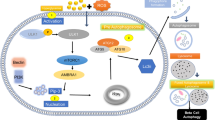
Abstract
β-cells represent the functional unit of pancreatic islets, and they are responsible for glucose homeostasis regulation. β-cells possess the ability to modify insulin secretion according to the organism-specific needs. Thus, during physiological changes such as pregnancy or obesity, glycemia is increased concomitantly with the ability of β-cells to secrete insulin. However, when demand for insulin chronically increases, a steady stimulation of β-cells eventually may lead to death. In spite of the conducted efforts in order to elucidate the glucotoxicity mechanisms acting on β-cells, they remain largely unknown. Hyperglycemia promotes several metabolic alterations such as glucolipotoxicity, mitochondrial alterations, oxidative stress, endoplasmic reticulum stress, amyloid polypeptide accumulation, and proinflammatory cytokine accumulation. The latter are commonly engaged during apoptosis triggering in β-cells. In recent years, p53 has been also proposed as a major trigger of apoptosis in β-cells during hyperglycemia conditions. Because insulin-producing cells are cultured using high glucose levels, the presence of p53 in mitochondria induces apoptosis. The insight on the mechanisms triggering cell death in pancreatic β-cells will support the proposal of alternatives for prevention and/or cell protection also contributing to the treatment of diabetic patients.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- AGE:
-
Advanced glycation end products
- AIF:
-
Apoptosis inducing factor
- Apaf-1:
-
Apoptotic protease-activating factor 1
- ATF6:
-
Activating transcription factor 6
- ATM:
-
ATM serine/threonine kinase protein
- Bak:
-
Bcl-2 homologous antagonist killer
- Bax:
-
Bcl-2-associated X protein
- Bcl-2:
-
B-cell lymphoma 2
- Bcl-xl:
-
B-cell lymphoma-extra large
- BH (1–4):
-
Bcl-2 homology domains
- Bok:
-
Bcl-2 related ovarian killer
- Caspasa:
-
Cysteine-aspartic proteases, cysteine aspartases
- CHOP:
-
C/EBP homologous protein
- ChREBP:
-
Carbohydrate response element-binding protein
- Drp1:
-
Dynamin-related protein 1
- ΔΨm:
-
Mitochondrial membrane potential
- eif2α:
-
Eukaryotic translation initiation factor 2α
- ER:
-
Reticulum stress
- ERE:
-
Endoplasmic reticulum stress
- EZH2:
-
Enhancer of Zeste Homologue 2
- FADD:
-
FAS-associating death domain-containing protein
- Fas:
-
Death receptor
- FFA:
-
Free fatty acids
- Fis1:
-
Mitochondrial fission 1 protein
- FOX A1/2:
-
Forkhead box
- G3P:
-
Glyceraldehyde 3-phosphate
- GADD34:
-
Downstream growth arrest and DNA damage-inducible protein
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- GATA4/6:
-
GATA-binding protein
- GLUT:
-
Glucose transporter
- GSIS:
-
Glucose-stimulated insulin secretion
- H3K27me3:
-
Histone H3 trimethyl K27
- HNF1β:
-
Hepatocyte nuclear factor
- IAPP:
-
Islet amyloid polypeptide
- IFNγ:
-
Interferon gamma
- IGF1:
-
Insulin-like growth factor 1
- IL-1β:
-
Interleukin 1 beta
- iNOS:
-
Nitric oxide synthases inducible
- Ins :
-
Insulin gene
- INS1:
-
Insulin secreting beta-cell-derived line
- IRE1α:
-
Inositol-requiring enzyme
- IRS-2:
-
Insulin receptor substrate
- Isl:
-
Islet
- MafA:
-
Musculo aponeurotic fibrosarcoma protein A
- Mdm2:
-
Murine double minute 2
- Mff:
-
Mitochondrial fission factor
- Mfn:
-
Mitofusin
- Mouse db/db:
-
Model of obesity, diabetes, and dyslipidemia with a mutation in leptin receptor
- mTOR:
-
Mammalian target of rapamycin
- NAD+:
-
Nicotinamide adenine dinucleotide
- NADH:
-
Nicotinamide adenine dinucleotide reduced
- NADPH oxi:
-
Nicotinamide adenine dinucleotide phosphate-oxidase
- NeuroD1:
-
Neurogenic differentiation 1
- NF-κB:
-
Nuclear factor kappa B
