Abstract
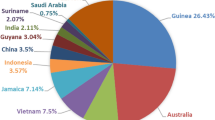
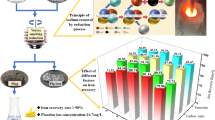
In order to realize the large-scale resourcefulness and harmless utilization of high-iron red mud, a pilot-scale experiment on vortex smelting reduction of high-iron red mud was conducted using conditions determined on the basis of laboratory research. The pilot study achieved good results. The iron recovery was 97.12%, and a pig iron product containing 93.27 wt % iron was obtained. The experimental conditions included a temperature of 1500 °C, a carbon ratio of 1.3, a holding time of 30 min, a stirring speed of 125 rpm, an alkalinity of 1.0, and fluorite was used as a co-solvent at 10% of the calcium oxide addition. The concentrations of P and S in the pig iron were low, which allowed them to meet the requirements of the national standard L03 for steelmaking. The main phases in the reduction slag were gehlenite, calcium titanate, and nepheline, which were effective components for cement clinker production.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Liu W C, Chen X Q, Li W X, et al. (2014) Environmental assessment management and utilization of red mud in China. Journal of Cleaner Production. 84: 606–610.
Khairul M A, Zanganeh J, Moghtaderi, B. (2019) The composition, recycling and utilization of Bayer red mud. Resources, Conservation & Recycling. 141: 483–498.
Luo D, Li Z L, Du Q, et al. (2020) Research progress on comprehensive utilization of red mud. Technology Innovation and Application. (15): 75–76.
Klauber C, Gräfe M, Power G. (2011) Bauxite residue issues: II. Options for residue utilization. Hydrometallurgy. 108:11–32.
Grafe M, Power G, Klauber C. (2011) Bauxite residue issues: III. Alkalinity and associated chemistry. Hydrometallurgy. 108: 60–79.
Li X F, Zhang T A, Wang K, et al. (2022) Experimental research on Vortex Melting Reduction of High-iron Red Mud (Bauxite residue). Bulletin of Environmental Contamination and Toxicology. 109:155–162.
Yan W Q. (2016) Domestic and international tailings storage stockpileings and applications. Metal Mine. 1(9): 17–21.
Nan X L, Zhang T A, Liu Y, et al. (2009) Main Categories of Red Mud and Its Environmental Impacts. The Chinese Journal of Process Engineering. (S1): 459–464.
Wu S C, Zhu L X, Sun T C, et al. (2019) Red mud comprehensive utilization status and outlook[J]. Metal Mine. (6): 38–44.
Ruyters S, Mertens J, Vassilieva E. (2011) The red mud accident in Ajka (Hungary): plant toxicity and trace metal bioavailability in red mud contaminated soil. Environmental Science & Technology. 45(4): 1616–1622.
Yang P, Wu W N. (2017) Analysis of the emergency response to the Hungarian red mud spill and implications for China. Technology and Innovation Herald. 14(9): 163–164.
Wang K, Dou Z H, Liu Y, et al. (2022) Summary of research progress on separation and extraction of valuable metals from Bayer red mud. Environmental Science and Pollution Research. 29: 89834–89852.
Agrawal S, Dhawan N. (2021) Evaluation of red mud as a polymetallic source – A review. Minerals Engineering. 171: 107084.
Gu N H, Guo T F, Ma S C, et al. (2018) Progress in the extraction and recycling of iron from red mud. Chemical Industry and Engineering Progress. (9): 3599–3608.
Liu Z, Li H. (2015) Metallurgical process for valuable elements recovery from red mud: a review. Hydrometallurgy. 155: 29–43.
Sadangi J K, Das S P, Tripathy A, et al. (2018) Investigation into recovery of iron values from red mud dumps. Separation Science and Technology. 53: 1–6.
Hu W, **ong D H. (2010) On Iron Ore Recovery out of Mineral Processing Tailing and Smelting Tailing by Magnetic-gravity Process. Nonferrous Metals Science and Engineering. 1(3): 52–54.
Guan J H. (2000) Study on recovering Fe from red mud with SLon vertical ring and pulsating high gradient magnetic separator. Jiangxi Nonferrous Metals. 14(4): 15–18.
Zhang R, Zheng S, Ma S, et al. (2011) Recovery of alumina and alkali in Bayer red mud by the formation of andradite-grossular hydrogarnet in hydrothermal process. Journal of Hazardous Materials. 189(3): 827–835.
Yang Y, Wang X, Wang M, et al. (2015) Recovery of iron from red mud by selective leach with oxalic acid. Hydrometallurgy. 157: 239–245.
Samouhos M, Taxiarchou M, Pilatos G et al. (2017) Controlled reduction of red mud by H2 followed by magnetic separation. Minerals Engineering. 105: 36–43.
Gostu S, Mishra B, Martins G P. (2017) Low temperature reduction of hematite in red mud to magnetite. The Minerals, Metals & Materials Society. 67–73.
Wang J S, Liu J, Xue Q G. (2013) Experimental research on comprehensive utilization of the high iron red mud based on direct reduction and melting by RHF iron bead technology [J]. Light metals. 1:19–22.
Liu Y T, Qin Z H, Chen B. (2020) Experimental research on magnesium phosphate cements modified by red mud. Construction and Building Materials. 231:117–131.
Zhang T A, Wang Y X, Dou Z H, et al. (2019) A method for direct cementation of high-iron red mud by eddy current melting reduction alkali removal and iron extraction. China Patent: ZL201910290643.7, 2019.
Li X F, Zhang T A, Wang K, et al. (2022) Research on the Advantages of Vortex Smelting Reduction of High-Iron Red Mud (Bauxite Residue). Russian Journal of Non-Ferrous Metals. 63(6): 731–737.
Wang K, Liu Y, Dou Z H, et al. (2022) A Novel Method of Extracting Iron from High-Iron Red Mud and Preparing Low-Carbon Cement Clinker from Tailings. JOM. 74(7):2750–2759.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (52204419), the National Natural Science Foundation of Liaoning Province (2022-BS-076), and the Guangxi Science and Technology Major Project (2021AA12013).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Minerals, Metals & Materials Society
About this paper
Cite this paper
Li, X., Zhang, Ta., Lv, G., Wang, K. (2024). Pilot Study on the Recovery of Iron from High-Iron Red Mud by Vortex Smelting Reduction. In: Wagstaff, S. (eds) Light Metals 2024. TMS 2024. The Minerals, Metals & Materials Series. Springer, Cham. https://doi.org/10.1007/978-3-031-50308-5_15
Download citation
DOI: https://doi.org/10.1007/978-3-031-50308-5_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-50307-8
Online ISBN: 978-3-031-50308-5
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)