Abstract
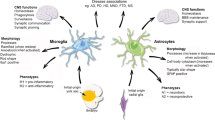
Emerging research has revealed that glial cells of the brain can produce many of the same signaling factors as cells of the peripheral immune system and, as such, can function as a brain immune system, referred to as the neuroimmune system. Both neurons and glial cells of the brain express receptors and intracellular signaling pathways that can interpret the signals communicated by these factors, which are called neuroimmune factors when produced by brain cells, and in response adjust their function. The neuroimmune system serves many roles both in normal brain biology and in brain pathology. To date, the majority of studies on the characteristics and function of the neuroimmune system comes from brain regions outside of the cerebellum, but recent studies of the cerebellum have shown that the neuroimmune system plays an important role in cerebellar development, function, and disease. This new and expanding area of research will likely bring greater understanding to the cellular and molecular mechanisms that mediate cerebellar function.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Araujo AP, Diniz LP, Eller CM, de Matos BG, Martinez R, Gomes FC (2016) Effects of transforming growth factor beta 1 in cerebellar development: role in synapse formation. Front Cell Neurosci 10:104
Araujo APB, Carpi-Santos R, Gomes FCA (2019) The role of astrocytes in the development of the cerebellum. Cerebellum (London, England) 18:1017–1035
Bacher M, Meinhardt A, Lan HY, Dhabhar FS, Mu W, Metz CN, Chesney JA, Gemsa D, Donnelly T, Atkins RC, Bucala R (1998) MIF expression in the rat brain: implications for neuronal function. Mol Med 4:217–230
Bajetto A, Bonavia R, Barbero S, Florio T, Schettini G (2001) Chemokines and their receptors in the central nervous system. Front Neuroendocrinol 22:147–184
Balzano T, Arenas YM, Dadsetan S, Forteza J, Gil-Perotin S, Cubas-Nuñez L, Casanova B, Gracià F, Varela-Andrés N, Montoliu C, Llansola M, Felipo V (2020) Sustained hyperammonemia induces TNF-α in Purkinje neurons by activating the TNFR1-NF-κB pathway. J Neuroinflammation 17:70
Bandtlow CE, Meyer M, Lindholm D, Spranger M, Heumann R, Thoenen H (1990) Regional and cellular codistribution of interleukin 1 beta and nerve growth factor mRNA in the adult rat brain: possible relationship to the regulation of nerve growth factor synthesis. J Cell Biol 111:1701–1711
Banisadr G, Gosselin RD, Mechighel P, Kitabgi P, Rostène W, Parsadaniantz SM (2005) Highly regionalized neuronal expression of monocyte chemoattractant protein-1 (MCP-1/CCL2) in rat brain: evidence for its colocalization with neurotransmitters and neuropeptides. J Comp Neurol 489:275–292
Bellamy TC (2006) Interactions between Purkinje neurones and Bergmann glia. Cerebellum (London, England) 5:116–126
Cerrato V (2020) Cerebellar astrocytes: much more than passive by standers in ataxia pathophysiology. J Clin Med 9:757
Chiang CS, Powell HC, Gold LH, Samimi A, Campbell IL (1996) Macrophage/microglial-mediated primary demyelination and motor disease induced by the central nervous system production of interleukin-3 in transgenic mice. J Clin Invest 97:1512–1524
Chibowska K, Korbecki J, Gutowska I, Metryka E, Tarnowski M, Goschorska M, Barczak K, Chlubek D, Baranowska-Bosiacka I (2020) Pre- and neonatal exposure to lead (pb) induces neuroinflammation in the forebrain cortex, hippocampus and cerebellum of rat pups. Int J Mol Sci 21:1083
Colonna M, Butovsky O (2017) Microglia function in the central nervous system during health and neurodegeneration. Annu Rev Immunol 35:441–468
Conroy SM, Nguyen V, Quina LA, Blakely-Gonzales P, Ur C, Netzeband JG, Prieto AL, Gruol DL (2004) Interleukin-6 produces neuronal loss in develo** cerebellar granule neuron cultures. J Neuroimmunol 155:43–54
Cowell RM, Silverstein FS (2003) Developmental changes in the expression of chemokine receptor CCR1 in the rat cerebellum. J Comp Neurol 457:7–23
Cunningham C, Dunne A, Lopez-Rodriguez AB (2019) Astrocytes: heterogeneous and dynamic phenotypes in neurodegeneration and innate immunity. Neuroscientist 25:455–474
Cvetanovic M, Ingram M, Orr H, Opal P (2015) Early activation of microglia and astrocytes in mouse models of spinocerebellar ataxia type 1. Neuroscience 289:289–299
Dinarello CA (2007) Historical insights into cytokines. Eur J Immunol 37(Suppl 1):S34–S45
