Abstract
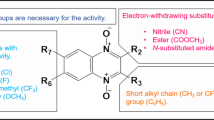
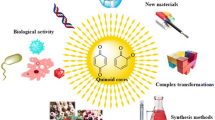
Natural products represent important drugs since ancient times, nowadays as endless opportunities to find new compounds with pharmacological activities, a great number of new drugs are still derived from natural sources. Quinones and their derivatives have a fundamental role in several applications, i.e., pharmaceutical, medicinal, and food industries. Quinones represent a large class of compounds showing fascinating chemistry: they interact as electron transfer agents with biological targets by the formation of covalent bindings in redox reactions. Quinones constitute an important class of natural and synthetic compounds. A wide variety of synthetic quinones were prepared for develo** structures with pharmacological activities.
Their structure makes them interfere/udergo in chemical transformations. A great interest by the scientific community is given to quinone-based compounds for their challenging structural elements and potential therapeutic properties.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Agarwala N, Rohani L, Hastings G (2022) Experimental and calculated infrared spectra of disubstituted naphthoquinones. Spectrochim Acta A Mol Biomol Spectrosc 2022:120674
Ahmadi ES, Tajbakhsh A, Iranshahy M, Asili J, Kretschmer N, Shakeri A, Sahebkar A (2020) Naphthoquinone derivatives isolated from plants: recent advances in biological activity. Mini Rev Med Chem 20(19):2019–2035. https://doi.org/10.2174/1389557520666200818212020
Aminin D, Polonik S (2020) 1,4-naphthoquinones: some biological properties and application. Chem Pharm Bull 68(1):46–57. https://doi.org/10.1248/cpb.c19-00911
Annisa R, Hendradi E, Yuwono M (2020) Analysis of 1,4 naphthoquinone in the Indonesian medical plant from extract Eleutherine palmifolia (L.) Merr by UHPLC. IOP Conf Ser Earth Environ Sci 456(1):012020
Aranda-López Y, López-López L, Castro K, Ponce-Regalado MD, Becerril-Villanueva LE, Girón-Pérez MI, Del Río-Araiza VH, Morales-Montor J (2021) Cysticidal effect of a pure naphthoquinone on Taenia crassiceps cysticerci. Parasitol Res 120(11):3783–3794. https://doi.org/10.1007/s00436-021-07281-x
Asha A, Suma S (2022) Synthesis, electrochemical and anti-microbial study of 2,5-diamino benzoquinones. J Indian Chem Soc 99:100316
Badary OA, Hamza MS, Tikamdas R (2021) Thymoquinone: a promising natural compound with potential benefits for COVID-19 prevention and cure. Drug Des Devel Ther 15:1819–1833. https://doi.org/10.2147/DDDT.S308863
Carcamo-Noriega EN, Sathyamoorthi S, Banerjee S, Gnanamani E, Mendoza-Trujillo M, Mata-Espinosa D, Hernández-Pando R, Veytia-Bucheli JI, Possani LD, Zare RN (2019) 1,4-benzoquinone antimicrobial agents against Staphylococcus aureus and mycobacterium tuberculosis derived from scorpion venom. Proc Natl Acad Sci U S A 116(26):12642–12647. https://doi.org/10.1073/pnas.1812334116
Çiçek SS, Ugolini T, Girreser U (2019) Two-dimensional qNMR of anthraquinones in Frangula alnus (Rhamnus frangula) using surrogate standards and delay time adaption. Anal Chim Acta 1081:131–137. https://doi.org/10.1016/j.aca.2019.06.046
Devi SP, Kumaria S, Rao SR, Tandon P (2016) Carnivorous plants as a source of potent bioactive compound: naphthoquinones. Trop Plant Biol 9(4):267–279
Duval J, Pecher V, Poujol M, Lesellier E (2016) Research advances for the extraction, analysis and uses of anthraquinones: a review. Ind Crop Prod 94:812–833
EuroFIR eBASIS (n.d.) Bioactive substances in food information system. Available at: http://ebasis.eurofir.org/Default.asp. Accessed 18 Jan 2022
Farooq U, Pan Y, Disasa D, Qi J (2019) Novel anti-aging benzoquinone derivatives from Onosma bracteatum wall. Molecules (Basel, Switzerland) 24(7):1428. https://doi.org/10.3390/molecules24071428
Ferraris D, Lapidus R, Truong P et al (2021) Pre-clinical activity of amino-alcohol dimeric naphthoquinones as potential therapeutics for acute myeloid leukemia. Anticancer Agents Med Chem. https://doi.org/10.2174/1871520621666210602131558
Gartman JA, Tambar UK (2022) Recent total syntheses of anthraquinone-based natural products. Tetrahedron 105:132501
