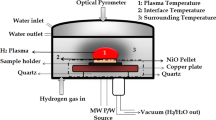
Abstract
Hydrogen is a candidate to replace carbon in metal production, as it can reduce some metal ores (e.g., iron ore). However, for other oxides, such as those of manganese and chromium, the situation is much more challenging. As the exotic species found in hydrogen plasma are much more reactive than molecular hydrogen, the use of hydrogen plasma can improve hydrogen reduction for all these oxides. Here, using a plasma arc melter, samples of Fe2O3, Cr2O3 and MnO have been exposed to hydrogen plasma. Reactions between oxides and hydrogen have been observed in all cases, producing metallic iron, chromium, and manganese, hinting that plasma technology can play a part in sustainable metal production, allowing for carbon free production of chromium and manganese. The paper also discusses possible reaction mechanisms.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kero IT, Dalaker Halvor, Osen KS, Ringdalen E (2021) Some carbon-free technologies for manganese ferroalloy production. In: Proceedings of the 16th international ferro-alloys congress (INFACON XVI). Available at SSRN: https://ssrn.com/abstract=3926069 or https://doi.org/10.2139/ssrn.3926069
European Commission (2020) Communication from the commission to the European parliament. The Council, The European economic and social committee and the committee of the regions—a hydrogen strategy for a climate-neutral Europe. https://ec.europa.eu/energy/sites/ener/files/hydrogen_strategy.pdf
Sabat KC, Rajput P, Paramguru RK, Bhoi B, Mishra BK (2014) Reduction of oxide minerals by hydrogen plasma: an overview. Plasma Chem Plasma Process 34:1–23. https://doi.org/10.1007/s11090-013-9484-2
Bale CW, Bélisle E, Chartrand P, Decterov SA, Eriksson G, Gheribi AE, Hack K, Jung IH, Kang YB, Melançon J, Pelton AD, Petersen S, Robelin C. Sangster J, Van Ende MA (2016) FactSage thermochemical software and databases, 2010–2016. Calphad 54:35–53. www.factsage.com
Barner HE, Mantell CL (1968) Kinetics of hydrogen reduction of manganese dioxide. Ind Eng Chem Process Des Dev 7(2):285–294. https://doi.org/10.1021/i260026a023
Safarian J (2022) A sustainable process to produce manganese and its alloys through hydrogen and aluminothermic reduction. Processes 10(1):27. https://doi.org/10.3390/pr10010027
Davies J, Paktunc D, Ramos-Hernandez J, Tangstad M, Ringdalen E, Beukes J, Bessarabov D, du Preez S (2022) The use of hydrogen as a potential reductant in the chromite smelting industry. Minerals 12:534. https://doi.org/10.3390/min12050534
SSAB (2021) The world’s first fossil-free steel ready for delivery. https://www.ssab.com/en/news/2021/08/the-worlds-first-fossilfree-steel-ready-for-delivery [Accessed 03 Aug 2022]
Thyssenkrupp (2021) Green hydrogen for the decarbonization of the steel industry: Thyssenkrupp Steel and STEAG agree on hydrogen supply. https://www.thyssenkrupp-steel.com/en/newsroom/press-releases/thyssenkrupp-steel-and-steag-agree-on-hydrogen-supply.html [Accessed 03 Aug 2022]
Seftejani MN, Schenk J (2018) Thermodynamic of liquid iron ore reduction by hydrogen thermal plasma. Metals 8(12):1051. https://doi.org/10.3390/met8121051
SINTEF (2021) HyPla-Hydrogen plasma for CO2-free metal production. https://www.sintef.no/en/projects/2020/hypla-hydrogen-plasma-for-co2-free-metal-production/ [Accessed 03 Aug 2022]
Souza Filho IR, Ma Y, Kulse M, Ponge D, Gault B, Springer H, Raabe D (2021) Sustainable steel through hydrogen plasma reduction of iron ore: process, kinetics, microstructure, chemistry. Acta Mater 213:116971. https://doi.org/10.1016/j.actamat.2021.116971
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Minerals, Metals & Materials Society
About this paper
Cite this paper
Dalaker, H., Hovig, E.W. (2023). Hydrogen Plasma-Based Reduction of Metal Oxides. In: Fleuriault, C., et al. Advances in Pyrometallurgy. TMS 2023. The Minerals, Metals & Materials Series. Springer, Cham. https://doi.org/10.1007/978-3-031-22634-2_8
Download citation
DOI: https://doi.org/10.1007/978-3-031-22634-2_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-22633-5
Online ISBN: 978-3-031-22634-2
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)