- Nkx:
-
Homeobox protein
- NLRP3:
-
NACHT, LRR, and PYD domain-containing protein 3
- NLRs:
-
Nucleotide oligomerization domain (NOD)-like receptors
- NO:
-
Nitric oxide
- NOD :
-
Nucleotide oligomerization domain
- Notch:
-
Transcription factor
- •OH:
-
Hydroxyl radical
- 8-OHdG:
-
8-hydroxy-2′-deoxyguanosine
- O−2:
-
Superoxide anion
- O-GlcNAc:
-
O-linked β-N-acetylglucosamine
- Opa1:
-
Protein of mitocondrial internal membrane
- P/CAF:
-
P300/CBP-associated factor
- p16:
-
Cyclin-dependent kinase inhibitor 2A, multiple tumor suppressor 1
- p21:
-
Cyclin-dependent kinase inhibitor 1 or CDK-interacting protein 1
- p27:
-
Cyclin-dependent kinase inhibitor 1B
- p300/CBP:
-
E1A binding protein p300/CREB-binding protein
- p38 MAPK:
-
P38 mitogen-activated protein kinases
- p53:
-
Tumor protein p53
- PARP:
-
Poly ADP ribose polymerase
- Pax4:
-
Transcription factors paired box gene 4
- Pdx1:
-
Pancreatic and duodenal homeobox 1
- PERK:
-
Protein kinase-like ER kinase
- PI3k:
-
Phosphatidylinositol-3-kinases
- PKC:
-
Protein kinase C
- PP-1:
-
Protein phosphatase 1
- Ptf1α:
-
Pancreas transcription factor 1α
- RAMP1:
-
Receptor activity-modifying protein 1
- Rfx 6:
-
Regulatory factor x
- RING-finger:
-
Really interesting new gene
- RINm5F:
-
Rat insulinoma cells
- ROS:
-
Reactive oxygen species
- Sox9 SRY:
-
Sex-determining region Y-box 9
- SPT:
-
Serine C-palmitoyltransferase
- T2D:
-
Type 2 diabetes
- TLRs:
-
Toll-like receptors
- TNFRI:
-
Tumor necrosis factor receptor type I
- TNFα:
-
Tumor necrosis factor alpha
- TXNIP:
-
Thioredoxin-interacting protein
- UCP2:
-
Uncoupling protein 2
- UDP-GlcNAc:
-
Uridine diphosphate N-acetylglucosamine
- UPR:
-
Unfolded protein response
References
Assmann A, Hinault C, Kulkarni RN. Growth factor control of pancreatic islet regeneration and function. Pediatr Diabetes. 2009;10:14–32.
Hudish LI, Reusch JE, Sussel L. β Cell dysfunction during progression of metabolic syndrome to type 2 diabetes. J Clin Invest. 2019;129:4001–8.
Ortega-Camarillo C, Guzmán-Grenfell AM, García-Macedo R, et al. Hyperglycemia induces apoptosis and p53 mobilization to mitochondria in RINm5F cells. Mol Cell Biochem. 2006;281:163–70.
Hinault C, Kawamori D, Liew CW, et al. D40 isoform of p53 controls b-cell proliferation and glucose homeostasis in mice. Diabetes. 2011;60:1210–22.
Jurczyk A, Bortell R, Alonso LC. Human β-cell regeneration: progress, hurdles, and controversy. Curr Opin Endocrinol Diabetes Obes. 2014;21:102–8.
Yagihashi S, Inaba W, Mizukami H. Dynamic pathology of islet endocrine cells in type 2 diabetes: β-cell growth, death, regeneration and their clinical implications. J Clin Invest. 2016;7:155–65.
Oliver-Krasinski JM, Stoffers DA. On the origin of the β cell. Genes Dev. 2008;22:1998–2021.
Gittes GK, Rutter WJ. Onset of cell-specific gene expression in the develo** mouse pancreas. Proc Natl Acad Sci U S A. 1992;89:1128–32.
Meier JJ, Butler AE, Saisho Y, et al. β-cell replication is the primary mechanism subserving the postnatal expansion of β-cell mass in humans. Diabetes. 2008;57:1584–94.
Mizukami H, Takahashi K, Inaba W, et al. Age-associated changes of islet endocrine cells and the effects of body mass index in Japanese. J Diabetes Investig. 2014;5:38–47.
Alejandro EU, Gregg B, Blandino-Rosano M, Cras-Méneur C, Bernal-Mizrachi E. Natural history of β-cell adaptation and failure in type 2 diabetes. Mol Aspects Med. 2015;42:19–41.
Butler AE, Janson J, Bonner-Weir S. β-Cell deficit and increased β-cell apoptosis in humans with type 2 diabetes. Diabetes. 2003;52:102–10.
Khadra A, Schnell S. Development, growth and maintenance of β-cell mass: models are also part of the story. Mol Aspects Med. 2015;42:78–90.