Gecibesler IH, Disli F, Bayindir S, Toprak M, Tufekci AR, Sahin Yaglıoglu A, Altun M, Kocak A, Demirtas I, Adem S (2021) The isolation of secondary metabolites from Rheum ribes L. and the synthesis of new semi-synthetic anthraquinones: isolation, synthesis and biological activity. Food Chem 342:128378. https://doi.org/10.1016/j.foodchem.2020.128378
Han Y, van der Heijden R, Lefeber AWM, Erkelens C, Verpoorte R (2002) Biosynthesis of anthraquinones in cell cultures of Cinchona “Robusta” proceeds via the methylerythritol 4-phosphate pathway. Phytochemistry 59:45–55
Harry NA, Saranya S, Krishnan KK, Anilkumar G (2017) Recent advances in the chemistry of masked ortho-benzoquinones and their applications in organic synthesis Asian. J Org Chem 6(8):945–966
Hosseini S, Pourmousavi SA, Mahdavi MT, P. (2021) Synthesis, and in vitro biological evaluations of novel naphthoquinone conjugated to aryl triazole acetamide derivatives as potential anti-Alzheimer agents. J Mol Struct 132229
Japp FR (1879) LIV. - On the action of organo-zinc compounds on quinones. (Preliminary note). J Chem Soc 35:526–528
Kamo S, Kuramochi K, Tsubaki K (2018) Recent topics in total syntheses of natural dimeric naphthoquinone derivatives. Tetrahedron Lett 59:224–23017
Kiely M, Black LJ, Plumb J, Kroon PA, Hollman PC, Larsen JC, Speijers GJ, Kapsokefalou M, Sheehan D, Gry J, Finglas P, EuroFIR consortium (2010) EuroFIR eBASIS: application for health claims submissions and evaluations. Eur J Clin Nutr 64(Suppl 3):S101–S107. https://doi.org/10.1038/ejcn.2010.219
Kim G, Lee S-E (2021) Antifungal and antiaflatoxigenic properties of naphthoquinones toward Aspergillus flavus and their mode of inhibitory action on aflatoxin biosynthesis. Food Control 119:107506
Kumagai Y, Shinkai Y, Miura T, Cho AK (2012) The chemical biology of naphthoquinones and its environmental implications. Annu Rev Pharmacol Toxicol 52:221–247. https://doi.org/10.1146/annurev-pharmtox-010611-134517
Li Y, Jiang JG (2018) Health functions and structure-activity relationships of natural anthraquinones from plants. Food Funct 9:6063–6080
Luo Y, Shen HY, Shen QX, Cao ZH, Zhang M, Long SY, Wang ZB, Tan JW (2017) A new anthraquinone and a new naphthoquinone from the whole plant of Spermacoce latifolia. J Asian Nat Prod Res 19(9):869–876. https://doi.org/10.1080/10286020.2017.1279609
MacMillan F, Hunte C (2010) Quinone binding and catalysis. Biochim Biophys Acta Bioenerg 1797(12):1841
Miao Y, Yanling W, Ying** J, Mengze L, Jixing N, Xue W (2020) Benzoquinone derivatives with antioxidant activity inhibit activated hepatic stellate cells and attenuate liver fibrosis in TAA-induced mice. Chem Biol Interact 317:108945. https://doi.org/10.1016/j.cbi.2020.108945
Minnikin DE, O’Donnell AG, Goodfellow M, Alderson G, Athalye M, Schaal A, Parlett JH (1984) An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J Microbiol Methods 2(5):233–241
Morgan I, Wessjohann LA, Kaluderović GN (2022) In vitro anticancer screening and preliminary mechanistic study of A-ring substituted Anthraquinone derivatives. Cell 11(1):168
Plumb J, Pigat S, Bompola F, Cushen M, Pinchen H, Nørby E, Astley S, Lyons J, Kiely M, Finglas P (2017) eBASIS (bioactive substances in food information systems) and bioactive intakes: major updates of the bioactive compound composition and beneficial bioeffects database and the development of a probabilistic model to assess intakes in Europe. Nutrients 9(4):320. https://doi.org/10.3390/nu9040320
Price ER, Johnson SC (2013) Quinones: occurrence, medicinal uses and physiological importance. Nova Science Publishers, Hauppauge, NY, pp 1–182
Qin D, **ang B, Zhou X, Qiu S, ** J (2022) Microemulsion as solvent for naphthoquinones extraction from walnut (Juglans mandshurica Maxim) green husk using high voltage electrical discharge. Sep Purif Technol 28115:119983
Qiu H-Y, Wang P-F, Lin H-Y, Tang C-Y, Zhu H-L, Yang Y-H (2018) Naphthoquinones: a continuing source for discovery of therapeutic antineoplastic agents. Chem Biol Drug Des 91(3):681–690
Radhika S, Saranya S, Harry NA, Anilkumar G (2019) Recent advances and prospects in the chemistry of o-benzoquinones. ChemistrySelect 4(31):9124–913423
Rani R, Narsiman B, Varma RS, Kumar R (2022) Gum-based nanocapsules comprising naphthoquinones enhance the apoptotic and trypanocidal activity against Trypanosoma evansi. Eur J Pharm Sci 171:106118. https://doi.org/10.1016/j.ejps.2022.106118
