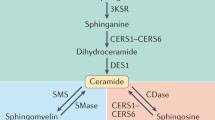
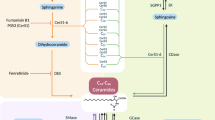
Abstract
Ceramides are components of all cell membranes, regulating immune cell function and inflammation. Metabolism-wise, ceramides increase fat use as an energy substrate instead of glucose, thus reducing insulin resistance. In parallel, mitochondrial performance is also reduced, enabling the consumption of more fat per unit of energy production, while disrupting the function of lipolysis enzymes. Increased ceramide concentrations are observed in the adipose tissue of individuals with obesity, while in mice, blocking ceramide production improved insulin sensitivity and prevented atherosclerosis and heart failure. Elevated ceramide concentrations are associated with adiposity and the development of insulin resistance, type 2 diabetes mellitus, and increased cardiovascular disease risk. Interventions with hypocaloric diets, adherence to the Mediterranean diet, low-sugar intake, substitution of unhealthy dietary fats, and supplementation with n-3 fatty acids and vitamin D can reduce ceramide concentrations, improving prognosis.
Similar content being viewed by others
Abbreviations
- C1P:
-
Ceramide 1-phosphate
- CAD:
-
Coronary artery disease
- CerS:
-
Ceramide synthetase
- CVD:
-
Cardiovascular disease
- EVOO:
-
Extra-virgin olive oil
- FFA:
-
Free fatty acids
- GLUT-4:
-
Glucose transporter type 4
- HELLP:
-
Hemolysis, elevated liver enzymes, and low platelets
- HOMA-IR:
-
Homeostatic model assessment of insulin resistance
- IR:
-
Insulin resistance
- MD:
-
Mediterranean diet
- mRNA:
-
Messenger RNA
- MUFA:
-
Monounsaturated fatty acids
- NAFLD:
-
Non-alcoholic fatty liver disease
- OA:
-
Oleic acid
- PA:
-
Palmitic acid
- PC:
-
Phosphatidylcholine
- PKB:
-
Akt kinase/protein B
- PREDIMED:
-
PREvención con DIeta MEDiterránea
- RCT:
-
Randomized controlled trial
- S1P:
-
Sphingosine 1-posphate
- SFA:
-
Saturated fatty acid
- SPT:
-
Serine palmitoyl-transferase
- T2DM:
-
Type 2 diabetes mellitus
- TAG:
-
Triacylglyceride
- TNFα:
-
Tumor necrosis factor-α
- VD:
-
Lacto-ovo-vegetarian diet
References
Adams JM, Pratipanawatr T, Berria R. Ceramide content is increased in skeletal muscle from obese insulin-resistant humans. Diabetes. 2004;53:25–31. https://doi.org/10.2337/DIABETES.53.1.25.
Blachnio-Zabielska AU, Hady HR, Markowski AR, et al. Inhibition of ceramide de novo synthesis affects adipocytokine secretion and improves systemic and adipose tissue insulin sensitivity. Int J Mol Sci. 2018;19:3995. https://doi.org/10.3390/ijms19123995.
Brozinick JT, Hawkins E, Bui HH. Plasma sphingolipids are biomarkers of metabolic syndrome in non-human primates maintained on a western-style diet. Int J Obes. 2013;37:1064–70. https://doi.org/10.1038/IJO.2012.191.
Butler TJ, Ashford D, Seymour AM. Western diet increases cardiac ceramide content in healthy and hypertrophied hearts. Nutr Metab Cardiovasc Dis. 2017;27:991–8. https://doi.org/10.1016/J.NUMECD.2017.08.007.
Charkiewicz K, Blachnio-Zabielska A, Zbucka-Kretowska M. Maternal plasma and amniotic fluid sphingolipids profiling in fetal down syndrome. PLoS One. 2015;10:1–10. https://doi.org/10.1371/journal.pone.0127732.
Chaurasia B, Kaddai VA, Lancaster GI. Adipocyte ceramides regulate subcutaneous adipose browning, inflammation, and metabolism. Cell Metab. 2016;24:820–34. https://doi.org/10.1016/J.CMET.2016.10.002.