Ward W, Bolgiano D, McKnight B, Halter J, Porte DJ. Diminished β cell secretory capacity in patients with noninsulin-dependent diabetes mellitus. J Clin Invest. 1984;74:1318–28.
Meier JJ. Beta cell mass in diabetes: a realistic therapeutic target? Diabetologia. 2008;51:703–13.
Unger RH. Minireview: weapons of lean body mass destruction: the role of ectopic lipids in the metabolic syndrome. Endocrinology. 2003;144:5159–65.
Kjørholt C, Åkerfeldt MC, Biden TJ, Laybutt DR. Chronic hyperglycemia, independent of plasma lipid levels, is sufficient for the loss of β-cell differentiation and secretory function in the db/db mouse model of diabetes. Diabetes. 2005;54:2755–63.
Thornberry AN, Lazebnik Y. Caspases: enemies within. Science. 1998;281:1312–6.
Schwartzman RA, Cidlowski JA. Apoptosis: the biochemistry and molecular biology of programmed cell death. Endocr Rev. 1993;14:133–51.
Höppener JW, Ahrén B, Lips CJ. Islet amyloid and type 2 diabetes mellitus. N Engl J Med. 2000;343:411–9.
Tomita T. Apoptosis in pancreatic β-islet cells in type 2 diabetes. Bosn J Basic Med Sci. 2016;16:162–79.
Kilpatrick ED, Robertson RP. Differentiation between glucose-induced desensitization of insulin secretion and β-cell exhaustion in the HIT-T15 cell line. Diabetes. 1998;47:606–11.
Robertoson RP. β-cell deterioration during diabetes: what’s in the gun? Trends Endocrinol Metab. 2009;20:388–93.
Kajimoto Y, Watada H, Matsuoka T-a, et al. Suppression of transcription factor PDX-1/IPF1/STF-1/IDX-1 causes no decrease in insulin mRNA in MIN6 cells. J Clin Invest. 1997;100:1840–6.
Federici M, Hribal M, Perego L, et al. High glucose causes apoptosis in cultured human pancreatic islets of Langerhans. A potential role for regulation of specific Bcl family genes toward an apoptotic cell death program. Diabetes. 2001;50:1290–300.
Donath MY, Gross DJ, Cerasi E, Kaiser N. Hyperglycemia-induced -cell apoptosis in pancreatic islets of Psammomys obesus during development of diabetes. Diabetes. 1999;48:738–44.
Kim W-H, Lee JW, Suh YH, et al. Exposure to chronic high glucose induces b-cell apoptosis through decreased interaction of glucokinase with mitochondria. Diabetes. 2005;54:2602–11.
Piro S, Anello M, Pietro CD, et al. Chronic exposure to free fatty acids or high glucose induces apoptosis in rat pancreatic islets: possible role of oxidative stress. Metabolism. 2002;51:1340–7.
Chan CB, Saleh MC, Koshkin V, Wheeler MB. Uncoupling protein 2 and islet function. Diabetes. 2004;53:S136–42.
Joseph JW, Koshkin V, Saleh MC, et al. Free fatty acid-induced -cell defects are dependent on uncoupling protein 2 expression. J Biol Chem. 2004;279:51049–56.
Laybutt DR, Preston AM, Åkerfeldt MC, et al. Endoplasmic reticulum stress contributes to beta cell apoptosis in type 2 diabetes. Diabetologia. 2007;50:752–63.
Ogihara T, Mirmira RG. An islet in distress: β cell failure in type 2 diabetes. J Diabetes Investig. 2010;1:123–33.
Kelpe CL, Moore PC, Parazzoli SD, et al. Palmitate inhibition of insulin gene expression is mediated at the transcriptional level via ceramide synthesis. J Biol Chem. 2003;278:30015–21.
Stein DT, Esser V, Stevenson BE, et al. Essentiality of circulating fatty acids for glucose-stimulated insulin secretion in the fasted rat. J Clin Invest. 1996;97:2728–35.
Véret J, Coant N, Berdyshev EV, et al. Ceramide synthase 4 and de novo production of ceramides with specific N-acyl chain lengths are involved in glucolipotoxicity-induced apoptosis of INS-1 β-cells. Biochem J. 2011;438:177–89.
Galadari S, Rahman A, Pallichankandy S, Galadari A, Thayyullathil F. Role of ceramide in diabetes mellitus: evidence and mechanisms. Lipids Health Dis. 2013;12:98–114.
Unger RH, Zhou Y-T. Lipotoxicity of β-cells in obesity and in other causes of fatty acid spillover. Diabetes. 2001;50:S118–21.
Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001;414:813–20.
Oliveira HR, Verlengia R, Carvalho CRO, et al. Pancreatic β-cells express phagocyte-like NAD(P)H oxidase. Diabetes. 2003;52:1457–63.
Yan L-J. Pathogenesis of chronic hyperglycemia: from reductive stress to oxidative stress. J Diabetes Res. 2014;2014:137919.
Grankvist K, Marklund SL, Täljedal I-B. CuZn-superoxide dismutase, Mn-superoxide dismutase, catalase and glutathione peroxidase in pancreatic islets and other tissues in the mouse. Biochem J. 1981;199:393–8.
Brownlee M. The pathology of diabetic complications: a unifying mechanism. Diabetes. 2005;54:1615–25.
Nishikawa T, Edelstein D, Du JX, et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature. 2000;404:787–90.
Maechler P, Wollheim CB. Mitochondrial function in normal and diabetic b-cells. Nature. 2001;414:807–12.
Orrenius S, Zhivotovsky B. Cardiolipin oxidation sets cytochrome c free. Nat Chem Biol. 2005;1:188–9.
Ma ZA, Zhao Z, Turk J. Mitochondrial dysfunction and β-cell failure in type 2 diabetes mellitus. Exp Diabetes Res. 2012;2012:1–11.
Kaufman BA, Li C, Soleimanpour SA. Mitochondrial regulation of β-cell function: maintaining the momentum for insulin release. Mol Aspects Med. 2015;2015:91–104.
Yoon Y, Krueger EW, Oswald BJ, McNiven MA. The mitochondrial protein hFis1 regulates mitochondrial fission in mammalian cells through an interaction with the dynamin-like protein DLP1. Mol Cell Biol. 2003;23:5409–20.
Maassen JA, Romijn JA, Heine RJ. Fatty acid-induced mitochondrial uncoupling in adipocytes as a key protective factor against insulin resistance and beta cell dysfunction: a new concept in the pathogenesis of obesity-associated type 2 diabetes mellitus. Diabetologia. 2007;50:2036–41.
Hasnain SZ, Prins JB, McGuckin MA. Oxidative and endoplasmic reticulum stress in b-cell dysfunction in diabetes. J Mol Endocrinol. 2016;56:R33–54.
Karunakaran U, Kim H-J, Kim J-Y, Lee I-K. Guards and culprits in the endoplasmic reticulum: glucolipotoxicity and β-cell failure in type II diabetes. Exp Diabetes Res. 2012;2012:9.
Sharma RB, Alonso LC. Lipotoxicity in the pancreatic beta cell: not just survival and function, but proliferation as well? Curr Diab Rep. 2014;14:492–508.
Cui W, Ma J, Wang X, et al. Free fatty acid induces endoplasmic reticulum stress and apoptosis of β-cells by Ca2+/Calpain-2 pathways. PLoS One. 2013;8:e59921.
Meigs JB, Wilson PWF, Fox CS, et al. Body mass index, metabolic syndrome, and risk of type 2 diabetes or cardiovascular disease. J Clin Endocrinol Metab. 2006;91:2906–12.
Meier JJ, Bonadonna RC. Role of reduced B-cell function in the pathogenesis of type 2 diabetes. Diabetes Care. 2013;36:S13–9.
Cantley J, Ashcroft FM. Q&A: insulin secretion and type 2 diabetes: why do β-cells fail? BMC Biol. 2015;13:33–40.
Saisho Y, Butler AE, Manesso E, et al. β-cell mass and turnover in humans: effects of obesity and aging. Diabetes Care. 2013;36:111–7.
Hull RL, Kodama K, Utzschneider KM, et al. Dietary-fat induced obesity in mice results in beta cell hyperplasia but not increased insulin release: evidence for specificity of impaired beta cell adaptation. Diabetologia. 2005;48:1350–8.
Cheng Z, Almeida FA. Mitochondrial alteration in type 2 diabetes and obesity. Cell Cycle. 2014;13:890–7.
Itariu BK, Stulnig TM. Autoimmune aspects of type 2 diabetes mellitus – a mini-review. Gerontology. 2014;60:189–96.
Araki E, Oyadomari S, Mori M. Impact of endoplasmic reticulum stress pathway on pancreatic b-cells and diabetes mellitus. Exp Biol Med. 2003;228:1213–7.
Chen J, Saxena G, Mungrue IN, Lusis AJ, Shalev A. Thioredoxin-Interacting protein: a critical link between glucose toxicity and beta cell apoptosis. Diabetes. 2008;57:938–44.