Ferro A, Sheeler C, Rosa JG, Cvetanovic M (2019) Role of microglia in ataxias. J Mol Biol 431:1792–1804
Gadient RA, Otten UH (1997) Interleukin-6 (IL-6)—a molecule with both beneficial and destructive potentials. Prog Neurobiol 52:379–390
Gayle D, Ilyin SE, Plata-Salamán CR (1997) Interleukin-1 receptor type I mRNA levels in brain regions from male and female rats. Brain Res Bull 42:463–467
Giovannelli A, Limatola C, Ragozzino D, Mileo AM, Ruggieri A, Ciotti MT, Mercanti D, Santoni A, Eusebi F (1998) CXC chemokines interleukin-8 (IL-8) and growth-related gene product alpha (GROalpha) modulate Purkinje neuron activity in mouse cerebellum. J Neuroimmunol 92:122–132
Gómez-Nicola D, Valle-Argos B, Pita-Thomas DW, Nieto-Sampedro M (2008) Interleukin 15 expression in the CNS: blockade of its activity prevents glial activation after an inflammatory injury. Glia 56:494–505
Gruol DL, Melkonian C, Huitron-Resendiz S, Roberts AJ (2020) Alcohol alters IL-6 signal transduction in the CNS of transgenic mice with increased astrocyte expression of IL-6. Cell Mol Neurobiol 442:124–137
Hickman S, Izzy S, Sen P, Morsett L, El Khoury J (2018) Microglia in neurodegeneration. Nat Neurosci 21:1359–1369
Hoxha E, Balbo I, Miniaci MC, Tempia F (2018) Purkinje cell signaling deficits in animal models of ataxia. Front Synaptic Neurosci 10:6
Jha MK, Jo M, Kim JH, Suk K (2019) Microglia-astrocyte crosstalk: an intimate molecular conversation. Neuroscientist 25:227–240
Kana V et al (2019) CSF-1 controls cerebellar microglia and is required for motor function and social interaction. J Exp Med 216:2265–2281
Kaur C, Sivakumar V, Zou Z, Ling EA (2014) Microglia-derived proinflammatory cytokines tumor necrosis factor-alpha and interleukin-1beta induce Purkinje neuronal apoptosis via their receptors in hypoxic neonatal rat brain. Brain Struct Funct 219:151–170
Kempuraj D, Thangavel R, Natteru PA, Selvakumar GP, Saeed D, Zahoor H, Zaheer S, Iyer SS, Zaheer A (2016) Neuroinflammation induces neurodegeneration. J Neurol Neurosurg Spine 1:1003
Lawson LJ, Perry VH, Dri P, Gordon S (1990) Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience 39:151–170
Ma SH, Li B, Huang HW, Peng YP, Qiu YH (2012) Interleukin-6 inhibits L-type calcium channel activity of cultured cerebellar granule neurons. J Physiol Sci 62:385–392
Mandolesi G, Musella A, Gentile A, Grasselli G, Haji N, Sepman H, Fresegna D, Bullitta S, De Vito F, Musumeci G, Di Sanza C, Strata P, Centonze D (2013) Interleukin-1β alters glutamate transmission at Purkinje cell synapses in a mouse model of multiple sclerosis. J Neurosci 33:12105–12121
Marcián V, Filip P, Bareš M, Brázdil M (2016) Cerebellar dysfunction and ataxia in patients with epilepsy: coincidence, consequence, or cause? Tremor Other Hyperkinet Mov (N Y) 6:376
Meera P, Pulst SM, Otis TS (2016) Cellular and circuit mechanisms underlying spinocerebellar ataxias. J Physiol 594:4653–4660
Meng SZ, Oka A, Takashima S (1999) Developmental expression of monocyte chemoattractant protein-1 in the human cerebellum and brainstem. Brain and Development 21:30–35
Motoki K, Kishi H, Hori E, Tajiri K, Nishijo H, Muraguchi A (2009) The direct excitatory effect of IL-1beta on cerebellar Purkinje cell. Biochem Biophys Res Commun 379:665–668
Nakayama H, Abe M, Morimoto C, Iida T, Okabe S, Sakimura K, Hashimoto K (2018) Microglia permit climbing fiber elimination by promoting GABAergic inhibition in the develo** cerebellum. Nat Commun 9:2830
Nedergaard M, Ransom B, Goldman SA (2003) New roles for astrocytes: redefining the functional architecture of the brain. Trends Neurosci 26:523–530
Nelson TE, Campbell IL, Gruol DL (1999) Altered physiology of Purkinje neurons in cerebellar slices from transgenic mice with chronic central nervous system expression of interleukin-6. Neuroscience 89:127–136
Nelson TE, Ur CL, Gruol DL (2002) Chronic interleukin-6 exposure alters electrophysiological properties and calcium signaling in develo** cerebellar Purkinje neurons in culture. J Neurophysiol 88:475–486
Nelson TE, Netzeband JG, Gruol DL (2004) Chronic interleukin-6 exposure alters metabotropic glutamate receptor-activated calcium signalling in cerebellar Purkinje neurons. Eur J Neurosci 20:2387–2400
Oldreive CE, Doherty GH (2010) Effects of tumour necrosis factor-alpha on develo** cerebellar granule and Purkinje neurons in vitro. J Mol Neurosci 42:44–52
