Abstract
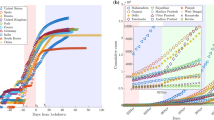
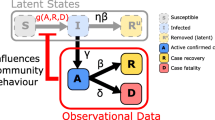
Mathematical models have been widely used to understand the dynamics of the ongoing coronavirus disease 2019 (COVID-19) pandemic as well as to predict future trends and assess intervention strategies. The asynchronicity of infection patterns during this pandemic illustrates the need for models that can capture dynamics beyond a single-peak trajectory to forecast the worldwide spread and for the spread within nations and within other sub-regions at various geographic scales. Here, we demonstrate a five-parameter sub-epidemic wave modeling framework that provides a simple characterization of unfolding trajectories of COVID-19 epidemics that are progressing across the world at different spatial scales. We calibrate the model to daily reported COVID-19 incidence data to generate six sequential weekly forecasts for five European countries and five hotspot states within the United States. The sub-epidemic approach captures the rise to an initial peak followed by a wide range of post-peak behavior, ranging from a typical decline to a steady incidence level to repeated small waves for sub-epidemic outbreaks. We show that the sub-epidemic model outperforms a three-parameter Richards model, in terms of calibration and forecasting performance, and yields excellent short- and intermediate-term forecasts that are not attainable with other single-peak transmission models of similar complexity. Overall, this approach predicts that a relaxation of social distancing measures would result in continuing sub-epidemics and ongoing endemic transmission. We illustrate how this view of the epidemic could help data scientists and policymakers better understand and predict the underlying transmission dynamics of COVID-19, as early detection of potential sub-epidemics can inform model-based decisions for tighter distancing controls.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jewell NP, Lewnard JA, Jewell BL. Caution Warranted: Using the Institute for Health Metrics and Evaluation Model for Predicting the Course of the COVID-19 Pandemic. Annals of Internal Medicine 2020;173:xxx-xxx. https://doi.org/107326/M20-1565.
Blower SM, McLean AR, Porco TC, Small PM, Hopewell PC, Sanchez MA, et al. The intrinsic transmission dynamics of tuberculosis epidemics [see comments]. Nature Medicine. 1995;95(8):815-21.
Garnett GP. The geographical and temporal evolution of sexually transmitted disease epidemics. Sexually Transmitted Infections. 2002;78(Suppl 1):14-9.
Rothenberg R, Voigt R. Epidemiologic Aspects of Control of Penicillinase-Producing Neisseria gonorrhoeae. Sexually Transmitted Diseases. 1988;15(4):211-6.
Rothenberg R, Dai D, Adams MA, Heath JW. The HIV endemic: maintaining disease transmission in at-risk urban areas. Sexually Transmitted Diseases. 2017;44(2):71-8.
Chowell G, Tariq A, Hyman JM. A novel sub-epidemic modeling framework for short-term forecasting epidemic waves. BMC Medicine. 2019;17(1):164.
Wang XS, Wu J, Yang Y. Richards model revisited: validation by and application to infection dynamics. Journal of theoretical biology. 2012;313:12-9.
Nossiter A. Male reports first death from Ebola. New York Times [2014 Oct 24]. Available from: http://www.nytimes.com/2014/10/25/world/africa/mali-reports-first-death-from-ebola.html (accessed on 2015 Jan 13). 2014.
Onishi N, Santora M. Ebola patient in Dallas lied on screening form, Liberian airport official says. New York Times [2014 Oct 2]. Available from: http://www.nytimes.com/2014/10/03/world/africa/dallas-ebola-patient-thomas-duncan-airport-screening.html (accessed on 2015 Feb 28). 2014.
Onishi N. Last known Ebola patient in Liberia is discharged. New York Times [2015 Mar 5]. Available from: http://www.nytimes.com/2015/03/06/world/africa/last-ebola-patient-in-liberia-beatrice-yardolo-discharged-from-treatment.html?ref=topics&_r=0 (accessed on 2015 Mar 6). 2015.
The COVID Tracking Project [Available from: https://covidtracking.com/data.
Chowell G. Fitting dynamic models to epidemic outbreaks with quantified uncertainty: A Primer for parameter uncertainty, identifiability, and forecasts. Infect Dis Model. 2017;2(3):379-98.
Banks HT, Hu S, Thompson WC. Modeling and inverse problems in the presence of uncertainty: CRC Press; 2014.
Myung IJ. Tutorial on maximum likelihood estimation. Journal of Mathematical Pyschology; 2003. p. 90-100.
Kashin K. Statistical Inference: Maximum Likelihood Estimation. 2014.