Teff KL, Elliott SS, Tschöp M, et al. Dietary fructose reduces circulating insulin and leptin, attenuates postprandial suppression of ghrelin, and increases triglycerides in women. J Clin Endocrinol Metab. 2004;89:2963–72.
Lowell B, Shulman I. Mitochondrial dysfunction and type 2 diabetes. Science. 2005;307:384–7.
Ritzel RA, Meier JJ, Lin C-Y, Veldhuis JD, Butler PC. Human islet amyloid polypeptide oligomers disrupt cell coupling, induce apoptosis, and impair insulin secretion in isolated human islets. Diabetes. 2007;56:65–71.
Arnelo U, Permert J, Larsson J, et al. Chronic low dose islet amyloid polypeptide infusion reduces food intake, but does not influence glucose metabolism, in unrestrained conscious rats: studies using a novel aortic catheterization technique. Endocrinology. 1997;138:4081–5.
Hull RL, Westermark GT, Westermark P, Kahn SE. Islet Amyloid: a critical entity in the pathogenesis of type 2 diabetes. J Clin Endocrinol Metab. 2004;89:629–3643.
Mihara M, Erster S, Zaika A, et al. p53 has a direct apoptogenic role at the mitochondria. Mol Cell. 2003;11:577–90.
Ortega-Camarillo C, Flores-López LA, Ávalos-Rodríguez A. The role of p53 in pancreatic b-cell apoptosis. Immunoendocrinology. 2015;2:E1075.
Balmanno K, Cook SJ. Tumour cell survival signalling by the ERK1/2 pathway. Cell Death Differ. 2009;16:368–77.
Elkholi R, Chipuk JE. How do I kill thee? Let me count the ways: p53 regulates PARP-1 dependent necrosis. Bioessays. 2014;36:46–51.
Lavin MF, Gueven N. The complexity of p53 stabilization and activation. Cell Death Differ. 2006;13:941–50.
Flores-López LA, Díaz-Flores M, García-Macedo R, et al. High glucose induces mitochondrial p53 phosphorylation by p38 MAPK in pancreatic RINm5F cells. Mol Biol Rep. 2013;40:4947–58.
Yang WH, Kim JE, Nam HW, et al. Modification of p53 with O-linked N-acetylglucosamine regulates p53 activity and stability. Nat Cell Biol. 2006;8:1074–83.
Flores-López LA, Cruz-López M, García-Macedo R, et al. Phosphorylation, ON-acetylglucosaminylation and poly-ADP-ribosylation of p53 in RINm5F cells cultured in high glucose. Free Radic Biol Med. 2012;53:S95.
Ogawara Y, Kishishita S, Obata T, et al. Akt enhances Mdm2-mediated ubiquitination and degradation of p53. J Biol Chem. 2002;277:21843–50.
Barzalobre-Gerónimo R, Flores-López LA, Baiza-Gutman LA, et al. Hyperglycemia promotes p53-Mdm2 interaction but reduces p53 ubiquitination in RINm5f cells. Mol Cell Biochem. 2015;405:257–64.
Fang S, Jensen JP, Ludwig RL, Vousden KH, Weissman AM. Mdm2 is a RING finger-dependent ubiquitin protein ligase for itself and p53. J Biol Chem. 2000;275:8945–51.
Zhong I, Georgia S, Tschen S-i, et al. Essential role of Skp2-mediated p27 degradation in growth and adaptive expansion of pancreatic b cells. J Clin Invest. 2007;117:2869–76.
Vetere A, Choudhary A, Burns SM, Warner BK. Targeting the pancreatic b-cell to treat diabetes. Nat Rev Drug Discov. 2014;13:278–89.
Wang Z, York NW, Nichols CG, Remedi MS. Pancreatic β-cell dedifferentiation in diabetes and re-differentiation following insulin therapy. Cell Metab. 2014;19:872–82.
Lorenzo PI, Fuente-Martín E, Brun T, et al. PAX4 defines an expandable β-cell subpopulation in the adult pancreatic islet. Sci Rep. 2015;5:15672.
Shirakawa J, Fernandez M, Takatani T, et al. Insulin signaling regulates the FoxM1/PLK1/CENP-A pathway to promote adaptive pancreatic β cell proliferation. Cell Metab. 2017;25:868–82.
Bock T, Pakkenberg B, Buschard K. Increased islet volume but unchanged islet number in ob/ob mice. Diabetes. 2003;52:1716–22.
Fransson L, Franzén S, Rosengren V, et al. β-cell adaptation in a mouse model of glucocorticoid-induced metabolic syndrome. J Endocrinol. 2013;219:231–41.
