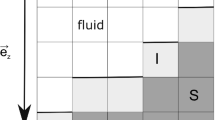
Abstract
Adsorption and fate of toxic ions in natural environments are complex phenomena involving a multitude of possible reactions at solid–fluid interfaces. Modelling of these reactions at realistic conditions defined by complex chemistry of natural waters can be a non-trivial task. Stochastic modelling approaches, such as Kinetic Monte Carlo (KMC), and the tools of statistical mechanics, can treat issues of system complexity by generation of possible system’s configurations and time-dependent system’s transitions. We present here basic theoretical background for these modelling approaches for beginners in this field. In particular, we show how KMC modelling can be applied to study adsorption of arsenic (V) on iron oxide nanocrystals. We begin this chapter with a description of molecular and microscopic structure of crystal-fluid interfaces as a necessary prerequisite for formulation of stochastic models. Stochastic modelling approach can be greatly beneficial for addressing phenomenon of toxic ion adsorption in natural water-containing systems. The application of this approach is in its embryonic state and requires a thorough and systematic development.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Allen MP, Tildesley DJ (2017) Computer simulation of liquids. Oxford University Press
Aredes S, Klein B, Pawlik M (2012) The removal of arsenic from water using natural iron oxide minerals. J Clean Prod 29–30:208–213. https://doi.org/10.1016/j.jclepro.2012.01.029
Backus EHG, Schaefer J, Bonn M (2021) Probing the mineral-water interface with nonlinear optical spectroscopy. Angew Chem Int Ed 60:10482–10501. https://doi.org/10.1002/anie.202003085
Blum AE, Lasaga AC (1987) Monte Carlo simulations of surface reaction rate laws. In: Stumm W (ed) Aquatic surface chemistry; chemical processes at the particle-water interface. Wiley, New York, pp 255–292
Bortz AB, Kalos MH, Lebowitz JL (1975) A new algorithm for Monte Carlo simulation of Ising spin systems. J Comput Phys 17:10–18. https://doi.org/10.1016/0021-9991(75)90060-1
Burghaus U (2006) A practical guide to Kinetic Monte Carlo simulations and classical molecular dynamics simulations: an example book. Nova Science Publishers
Chen Y, Bylaska EJ, Weare JH (2017) Weakly bound water structure, bond valence saturation and water dynamics at the goethite (100) surface/aqueous interface: ab initio dynamical simulations. Geochem Trans 18:3. https://doi.org/10.1186/s12932-017-0040-5
Chiavola A, D’Amato E, Boni MR (2019) Comparison of different iron oxide adsorbents for combined arsenic, vanadium and fluoride removal from drinking water. Int J Environ Sci Technol 16:6053–6064. https://doi.org/10.1007/s13762-019-02316-4
Churakov SV, Labbez C, Pegado L, Sulpizi M (2014) Intrinsic acidity of surface sites in calcium silicate hydrates and its implication to their electrokinetic properties. J Phys Chem C 118:11752–11762. https://doi.org/10.1021/jp502514a
Criscenti LJ, Kubicki JD, Brantley SL (2006) Silicate glass and mineral dissolution: calculated reaction paths and activation energies for hydrolysis of a Q3 Si by H3O+ Using Ab Initio Methods. J Phys Chem A 110:198–206. https://doi.org/10.1021/jp044360a
Exner KS, Hess F, Over H, Seitsonen AP (2015) Combined experiment and theory approach in surface chemistry: stairway to heaven? Surf Sci 640:165–180
Fouad DE, Zhang C, El-Didamony H et al (2019) Improved size, morphology and crystallinity of hematite (α-Fe2O3) nanoparticles synthesized via the precipitation route using ferric sulfate precursor. Results Phys 12:1253–1261. https://doi.org/10.1016/j.rinp.2019.01.005
Gillespie DT (1977) Exact stochastic simulation of coupled chemical reactions. J Phys Chem 81:2340–2361. https://doi.org/10.1021/j100540a008
Guo H, Xu H, Barnard AS (2013) Can hematite nanoparticles be an environmental indicator? Energy Environ Sci 6:561–569. https://doi.org/10.1039/C2EE23253G
Han Y-S, Park J-H, Min Y, Lim D-H (2020) Competitive adsorption between phosphate and arsenic in soil containing iron sulfide: XAS experiment and DFT calculation approaches. Chem Eng J 397:125426. https://doi.org/10.1016/j.cej.2020.125426
Hess F, Smarsly BM, Over H (2020) Catalytic stability studies employing dedicated model catalysts. Acc Chem Res 53:380–389. https://doi.org/10.1021/acs.accounts.9b00467
Kubicki JD, Sofo JO, Skelton AA, Bandura AV (2012) A new hypothesis for the dissolution mechanism of silicates. J Phys Chem C 116:17479–17491. https://doi.org/10.1021/jp300623v
