Abstract
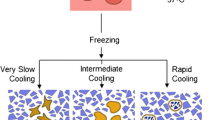
Cell freezing and thawing have long been a standard procedure in cell biology. The procedure, also known as cryonics, has effectively stopped the cellular clock, thereby adds tremendous repeatability to studies performed at different times by allowing extremely similar samples of cells to be utilised weeks, months or even ages apart. It also saves time, resources and money. The primary issue during the entire freezing–storage–thawing process is to obtain higher cell viability and, perhaps more crucially, predictable and consistent cell behaviour following cryopreservation. Many various rules and regulations have been evolved over time to accomplish this. The development of intracellular ice is the primary cause of cell death during cryopreservation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Akhtar, T., Pegg, D. E., & Foreman, J. (1979). The effect of cooling and warming rates on the survival of cryopreserved L-cells. Cryobiology, 16(5), 424–429.
Arai, K., Murata, D., Takao, S., Verissiomo, A. R., & Nakayama, K. (2020). Cryopreservation method for spheroids and fabrication of scaffold-free tubular constructs. PLoS One, 15(11), e0230428.
Armitage, W. J., & Mazur, P. (1984). Toxic and osmotic effects of glycerol on human granulocytes. American Journal of Physics, 247(5 Pt 1), C382–C389. https://doi.org/10.1152/ajpcell.1984.247.5.C382
Chua, K. J., & Chou, S. K. (2009). On the study of the freeze–thaw thermal process of a biological system. Applied Thermal Engineering, 29(17), 3696–3709.
Hunt, C. J. (2019). Technical considerations in the freezing, low-temperature storage and thawing of stem cells for cellular therapies. Transfusion Medicine and Hemotherapy, 46(3), 134–150.
Klossner, D. P., Robilotto, A. T., Clarke, D. M., VanBuskirk, R. G., Baust, J. M., Gage, A. A., & Baust, J. G. (2007). Cryosurgical technique: Assessment of the fundamental variables using human prostate cancer model systems. Cryobiology, 55(3), 189–199.
Mazur, P., Miller, R. H., & Leibo, S. P. (1974). Survival of frozen-thawed bovine red cells as a function of the permeation of glycerol and sucrose. The Journal of Membrane Biology, 15(1), 137–158.
Norkus, M., Kilmartin, L., Fay, D., Murphy, M. J., Olaighin, G., & Barry, F. (2013). The effect of temperature elevation on cryopreserved mesenchymal stem cells. Cryo Letters, 34(4), 349–359.
Cosentino, L. M., Corwin, W., Baust, J. M., Diaz-Mayoral, N., Cooley, H., Shao, W., et al. (2007). Preliminary report: evaluation of storage conditions and Cryococktails during peripheral blood mononuclear cell cryopreservation. Cell Preservation and Technology, 5(4), 189–204.
Strober, W. (2015). Trypan blue exclusion test of cell viability. Current Protocols in Immunology, 111, A3.B.1–A3.B.3. https://doi.org/10.1002/0471142735.ima03bs111.
Thompson, M. L., Kunkel, E. J., & Ehrhardt, R. O. (2014). Cryopreservation and thawing of mammalian cells. eLS, 9, 3417.
Yokoyama, W. M., Thompson, M. L., & Ehrhardt, R. O. (2012). Cryopreservation and thawing of cells. Current Protocols in Immunology, 99(1), A.3G.1–A.3G.5.
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Agarwal, V., Singh, M. (2023). Resuscitation of Frozen Cell Lines. In: Animal Cell Culture: Principles and Practice. Techniques in Life Science and Biomedicine for the Non-Expert. Springer, Cham. https://doi.org/10.1007/978-3-031-19485-6_11
Download citation
DOI: https://doi.org/10.1007/978-3-031-19485-6_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-19484-9
Online ISBN: 978-3-031-19485-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)