Maiztegui B, Borelli MI, Raschia MA, Zotto HHD, Gagliardino JJ. Islet adaptive changes to fructose-induced insulin resistance: β-cell mass, glucokinase, glucose metabolism, and insulin secretion. J Endocrinol. 2009;200:139–49.
Rizkalla SW. Health implications of fructose consumption: a review of recent data. Nutr Metab (Lond). 2010;7:82.
Prentki M, Nolan CJ. Islet B cell failure in type 2 diabetes. J Clin Invest. 2006;116:1802–12.
Jones A, Danielson KM, Benton MC, et al. miRNA Signatures of insulin resistance in obesity. Obesity. 2017;25:1734–44.
Guay C, Regazzi R. MicroRNAs and the functional cell mass: for better or worse. Diabetes Metab. 2015;41:369–77.
Kalis M, Bolmeson C, Esguerra JLS, et al. Beta-cell specific deletion of Dicer1 leads to defective insulin secretion and diabetes mellitus. PLoS One. 2011;6:e29166.
Fischer-Posovszky P, Roos J, Kotnik P, et al. Functional significance and predictive value of MicroRNAs in pediatric obesity: tiny molecules with huge impact? Horm Res Paediatr. 2016;86:3–10.
Peng Y, Yu S, Li H, et al. MicroRNAs: emerging roles in adipogenesis and obesity. Cell Signal. 2015;26:1886–96.
Grieco E, Brusco N, Licata G, et al. Landscape of microRNAs in cell: between phenotype maintenance and protection. Int J Mol Sci. 2021;22:803–21.
Esguerra JLS, Bolmeson C, Cilio CM, Eliasson L. Differential glucose-regulation of MicroRNAs in pancreatic islets of non-obese type 2 diabetes model Goto-Kakizaki Rat. PLoS One. 2011;6:e18613.
Gaddam RR, Kim Y-R, Li Q, et al. Genetic deletion of miR-204 improves glycemic control despite obesity in db/db mice. Biochem Biophys Res Commun. 2020;532:167–72.
Lovis P, Roggli E, Laybutt DR, et al. Alterations in microRNA expression contribute to fatty acid–induced pancreatic β-cell dysfunction. Diabetes. 2008;57:2728–36.
Green DR, Kroemer G. Cytoplasmic functions of the tumour suppressor p53. Nature. 2009;458:1127–30.
Zhang X-D, Qin Z-H, Wang J. The role of p53 in cell metabolism. Acta Pharmacol Sin. 2010;31:1208–12.
Vienberg S, Geiger J, Madsen S, Dalgaard LT. MicroRNAs in metabolism. Acta Physiol. 2017;219:346–61.
Belongie K, Ferrannini E, Johnson K, et al. Identification of novel biomarkers to monitor β-cell function and enable early detection of type 2 diabetes risk. PLoS One. 2017;12:e0182932.
Zhu Y, You W, Wang H, et al. MicroRNA-24/MODY gene regulatory pathway mediates pancreatic β-cell dysfunction. Diabetes. 2013;62:3194–206.
Nesca V, Guay C, Jacovetti C, et al. Identification of particular groups of microRNAs that positively or negatively impact on beta cell function in obese models of type 2 diabetes. Diabetologia. 2013;56:2203–12.
Shaer A, Azarpira N, Karimi MH. Differentiation of human induced pluripotent stem cells into insulin-like cell clusters with miR-186 and miR-375 by using chemical transfection. Appl Biochem Biotechnol. 2014;174:242–58.
Regazzi R. MicroRNAs as therapeutic targets for the treatment of diabetes mellitus and its complications. Expert Opin Ther Targets. 2018;22:153–60.
Accili D. Can COVID-19 cause diabetes? Nat Metab. 2021;3:123–5.
Shang J, Wan Y, Luo C, et al. Cell entry mechanisms of SARS-CoV-2. Proc Natl Acad Sci U S A. 2020;117:11727–34.
Ibrahim S, Monaco GS, Sims EK. Not so sweet and simple: impacts of SARS-CoV-2 on the β cell. Islets. 2021;13:66–79.
Geravandi S, Mahmoudi-aznaveh A, Azizi Z, Maedler K, Ardestani A. SARS-CoV-2 and pancreas: a potential pathological interaction? Trends Endocrinol Metab. 2021;32:842–5.
Hayden MR. An immediate and long-term complication of COVID-19 may be type 2 diabetes mellitus: the central role of β-cell dysfunction, apoptosis and exploration of possible mechanisms. Cell. 2020;9:2475–98.