Kurganskaya I, Churakov SV (2018) Carbonate dissolution mechanisms in the presence of electrolytes revealed by grand Canonical and Kinetic Monte Carlo modeling. J Phys Chem C 122:29285–29297. https://doi.org/10.1021/acs.jpcc.8b08986
Kurganskaya I, Niazi NK, Luttge A (2021) A modeling approach for unveiling adsorption of toxic ions on iron oxide nanocrystals. J Hazard Mater 417:126005. https://doi.org/10.1016/j.jhazmat.2021.126005
Kurganskaya I, Luttge A, Barron AR (2009) The application of VSI (Vertical scanning interferometry) to the study of crystal surface processes. In: Physical methods in inorganic and nano chemistry, 1st edn. Connexions, Rice University, Houston, Texas, USA
Labbez C, Jönsson B, Pochard I et al (2006) Surface charge density and electrokinetic potential of highly charged minerals: experiments and Monte Carlo simulations on calcium silicate hydrate. J Phys Chem B 110:9219–9230. https://doi.org/10.1021/jp057096+
Labbez C, Jönsson B, Skarba M, Borkovec M (2009) Ion−Ion correlation and charge reversal at titrating solid interfaces. Langmuir 25:7209–7213. https://doi.org/10.1021/la900853e
Labbez C, Jönsson B (2006) A new Monte Carlo method for the titration of molecules and minerals. In: Applied parallel computing. State of the art in scientific computing. Springer, Berlin, Heidelberg, pp 66–72
Langmuir I (1918) The adsorption of gases on plane surfaces of glass. Mica Platinum 40:1361–1403. https://doi.org/10.1021/ja02242a004
Morrow CP, Nangia S, Garrison BJ (2009) Ab initio investigation of dissolution mechanisms in aluminosilicate minerals. J Phys Chem A 113:1343–1352. https://doi.org/10.1021/jp8079099
Polowczyk I, Cyganowski P, Ulatowska J et al (2018) Synthetic iron oxides for adsorptive removal of arsenic. Water Air Soil Pollut 229:203. https://doi.org/10.1007/s11270-018-3866-2
Porus M, Labbez C, Maroni P, Borkovec M (2011) Adsorption of monovalent and divalent cations on planar water-silica interfaces studied by optical reflectivity and Monte Carlo simulations. J Chem Phys 135:064701. https://doi.org/10.1063/1.3622858
Reif F (2009) Fundamentals of statistical and thermal physics. Waveland Press
Rozanov YA (2013) Probability theory: a concise course. Courier Corporation
Stokes RH (1972) Debye model and the primitive model for electrolyte solutions. J Chem Phys 56:3382–3383. https://doi.org/10.1063/1.1677708
Usman M, Zarebanadkouki M, Waseem M et al (2020) Mathematical modeling of arsenic(V) adsorption onto iron oxyhydroxides in an adsorption-submerged membrane hybrid system. J Hazard Mater 400:123221. https://doi.org/10.1016/j.jhazmat.2020.123221
Valleau JP, Cohen LK (1980) Primitive model electrolytes. I. Grand canonical Monte Carlo computations. J Chem Phys 72:5935–5941. https://doi.org/10.1063/1.439092
Voter AF (2007) Introduction to the Kinetic Monte Carlo method. In: Sickafus KE, Kotomin EA, Uberuaga BP (eds) Radiation effects in solids. Springer, Netherlands, Dordrecht, pp 1–23
Wentzel ES (1969) Probability theory. Nauka, Moscow
Wheeler DA, Wang G, Ling Y et al (2012) Nanostructured hematite: synthesis, characterization, charge carrier dynamics, and photoelectrochemical properties. Energy Environ Sci 5:6682–6702. https://doi.org/10.1039/C2EE00001F
**ao Y, Lasaga AC (1994) Ab initio quantum mechanical studies of the kinetics and mechanisms of silicate dissolution: H+(H3O+) catalysis. Geochim Cosmochim Acta 58:5379–5400. https://doi.org/10.1016/0016-7037(94)90237-2
**ao Y, Lasaga AC (1996) Ab initio quantum mechanical studies of the kinetics and mechanisms of quartz dissolution: OH− catalysis. Geochim Cosmochim Acta 60:2283–2295. https://doi.org/10.1016/0016-7037(96)00101-9
Yan L, Chan T, **g C (2020) Arsenic adsorption on hematite facets: spectroscopy and DFT study. Environ Sci: Nano 7:3927–3939. https://doi.org/10.1039/D0EN00812E
Zhang S, Liu Y (2014) Molecular-level mechanisms of quartz dissolution under neutral and alkaline conditions in the presence of electrolytes. Geochem J 48:189–205. https://doi.org/10.2343/geochemj.2.0298
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kurganskaya, I. (2023). Approaches for Stochastic Modelling of Toxic Ion Adsorption at Crystal-Water Interfaces: A Case Study of Arsenic. In: Niazi, N.K., Bibi, I., Aftab, T. (eds) Global Arsenic Hazard. Environmental Science and Engineering. Springer, Cham. https://doi.org/10.1007/978-3-031-16360-9_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-16360-9_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-16359-3
Online ISBN: 978-3-031-16360-9
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)