Chaurasia B, Talbot CL, Summers SA. Adipocyte ceramides-the nexus of inflammation and metabolic disease. Front Immunol. 2020;11:576347. https://doi.org/10.3389/FIMMU.2020.576347.
Chaurasia B, Ying L, Talbot CL. Ceramides are necessary and sufficient for diet-induced impairment of thermogenic adipocytes. Mol Metab. 2021;45:101145. https://doi.org/10.1016/J.MOLMET.2020.101145.
Chen L, Dong Y, Bhagatwala J. Vitamin D3 supplementation increases long-chain ceramide levels in overweight/obese African Americans: a post-hoc analysis of a randomized controlled trial. Nutrients. 2020;12:981. https://doi.org/10.3390/NU12040981.
Chiu S, Siri-Tarino P, Bergeron N. A randomized study of the effect of replacing sugar-sweetened soda by reduced fat milk on cardiometabolic health in male adolescent soda drinkers. Nutrients. 2020;12. https://doi.org/10.3390/NU12020405.
Choromańska B, Myśliwiec P, Razak Hady H. Metabolic syndrome is associated with ceramide accumulation in visceral adipose tissue of women with morbid obesity. Obesity (Silver Spring). 2019;27:444–53. https://doi.org/10.1002/OBY.22405.
Coen PM, Menshikova EV, Distefano G. Exercise and weight loss improve muscle mitochondrial respiration, lipid partitioning, and insulin sensitivity after gastric bypass surgery. Diabetes. 2015;64:3737–50. https://doi.org/10.2337/DB15-0809.
Cuvillier O, Pirianov G, Kleuser B. Suppression of ceramide-mediated programmed cell death by sphingosine-1-phosphate. Nature. 1996;381(6585):800–3. https://doi.org/10.1038/381800a0.
de Carvalho LP, Tan SH, Ow GS. Plasma ceramides as prognostic biomarkers and their arterial and myocardial tissue correlates in acute myocardial infarction. JACC Basic Transl Sci. 2018;3:163–75. https://doi.org/10.1016/J.JACBTS.2017.12.005.
Djekic D, Shi L, Calais F. Effects of a lacto-ovo-vegetarian diet on the plasma lipidome and its association with atherosclerotic burden in patients with coronary artery disease-a randomized, open-label, cross-over study. Nutr. 2020;12:3586. https://doi.org/10.3390/NU12113586.
Dubé JJ, Amati F, Toledo FGS. Effects of weight loss and exercise on insulin resistance, and intramyocellular triacylglycerol, diacylglycerol and ceramide. Diabetologia. 2011;54:1156. https://doi.org/10.1007/S00125-011-2065-0.
Fretts AM, Jensen PN, Hoofnagle A. Plasma ceramide species are associated with diabetes risk in participants of the strong heart study. J Nutr. 2020;150:1214–22. https://doi.org/10.1093/jn/nxz259.
Fucho R, Casals N, Serra D, Herrero L. Ceramides and mitochondrial fatty acid oxidation in obesity. FASEB J. 2017;31:1263–72. https://doi.org/10.1096/FJ.201601156R.
Gault CR, Obeid LM, Hannun YA. An overview of sphingolipid metabolism: from synthesis to breakdown. In: Chalfant C, Del Poeta M, editors. Sphingolipids as signalings and regulatory molecules. New York: Springer; 2010. p. 1–23.
Górska M, Dobrzyń A, Zendzian-Piotrowska M, Namiot Z. Concentration and composition of free ceramides in human plasma. Horm Metab Res. 2002;34:466–8. https://doi.org/10.1055/s-2002-33597.
Hannun YA, Luberto C. Lipid metabolism: ceramide transfer protein adds a new dimension. Curr Biol. 2004;14:R163–5. https://doi.org/10.1016/J.CUB.2004.01.049.
Hannun YA, Obeid LM. Sphingolipids and their metabolism in physiology and disease. Nat Rev Mol Cell Biol. 2018;19:191. https://doi.org/10.1038/NRM.2017.107.
