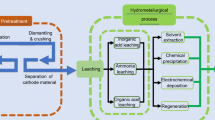
Abstract.
Demand of high-purity lithium hydroxide (LiOH) has increased significantly with the rapid advancement of lithium batteries for clean technologies and energy storage. Lithium hydroxide is commercially produced by the addition of lime into lithium carbonate. While this process produces a low concentration of LiOH, a significant amount of lithium is lost. To overcome these limitations, substantial studies have been conducted to develop new technologies to produce battery-grade LiOH from lithium sulfate and chloride solutions by causticization and membrane electrolysis. Furthermore, the membrane electrolysis processes have been shown to be a promising new technology for battery-grade LiOH production. Therefore, this paper reviews these technologies with emphasis on the process chemistry, key flowsheets, and challenges and potential improvement opportunities of the technologies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
González A, Grágeda M, Quispe A, Ushak S, Sistat P, Cretin M. Application and analysis of bipolar membrane electrodialysis for LiOH production at high electrolyte concentrations: current scope and challenges. Membranes. 2021;11:575.
Wietelmann U, Bauer RJ. Ullmann’s encyclopedia of industrial chemistry. Weinheim: Wiley-VCH; 2000. https://doi.org/10.1002/14356007.a15_393.
Buckley D, Genders JD, Atherton D. Method of making high purity lithium hydroxide and hydrochloric acid. US 2011/0044882 A1. 14 Feb 2011.
Turan Z, Baloglu H, Ünveren E, Bulutcu AN. The behaviour of Nafion® 424 membrane in the electrochemical production of lithium hydroxide. J Chem Technol Biotechnol. 2016;91(9):2529–38. https://doi.org/10.1002/jctb.4853.
Grageda M, Gonzalez A, Quispe A, Ushak SA. Analysis of a process for producing battery grade lithium hydroxide by membrane electrodialysis. Membranes. 2020;10:198.
Nemkov NM, Ryabtsev AD, Ìu** VV. Preparation of high purity lithium hydroxide monohydrate from lithium-containing wastes of various industries. Izv Tomsk Polytech Univ. 2004;77:80–4.
Grágeda M, González A, Alavia W, Ushak S. Development and optimización of a modified process for producing the battery grade LiOH: optimization of energy and water consumption. Energy. 2015;89:667–77.
Liu H, Azimi G. Production of battery grade lithium hydroxide monohydrate using barium hydroxide causticizing agent. Resour Conserv Recycl. 2022;179:106115.
Nan** L, Jianmin G, **%20L%2C%20Jianmin%20G%2C%20**aohui%20J.%20Production%20process%20of%20lithium%20hydroxide%20monohydrate.%20CN1214981C.%2017%20Aug%202005%20%28Chinese%29."> Google Scholar
Gmar S, Chagnes A. Recent advances on electrodialysis for the recovery of lithium from primary and secondary resources. Hydrometallurgy. 2019;189:105124.
Nemkov NM, Ryabtsev AD, Kotsupalo NP, Menzheres LT, Mamylova EV, Chayukova OI. Preparing high-purity lithium hydroxide monohydrate by the electrochemical conversion of highly soluble lithium salts. Theor Found Chem Eng. 2018;54(4):710–8.
Sharma Y. Production of lithium hydroxide. WO2016/070217A1. 12 May 2016.
Magnan J-F, Bourassa G, Pearse G, Symons P, Genders JD, Clayto G. Processes and system for preparing lithium hydroxide. US10036094B2. 31 July 2018.
Magnan J-F, Bourassa G, Pearse G, Symons P, Genders JD, Bar D, Langevin M-E. Processes for preparing lithium hydroxide. EP2971252B1. 30 Dec 2020.
Binnemans K. Method for producing battery grade lithium hydroxide monohydrate. WO2021228936A1. 18 Nov 2021.
Mukul MS, Barton SB. Electrolysis process for making lithium hydroxide. WO2021212018A1. 21 Oct 2021.
Hader RN, Nielsen RL, Herre MG. Lithium and its compounds. Ind Eng Chem. 1951;1951:2636.
Yao K, ** P, Huo L, Tu M, Dong H, Huang C, **a G. Method for preparing battery-stage monohydrate lithium hydroxide. CN 200710051016.5. 9 July 2007.
Jian G, Peng W, Yaoyao L, Min Z, Wenxuan L, Huan Z. Solid-liquid phase equilibria in the process of producing lithium hydroxide from lithium sulfate. Chem Res Chin Univ. 2019. https://doi.org/10.1007/s40242-019-9197-z.
Ryabtsev AD, Nemkov NM, Kotsupalo NP, Serikova LA. Preparation of high-purity lithium hydroxide monohydrate from technical-grade lithium carbonate by membrane electrolysis. Zhurnal Prikladnoj Khimii. 2004;77:1123–31.
Koter S, Warszawski A. A new model for characterization of bipolar membrane electrodialysis of brine. Desalination. 2006;198:111–23.
Jiang C, Wang Y, Wang Q, Feng H, Xu T. Production of lithium hydroxide from lake brines through electro-electrodialysis with bipolar membranes (EEDBM). Ind Eng Chem Res. 2014;53:6103–12.
Melnikov S, Sheldeshov N, Zabolotsky V, Loza S, Achoh A. Pilot scale complex electrodialysis technology for processing a solution of lithium chloride containing organic solvents. Sep Purif Technol. 2017;189:74–81.
Bunani S, Yoshizuka K, Nishihama S, Arda M, Kabay N. Application of bipolar membrane electrodialysis (BMED) for simultaneous separation and recovery of boron and lithium from aqueous solutions. Desalination. 2017;424:37–44.
Harrison S, Blanchet R. Processes for preparing highly pure lithium carbonate and other highly pure lithium containing compounds. EP 2749535 A1. 16 Oct 2012.
Buckley D, Genders JD, Atherton D. Method of making high purity lithium hydroxide and hydrochloric acid. US Patent 2011/0044882. 24 Feb 2011.
Chen X, Ruan X, Kentish SE, Li GK, Xu T, Chen GQ. Production of lithium hydroxide by electrodialysis with bipolar membranes. Sep Purif Technol. 2021;274:119026.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Halim, M.A., Lakshmanan, V.I., Chen, J., Kumar, S., DasGupta, S., Dehn, M. (2023). Potential Processes for Producing High-Purity Lithium Hydroxide: A Critical Review. In: Proceedings of the 61st Conference of Metallurgists, COM 2022. COM 2022. Springer, Cham. https://doi.org/10.1007/978-3-031-17425-4_78
Download citation
DOI: https://doi.org/10.1007/978-3-031-17425-4_78
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-17424-7
Online ISBN: 978-3-031-17425-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)