Wu C-T, Lidsk PV, **ao Y, et al. SARS-CoV-2 infects human pancreatic β cells and elicits β cell impairment. Cell Metab. 2021;33:1565–76.
Steenblock C, Richter S, Berger I, et al. Viral infiltration of pancreatic islets in patients with COVID-19. Nat Commun. 2021;12:3534.
Coate KC, Cha J, Shrestha S, et al. SARS-CoV-2 cell entry factors ACE2 and TMPRSS2 are expressed in the microvasculature and ducts of human pancreas but are not enriched in β cells. Cell Metab. 2020;32:1028–40.
Further Reading
Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54(6):1615–25. The author presents a unified mechanism that links overproduction of superoxide by the mitochondrial electron-transport chain to high glucose-mediated damage and diabetes complications. This paper provides the basis for understanding of the origin of ROS and oxidative stress in diabetes.
Grieco E, Brusco N, Licata G, Fignani D, Formichi C, Nigi L, Sebastiani G, Dotta F. Landscape of microRNAs in cell: between phenotype maintenance and protection. Int J Mol Sci. 2021;22:803–21. https://doi.org/10.3390/ijms22020803. This review is about the most important miRNAs regulating the maintenance and the robustness of β cell identity.
Hasnain SZ, et al. Oxidative and endoplasmic reticulum stress in b-cell dysfunction in diabetes. J Mol Endocrinol. 2016;56:R33–54. https://doi.org/10.1530/JME-15-0232. Here, the importance of deleterious effects of oxidative stress and endoplasmic reticulum stress-induced unfolded protein response is evaluated on β-cell insulin synthesis and secretion as well as on inflammatory signaling and apoptosis. Additionally, the authors describe recent findings on how inflammatory cytokines contribute to β-cell dysfunction and protect interleukin 22.
Hayden MR. An immediate and long-term complication of COVID-19 may be type 2 diabetes mellitus: the central role of β-cell dysfunction, apoptosis and exploration of possible mechanisms. Cell. 2020;9:2475–98. https://doi.org/10.3390/cells9112475. The author review COVID-19 participation on development of type 2 diabetes.
Hudish LI, Reusch JE, Sussel L. β Cell dysfunction during progression of metabolic syndrome to type 2 diabetes. J Clin Investig. 2019;129:4001–8. https://doi.org/10.1172/JCI129188. These authors focus on the current understanding from rodent and human studies of the progression of β cell responses during the development of MetS.
Kaufman B, et al. Mitochondrial regulation of β-cell function: maintaining the momentum for insulin release. Mol Aspects Med. 2015;42:91–104. https://doi.org/10.1016/j.mam.2015.01.004. Pancreatic β-cell function and insulin release is mitochondria dependent. In this work, the authors review mitochondrial metabolism and control of mitochondrial mass as they relate to pancreatic β-cell function.
Ortega-Camarillo C, et al. The role of p53 in pancreatic β-cell apoptosis. Immunoendocrinology. 2015;2:e1075. https://doi.org/10.14800/ie.1075. © 2015. This paper examines p53 mobilization to a mitochondrion and its phosphorylation, as well as the activation of the intrinsic route of β-cell apoptosis by hyperglycemia. They also describe how hyperglycemia affects the p53 degradation pathways.
Sharma RB, Alonso LC. Lipotoxicity in the pancreatic beta cell: not just survival and function, but proliferation as well? Curr Diab Rep. 2014;14(6):492. https://doi.org/10.1007/s11892-014-0492-2. This paper reviews free fatty acids’ (FFAs) positive and negative effects on beta cell survival and insulin secretion. It also examines strong new findings that lipids may also impair compensatory beta cell proliferation.
Strycharz J, et al. Is p53 involved in tissue-specific insulin resistance formation? Oxid Med Cell Longev. 2017;2017:9270549, 23p. https://doi.org/10.1155/2017/9270549. The protein p53 is connected with metabolic defects underlying cellular aging, obesity, inflammation and β-cells apoptosis. Additionally, the authors discuss p53 regulation of multiple biochemical processes such as glycolysis, oxidative phosphorylation, lipolysis, lipogenesis, 𝛽-oxidation, gluconeogenesis, and glycogen synthesis.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Glossary
- Adipokines
-
Cytokines (cell signaling proteins) secreted by adipose tissue.
- Amylin
-
A 37-amino acid peptide hormone, discovered in 1987, which is co-located and co-secreted with insulin by the pancreatic beta-cells in response to nutrient stimuli.
- Antioxidant
-
Molecule that inhibits the oxidation of other molecules.
- Apoptosis (a-po-toe-sis)
-
Was first used by Kerr, Wyllie, and Currie in 1972 to describe a morphologically distinct form of cell death and energy-dependent biochemical mechanisms.