Ozawa PM, Ariza CB, Ishibashi CM, Fujita TC, Banin-Hirata BK, Oda JM, Watanabe MA (2016) Role of CXCL12 and CXCR4 in normal cerebellar development and medulloblastoma. Int J Cancer 138:10–13
Palomo J, Dietrich D, Martin P, Palmer G, Gabay C (2015) The interleukin (IL)-1 cytokine family—Balance between agonists and antagonists in inflammatory diseases. Cytokine 76:25–37
Park CR, Kim DK, Cho EB, You DJ, do Rego JL, Vaudry D, Sun W, Kim H, Seong JY, Hwang JI (2012) Spatiotemporal expression and functional implication of CXCL14 in the develo** mice cerebellum. Mol Cells 34:289–293
Pringle AK, Gardner CR, Walker RJ (1996) Reduction of cerebellar GABAA responses by interleukin-1 (IL-1) through an indomethacin insensitive mechanism. Neuropharmacology 35:147–152
Qiu Z, Sweeney DD, Netzeband JG, Gruol DL (1998) Chronic interleukin-6 alters NMDA receptor-mediated membrane responses and enhances neurotoxicity in develo** CNS neurons. J Neurosci 18:10445–10456
Ragozzino D (2002) CXC chemokine receptors in the central nervous system: role in cerebellar neuromodulation and development. J Neurovirol 8:559–572
Ragozzino D, Giovannelli A, Mileo AM, Limatola C, Santoni A, Eusebi F (1998) Modulation of the neurotransmitter release in rat cerebellar neurons by GRO beta. Neuroreport 9:3601–3606
Revuelta M, Scheuer T, Chew LJ, Schmitz T (2020) Glial factors regulating white matter development and pathologies of the cerebellum. Neurochem Res 45:643–655
Schobitz B, de Kloet ER, Sutanto W, Holsboer F (1993) Cellular localization of interleukin 6 mRNA and interleukin 6 receptor mRNA in rat brain. Eur J Neurosci 5:1426–1435
Schobitz B, De Kloet ER, Holsboer F (1994) Gene expression and function of interleukin 1, interleukin 6 and tumor necrosis factor in the brain. Prog Neurobiol 44:397–432
Stoessel MB, Majewska AK (2021) Little cells of the little brain: microglia in cerebellar development and function. Trends Neurosci 44:564–578
Stowell RD, Wong EL, Batchelor HN, Mendes MS, Lamantia CE, Whitelaw BS, Majewska AK (2018) Cerebellar microglia are dynamically unique and survey Purkinje neurons in vivo. Dev Neurobiol 78:627–644
Tiveron MC, Cremer H (2008) CXCL12/CXCR4 signalling in neuronal cell migration. Curr Opin Neurobiol 18:237–244
Tomita M, Holman BJ, Williams LS, Pang KC, Santoro TJ (2001) Cerebellar dysfunction is associated with overexpression of proinflammatory cytokine genes in lupus. J Neurosci Res 64:26–33
Vainchtein ID, Molofsky AV (2020) Astrocytes and microglia: in sickness and in health. Trends Neurosci 43:144–154
van Gassen KL, Netzeband JG, de Graan PN, Gruol DL (2005) The chemokine CCL2 modulates Ca2+ dynamics and electrophysiological properties of cultured cerebellar Purkinje neurons. Eur J Neurosci 21:2949–2957
Vargas DL, Nascimbene C, Krishnan C, Zimmerman AW, Pardo CA (2005) Neuroglial activation and neuroinflammation in the brain of patients with autism. Ann Neurol 57:67–81
Verkhratsky A, Nedergaard M (2018) Physiology of astroglia. Physiol Rev 98:239–389
Vilz TO, Moepps B, Engele J, Molly S, Littman DR, Schilling K (2005) The SDF-1/CXCR4 pathway and the development of the cerebellar system. Eur J Neurosci 22:1831–1839
Wicher G, Husic E, Nilsson G, Forsberg-Nilsson K (2013) Developmental expression of IL-33 in the mouse brain. Neurosci Lett 555:171–176
Wong ML, Licinio J (1994) Localization of interleukin 1 type I receptor mRNA in rat brain. Neuroimmunomodulation 1:110–115
Wright-** EC, Gutmann DH (2019) Microglia as dynamic cellular mediators of brain function. Trends Mol Med 25:967–979
Yu T, Huang H, Li HF (2010) Stromal cell-derived factor-1 promotes migration of cells from the upper rhombic lip in cerebellar development. J Neurosci Res 88:2775–2786
Zhuang JL, Wang CY, Zhou MH, Duan KZ, Mei YA (2012) TGF-β1 enhances Kv2.1 potassium channel protein expression and promotes maturation of cerebellar granule neurons. J Cell Physiol 227:297–307
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Gruol, D.L. (2023). The Cerebellar Neuroimmune System. In: Gruol, D.L., Koibuchi, N., Manto, M., Molinari, M., Schmahmann, J.D., Shen, Y. (eds) Essentials of Cerebellum and Cerebellar Disorders. Springer, Cham. https://doi.org/10.1007/978-3-031-15070-8_47
Download citation
DOI: https://doi.org/10.1007/978-3-031-15070-8_47
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-15069-2
Online ISBN: 978-3-031-15070-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)