Haus JM, Kashyap SR, Kasumov T. Plasma ceramides are elevated in obese subjects with type 2 diabetes and correlate with the severity of insulin resistance. Diabetes. 2009;58:337–43. https://doi.org/10.2337/db08-1228.
Havulinna AS, Sysi-Aho M, Hilvo M. Circulating ceramides predict cardiovascular outcomes in the population-based finrisk 2002 cohort. Arterioscler Thromb Vasc Biol. 2016;36:2424–30. https://doi.org/10.1161/ATVBAHA.116.307497.
Kasumov T, Li L, Li M. Ceramide as a mediator of non-alcoholic fatty liver disease and associated atherosclerosis. PLoS One. 2015;10:1–26. https://doi.org/10.1371/journal.pone.0126910.
Kien CL, Bunn JY, Poynter ME. A lipidomics analysis of the relationship between dietary fatty acid composition and insulin sensitivity in young adults. Diabetes. 2013;62:1054–63. https://doi.org/10.2337/DB12-0363.
Kihara A, Mitsutake S, Mizutani Y, Igarashi Y. Metabolism and biological functions of two phosphorylated sphingolipids, sphingosine 1-phosphate and ceramide 1-phosphate. Prog Lipid Res. 2007;46:126–44. https://doi.org/10.1016/J.PLIPRES.2007.03.001.
Kirwan JP. Plasma ceramides target skeletal muscle in type 2 diabetes. Diabetes. 2013;62:352–4. https://doi.org/10.2337/DB12-1427.
Kitatani K, Idkowiak-Baldys J, Hannun YA. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell Signal. 2008;20:1010–8. https://doi.org/10.1016/J.CELLSIG.2007.12.006.
Koch A, Grammatikos G, Trautmann S, et al. Vitamin D supplementation enhances c18(dihydro)ceramide levels in type 2 diabetes patients. Int J Mol Sci. 2017;18:1532. https://doi.org/10.3390/IJMS18071532.
Kolak M, Westerbacka J, Velagapudi VR. Adipose tissue inflammation and increased ceramide content characterize subjects with high liver fat content independent of obesity. Diabetes. 2007;56:1960–8. https://doi.org/10.2337/DB07-0111.
Kolak M, Gertow J, Westerbacka J. Expression of ceramide-metabolising enzymes in subcutaneous and intra-abdominal human adipose tissue. Lipids Heal Dis. 2012;111(11):1–12. https://doi.org/10.1186/1476-511X-11-115.
Kurz J, Parnham MJ, Geisslinger G, Schiffmann S. Ceramides as novel disease biomarkers. Trends Mol Med. 2019;25:20–32. https://doi.org/10.1016/j.molmed.2018.10.009.
Kwon Y-J, Lee G-M, Liu K-H, Jung D-H. Effect of Korean red ginseng on plasma ceramide levels in postmenopausal women with hypercholesterolemia: a pilot randomized controlled trial. Metabolites. 2021;11:417. https://doi.org/10.3390/METABO11070417.
Laaksonen R, Ekroos K, Sysi-Aho M. Plasma ceramides predict cardiovascular death in patients with stable coronary artery disease and acute coronary syndromes beyond LDL-cholesterol. Eur Heart J. 2016;37:1967–76. https://doi.org/10.1093/EURHEARTJ/EHW148.
Lair B, Laurens C, Van Den Bosch B, Moro C. Novel insights and mechanisms of lipotoxicity-driven insulin resistance. Int J Mol Sci. 2020;21:1–27. https://doi.org/10.3390/ijms21176358.
Lankinen M, Schwab U, Erkkilä A. Fatty fish intake decreases lipids related to inflammation and insulin signaling-a lipidomics approach. PLoS One. 2009;4. https://doi.org/10.1371/JOURNAL.PONE.0005258.
Lemaitre RN, Yu C, Hoofnagle A. Circulating sphingolipids, insulin, HOMA-IR and HOMA-B: the strong heart family study running title: sphingolipids and insulin resistance markers. Diabetes. 2018;67:1663–72.