Sahoo PMS, Behera S, Behura R, Acharya A, Biswal D, Suna SK, Sahoo R, Soren RC, Jali BR (2022) A brief review: antibacterial activity of Quinone derivatives. Biointerface Res Appl Chem 12(3):3247–3258
Shakour ZT, Farag MA (2021) Diverse host-associated fungal systems as a dynamic source of novel bioactive anthraquinones in drug discovery: current status and future perspectives. J Adv Res. https://doi.org/10.1016/j.jare.2021.11.007
Shi BJ, Zhang WD, Jiang HF, Zhu YY, Chen L, Zha XM, Lu YY, Zhang WM (2016) A new anthraquinone from seed of Cassia obtusifolia. Nat Prod Res 30(1):35–41. https://doi.org/10.1080/14786419.2015.1032280
Sies H, Packer L (2004a) Quinones and quinone enzymes: Part A - Preface. Methods Enzymol 378:xv–xvi
Sies H, Packer L (2004b) quinones and quinone enzymes: Part B - Preface. Methods Enzymol 382:xv–xvi
Silakari P, Priyanka, Piplani P (2020) P-benzoquinone as a privileged scaffold of pharmacological significance: a review. Mini Rev Med Chem 20(16):1586–1609
Souto EB, Silva GF, Dias-Ferreira J, Zielinska A, Ventura F, Durazzo A, Lucarini M, Novellino E, Santini A (2020a) Nanopharmaceutics: Part II—Production scales and clinically compliant production methods. Nanomaterials (Basel) 10(3):E455. https://doi.org/10.3390/nano10030455
Souto EB, Silva GF, Dias-Ferreira J, Zielinska A, Ventura F, Durazzo A, Lucarini M, Novellino E, Santini A (2020b) Nanopharmaceutics: Part I—Clinical trials legislation and good manufacturing practices (GMP) of nanotherapeutics in the EU. Pharmaceutics 12(2):E146. https://doi.org/10.3390/pharmaceutics12020146
Wellington KW (2015) Understanding cancer and the anticancer activities of naphthoquinones: a review. RSC Adv 5(26):20309–20338
Wróbel-Biedrawa D, Grabowska K, Galanty A, Sobolewska D, Żmudzki P, Podolak I (2020) Anti-melanoma potential of two benzoquinone homologues embelin and rapanone - a comparative in vitro study. Toxicol In Vitro 65:104826. https://doi.org/10.1016/j.tiv.2020.104826
Wu W, Wang S, Zhang H, Guo W, Lu H, Xu H, Zhan R, Fidan O, Sun L (2021a) Biosynthesis of novel naphthoquinone derivatives in the commonly-used chassis cells Saccharomyces cerevisiae and Escherichia coli. Appl Biochem Microbiol 57:S11–S26
Wu Q, Er-Bu A, Liang X, Luan S, He C, Yin L, Yin Z, Zou Y, Li L, Song X (2021b) Determination of the main naphthoquinones in Onosma hookeri Clarke. var. longiforum Duthie and its optimization of the ultrasound-assisted extraction using response surface methodology. J Food Sci 86(2):357–365. https://doi.org/10.1111/1750-3841.15460
Yeung AWK, Souto EB, Durazzo A, Lucarini M, Novellino E, Tewari D, Wang D, Atanasov AG, Santini A (2020) Big impact of nanoparticles: analysis of the most cited nanopharmaceuticals and nanonutraceuticals research. Curr Res Biotechnol 2:53–63. https://doi.org/10.1016/j.crbiot.2020.04.002
Yu N, Ye Q, Nie X, **a C, Meng X (2021) Distribution of 2-tert-butyl-1,4-benzoquinone and its precursor, tert-butylhydroquinone, in typical edible oils and oleaginous foods marketed in Hangzhou City, China. Food Chem 361:130039
Zamora R, Hidalgo FJ (2021) Formation of naphthoquinones and anthraquinones by carbonyl-hydroquinone/benzoquinone reactions: a potential route for the origin of 9,10-anthraquinone in tea. Food Chem 354:129530
Zhang R, Zhang R, Jian R, Zhang L, Zhang MT, **a Y, Luo S (2022) Bio-inspired lanthanum-ortho-quinone catalysis for aerobic alcohol oxidation: semi-quinone anionic radical as redox ligand. Nat Commun 13(1):428. https://doi.org/10.1038/s41467-022-28102-4
Zhao PP, Tong JM, Tian YF, Zhang SF (2016) Research progress in pharmacological effects of anthraquinones. J Chengde Med Coll 33:152–155
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Durazzo, A., Gabrielli, P., Lucarini, M. (2023). Anthra-, Benzo-, and Naphthoquinones. In: Carocho, M., Heleno, S.A., Barros, L. (eds) Natural Secondary Metabolites. Springer, Cham. https://doi.org/10.1007/978-3-031-18587-8_9
Download citation
DOI: https://doi.org/10.1007/978-3-031-18587-8_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-18586-1
Online ISBN: 978-3-031-18587-8
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)