Educ. Psychol. Measur. 20, 37–46 (1960)
Zhao, X., et al.: Morphological characteristics and visual acuity of highly myopic eyes with different severities of myopic maculopathy. Retina 40, 461–467 (2020)
Szegedy, C., Vanhoucke, V., Ioffe, S., Shlens, J., Wojna, Z.: Rethinking the inception architecture for computer vision (2015). ar**v:1512.00567
Perez, F., Vasconcelos, C., Avila, S., Valle, E.: Data augmentation for skin lesion analysis (2018). ar**v:1809.01442
Matsunaga, K., Hamada, A., Minagawa, A., Koga, H.: Image classification of melanoma, nevus and seborrheic keratosis by deep neural network ensemble (2017). ar**v:1703.03108
Chua, S.Y.L., et al.: Cohort profile: design and methods in the eye and vision consortium of UK Biobank. BMJ Open 9, e025077 (2019)
Wosiak, A., Glinka, K., Zakrzewska, D.: Multi-label classification methods for improving comorbidities identification. Comput. Biol. Med. 100, 279–288 (2018)
Weng, W., Lin, Y., Wu, S., Li, Y., Kang, Y.: Multi-label learning based on label-specific features and local pairwise label correlation. Neurocomputing 273, 385–394 (2018)
Mosqueira-Rey, E., Hernández-Pereira, E., Alonso-Ríos, D., Bobes-Bascarán, J., Fernández-Leal, Á.: Human-in-the-loop machine learning: a state of the art. Artif. Intell. Rev. 56, 3005–3054 (2023)
Wang, Z., et al.: Generative adversarial networks in ophthalmology: what are these and how can they be used? Curr. Opin. Ophthalmol. 32, 459–467 (2021)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Yii, F. (2024). A Clinically Guided Approach for Training Deep Neural Networks for Myopic Maculopathy Classification. In: Sheng, B., Chen, H., Wong, T.Y. (eds) Myopic Maculopathy Analysis. MICCAI 2023. Lecture Notes in Computer Science, vol 14563. Springer, Cham. https://doi.org/10.1007/978-3-031-54857-4_8
Download citation
DOI: https://doi.org/10.1007/978-3-031-54857-4_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-54856-7
Online ISBN: 978-3-031-54857-4
eBook Packages: Computer ScienceComputer Science (R0)