León-Aguilar LF, Croyal M, Ferchaud-Roucher V. Maternal obesity leads to long-term altered levels of plasma ceramides in the offspring as revealed by a longitudinal lipidomic study in children. Int J Obes. 2019;43:1231–43. https://doi.org/10.1038/s41366-018-0291-y.
Li Q, Wang X, Pang J. Associations between plasma ceramides and mortality in patients with coronary artery disease. Atherosclerosis. 2020a;314:77–83. https://doi.org/10.1016/J.ATHEROSCLEROSIS.2020.09.004.
Li Y, Talbot CL, Chaurasia B. Ceramides in adipose tissue. Front Endocrinol (Lausanne). 2020b;11:1–8. https://doi.org/10.3389/fendo.2020.00407.
Luukkonen PK, Sädevirta S, Zhou Y. Saturated fat is more metabolically harmful for the human liver than unsaturated fat or simple sugars. Diabetes Care. 2018;41:1732–9. https://doi.org/10.2337/DC18-0071.
Mah M, Febbraio M, Turpin-Nolan S. Circulating ceramides- are origins important for sphingolipid biomarkers and treatments? Front Endocrinol (Lausanne). 2021;0:834. https://doi.org/10.3389/FENDO.2021.684448.
Merrill AHJ. De novo sphingolipid biosynthesis: a necessary, but dangerous, pathway. J Biol Chem. 2002;277:25843–6. https://doi.org/10.1074/JBC.R200009200.
Moro K, Nagahashi M, Gabriel E. Clinical application of ceramide in cancer treatment. Breast Cancer. 2019;264(26):407–15. https://doi.org/10.1007/S12282-019-00953-8.
Nakahara K, Ohkuni A, Kitamura T. The sjögren-larsson syndrome gene encodes a hexadecenal dehydrogenase of the sphingosine 1-phosphate degradation pathway. Mol Cell. 2012;46:461–71. https://doi.org/10.1016/J.MOLCEL.2012.04.033.
Neeland IJ, Singh S, McGuire DK. Relation of plasma ceramides to visceral adiposity, insulin resistance and the development of type 2 diabetes mellitus: the Dallas heart study. Diabetologia. 2018;61:2570–9. https://doi.org/10.1007/s00125-018-4720-1.
Norris GH, Blesso CN. Dietary and endogenous sphingolipid metabolism in chronic inflammation. Nutrients. 2017;9:1180. https://doi.org/10.3390/NU9111180.
Nwabuo CC, Duncan M, Xanthakis V. Association of circulating ceramides with cardiac structure and function in the community: the Framingham heart study. J Am Heart Assoc. 2019;8. https://doi.org/10.1161/JAHA.119.013050.
Öörni K, Jauhiainen M, Kovanen PT. Why and how increased plasma ceramides predict future cardiovascular events? Atherosclerosis. 2020;314:71–3. https://doi.org/10.1016/J.ATHEROSCLEROSIS.2020.09.030.
Palomer X, Pizarro-Delgado J, Barroso E, Vázquez-Carrera M. Palmitic and oleic acid: the yin and yang of fatty acids in type 2 diabetes mellitus. Trends Endocrinol Metab. 2018;29:178–90. https://doi.org/10.1016/J.TEM.2017.11.009.
Parveen F, Bender D, Law S-H. Role of ceramidases in sphingolipid metabolism and human diseases. Cell. 2019;8. https://doi.org/10.3390/CELLS8121573.
Peterson LR, Xanthakis V, Duncan MS. Ceramide remodeling and risk of cardiovascular events and mortality. J Am Heart Assoc. 2018;7. https://doi.org/10.1161/JAHA.117.007931.
Pewzner-Jung Y, Ben-Dor S, Futerman AH. When do lasses (longevity assurance genes) become cers (ceramide synthases)?: insights into the regulation of ceramide synthesis*. J Biol Chem. 2006;281:25001–5. https://doi.org/10.1074/JBC.R600010200.
Powell DP, Hajduch E, Kular G, Hundal HS. Ceramide disables 3-phosphoinositide binding to the pleckstrin homology domain of protein kinase B (PKB)/Akt by a PKCzeta-dependent mechanism. Mol Cell Biol. 2003;23:7794–808. https://doi.org/10.1128/MCB.23.21.7794-7808.2003.