- Apoptosome
-
Molecular complex of two major components—the adapter protein apoptotic protease activating factor 1 (Apaf1) and the pro caspase-9. These are assembled during apoptosis upon Apaf1 interaction with cytochrome c. Apoptosome assembly triggers effector caspase activation.
- Cardiolipin
-
Phospholipid important of the inner mitochondrial membrane, where it constitutes about 20% of the total lipid composition.
- Caspase (cysteine-aspartic proteases, cysteine aspartases, or cysteine-dependent aspartate-directed proteases)
-
Family of protease enzymes playing essential roles in apoptosis and inflammation.
- Ceramides
-
Family of waxy lipid molecules. A ceramide is composed of sphingosine and a fatty acid.
- Cytochrome c
-
Heme protein serving as electron carrier in respiration. Cytochrome c is also an intermediate of apoptosis.
- Cytokines
-
Cell signaling small proteins. Involved in autocrine signalling, paracrine signalling, and endocrine signalling as immunomodulating agents
- Dedifferentiation process
-
Processes by which cell that were specialized for a specific function lose their specialization.
- Fission
-
Division of mitochondria into new mitochondria.
- Flavoprotein
-
Proteins that contain a nucleic acid derivative of riboflavin: the flavin adenine dinucleotide (FAD) or flavin mononucleotide (FMN).
- Fusion
-
Process mediated by several large GTPases whose combined effects lead to the dynamic mitochondrial networks seen in many cell types.
- Glucolipotoxicity
-
Combined, deleterious effects of elevated glucose, and fatty acid levels on pancreatic beta-cell function and survival.
- Hyperlipidemia
-
Elevation of fats or lipids in the blood.
- Hyperplasia
-
Enlargement of an organ or tissue caused by an increase in the cell proliferation rate.
- Inflammasome
-
A multiprotein cytoplasmic complex which activates one or more caspases, leading to the processing and secretion of pro-inflammatory cytokines—e.g., IL-1 beta, IL-18, and IL-33. Assembly of inflammasomes depends on the NOD-like receptor family members, such as the NALP proteins kinase: enzyme-catalyzing phosphorylation of an acceptor molecule by ATP.
- Misfolded proteins
-
Are proteins structurally abnormal, and thereby disrupt the function of cells, tissues, and organs. Proteins that fail to fold into their normal configuration; in this misfolded state, the proteins can become noxious in some way and can lose their normal function.
- Mitofusins
-
Proteins that participate in mitochondrial fusion.
- Necrosis
-
Morphological changes in cell death caused by enzymatic degradation.
- Neogenesis
-
Generation of new cells.
- Oxidative stress
-
Pathological changes in living organisms in response to excessive levels of intracellular free radicals.
- Proenzyme
-
Precursor of an enzyme, requiring some change (hydrolysis of an inhibiting fragment that masks an active grou**) to render it active form.
- Proteasome
-
An intracellular complex enzymatic that degrades misfolded or damaged proteins (proteolysis), after damaged proteins are tagged by ubiquitin.
- Resistance to insulin
-
Pathological condition in which cells fail to respond normally to the hormone insulin.
- RING finger domain
-
Really interesting new gene-finger is a proteins domain that plays a key role in the ubiquitination process.
- Stem cells
-
Undifferentiated biological cells that can differentiate into specialized cells and can divide.
- Sumoylation
-
Small ubiquitin-like modifier (or SUMO) proteins are a family of small proteins that are covalently attached to and detached from other proteins in cells to modify their function. Post-translational modification involved in various cellular processes.
- Triacylglycerol
-
Ester of glycerol with three molecules of fatty acid.
- Ubiquitin
-
Small (8.5 kDa) regulatory protein that has been found in almost all tissues (ubiquitously) of eukaryotic organisms and regulated proteolysis.
- Ubiquitin ligase
-
Protein that recruits, recognizes a protein substrate, and catalyzes the transfer of ubiquitin from the E2 enzyme to the protein substrate.
- Uncoupling proteins
-
Proteins that uncouples phosphorylation of ADP from electron transport.
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Camarillo, C.O. (2023). Dysfunction and Death of Pancreatic Beta-Cells in Type 2 Diabetes. In: Rodriguez-Saldana, J. (eds) The Diabetes Textbook. Springer, Cham. https://doi.org/10.1007/978-3-031-25519-9_13
Download citation
DOI: https://doi.org/10.1007/978-3-031-25519-9_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-25518-2
Online ISBN: 978-3-031-25519-9
eBook Packages: MedicineMedicine (R0)