Rago D, Rasmussen MA, Lee-Sarwar KA. Fish-oil supplementation in pregnancy, child metabolomics and asthma risk. EBioMedicine. 2019;46:399–410. https://doi.org/10.1016/j.ebiom.2019.07.057. NC-ND license http://creativecommons.org/licenses/by-nc-nd/4.0/
Raichur S, Brunner B, Bielohuby M. The role of C16:0 ceramide in the development of obesity and type 2 diabetes: CerS6 inhibition as a novel therapeutic approach. Mol Metab. 2019;21:36–50. https://doi.org/10.1016/j.molmet.2018.12.008.
Reidy PT, McKenzie AI, Mahmassani Z. Skeletal muscle ceramides and relationship with insulin sensitivity after 2 weeks of simulated sedentary behaviour and recovery in healthy older adults. J Physiol. 2018;596:5217–36. https://doi.org/10.1113/JP276798.
Rosqvist F, Kullberg J, Ståhlman M. Overeating saturated fat promotes fatty liver and ceramides compared with polyunsaturated fat: a randomized trial. J Clin Endocrinol Metab. 2019;104:6207–19. https://doi.org/10.1210/JC.2019-00160.
Sokolowska E, Blachnio-Zabielska A. The role of ceramides in insulin resistance. Front Endocrinol (Lausanne). 2019;0:577. https://doi.org/10.3389/FENDO.2019.00577.
Summers SA. Ceramides: nutrient signals that drive hepatosteatosis. J Lipid Atheroscler. 2020;9:50–65.
Summers SA, Chaurasia B, Holland WL. Metabolic messengers: ceramides. Nat Metab. 2019;1:1051–8. https://doi.org/10.1038/S42255-019-0134-8.
Tuccinardi D, Di Mauro A, Lattanzi G, et al. An extra virgin olive oil-enriched chocolate spread positively modulates insulin-resistance markers compared with a palm oil-enriched one in healthy young adults: a double-blind, cross-over, randomized controlled trial. Diabetes Metab Res Rev. 2021;e3492. https://doi.org/10.1002/DMRR.3492.
Turpin-Nolan SM, Hammerschmidt P, Chen W, et al. CerS1-derived C18:0 ceramide in skeletal muscle promotes obesity-induced insulin resistance. Cell Rep. 2019;26:1–10.e7. https://doi.org/10.1016/J.CELREP.2018.12.031.
Walker M, Xanthakis V, Ma J, et al. A Mediterranean style diet is favorably associated with concentrations of circulating ceramides and ceramide ratios in the Framingham Offspring cohort (P18-048-19). Curr Dev Nutr. 2019;3. https://doi.org/10.1093/CDN/NZZ039.P18-048-19.
Wang DD, Toledo E, Hruby A, et al. Plasma ceramides, Mediterranean diet, and incident cardiovascular disease in the PREDIMED trial. Circulation. 2017;135:2028–40. https://doi.org/10.1161/CIRCULATIONAHA.116.024261.
Watt M, Boon J, Hoy A, et al. Plasma ceramides are elevated in patients with type 2 diabetes and promote skeletal muscle insulin resistance and inflammation – the physiological society. Proc Physiol. 2012;27:C97.
Zabielski P, Błachnio-Zabielska AU, Wójcik B, et al. Effect of plasma free fatty acid supply on the rate of ceramide synthesis in different muscle types in the rat. PLoS One. 2017;12:e0187136. https://doi.org/10.1371/JOURNAL.PONE.0187136.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Alexandropoulou, I., Lantzanaki-Syrpou, M., Grammatikopoulou, M.G., Goulis, D.G. (2022). Ceramides as Dietary Biomarkers. In: Patel, V.B., Preedy, V.R. (eds) Biomarkers in Nutrition . Biomarkers in Disease: Methods, Discoveries and Applications. Springer, Cham. https://doi.org/10.1007/978-3-031-07389-2_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-07389-2_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-07388-5
Online ISBN: 978-3-031-07389-2
eBook Packages: Biomedical and Life SciencesReference Module Biomedical and Life Sciences