Roosa K, Luo R, Chowell G. Comparative assessment of parameter estimation methods in the presence of overdispersion: a simulation study. Mathematical biosciences and engineering : MBE. 2019;16(5):4299-313.
Yan P, Chowell G. Quantitative methods for investigating infectious disease outbreaks. Switzerland: Springer Nature; 2019.
Friedman J, Hastie T, Tibshirani R. The Elements of Statistical Learning: Data Mining, Inference, and Prediction. New York, NY.: Springer-Verlag New York; 2009.
Smirnova A, Chowell G. A primer on stable parameter estimation and forecasting in epidemiology by a problem-oriented regularized least squares algorithm. Infect Dis Model. 2017;2(2):268-75.
Viboud C, Simonsen L, Chowell G. A generalized-growth model to characterize the early ascending phase of infectious disease outbreaks. Epidemics. 2016;15:27-37.
Chowell G. Fitting dynamic models to epidemic outbreaks with quantified uncertainty: A primer for parameter uncertainty, identifiability, and forecasts. Infectious Disease Modelling. 2017;2(3):379-98.
Chowell G, Sattenspiel L, Bansal S, Viboud C. Mathematical models to characterize early epidemic growth: A review. Physics of Life Reviews. 2016;18:66-97.
Roosa K, Lee Y, Luo R, Kirpich A, Rothenberg R, Hyman JM, et al. Real-time forecasts of the COVID-19 epidemic in China from February 5th to February 24th, 2020. Infect Dis Model. 2020;5:256-63.
Roosa K, Lee Y, Luo R, Kirpich A, Rothenberg R, Hyman JM, et al. Short-term Forecasts of the COVID-19 Epidemic in Guangdong and Zhejiang, China: February 13-23, 2020. J Clin Med. 2020;9(2).
Pell B, Kuang Y, Viboud C, Chowell G. Using phenomenological models for forecasting the 2015 Ebola challenge. Epidemics. 2018;22:62-70.
Shanafelt DW, Jones G, Lima M, Perrings C, Chowell G. Forecasting the 2001 Foot-and-Mouth Disease Epidemic in the UK. Ecohealth. 2018;15(2):338-47.
Chowell G, Hincapie-Palacio D, Ospina J, Pell B, Tariq A, Dahal S, et al. Using Phenomenological Models to Characterize Transmissibility and Forecast Patterns and Final Burden of Zika Epidemics. PLoS currents. 2016;8:ecurrents.outbreaks.f14b2217c902f453d9320a43a35b583.
Shanafelt DW, Jones G, Lima M, Perrings C, Chowell G. Forecasting the 2001 Foot-and-Mouth Disease Epidemic in the UK. EcoHealth. 2017.
Richards FJ. A Flexible Growth Function for Empirical Use. Journal of Experimental Botany. 1959;10(2):290-301.
Granovetter MS. The strength of weak ties. American Journal of Sociology. 1973;78(6):1360-80.
Cheng VCC, Wong S-C, To KKW, Ho PL, Yuen K-Y. Preparedness and proactive infection control measures against the emerging Wuhan coronavirus pneumonia in China. Journal of Hospital Infection. 2020.
Pan J, Yao Y, Liu Z, Li M, Wang Y, Dong W, et al. Effectiveness of control strategies for Coronavirus Disease 2019: a SEIR dynamic modeling study. https://doi.org/10.1101/2020.02.19.200253872020.
Prem K, Liu Y, Russell T, Kucharski AJ, Eggo RM, Davies N, et al. The effect of control strategies that reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China. 2020. https://doi.org/10.1101/2020.03.09.20033050.
Lai S, Ruktanonchai NW, Zhou L, Prosper O, Luo W, Floyd JR, et al. Effect of non-pharmaceutical interventions for containing the COVID-19 outbreak: an observational and modelling study. 2020. https://doi.org/10.1101/2020.03.03.20029843.
Chowell G, Ammon CE, Hengartner NW, Hyman JM. Transmission dynamics of the great influenza pandemic of 1918 in Geneva, Switzerland: Assessing the effects of hypothetical interventions. Journal of theoretical biology. 2006;241(2):193-204.
Chowell G, Tariq A, Hyman JM. A novel sub-epidemic modeling framework for short-term forecasting epidemic waves: Datasets and fitting code. figshare. Available from: https://doi.org/10.6084/m9.figshare.8867882. 2019.
Hsieh YH, Cheng YS. Real-time forecast of multiphase outbreak. Emerging infectious diseases. 2006;12(1):122-7.
Gneiting T, Raftery AE. Strictly proper scoring rules, prediction, and estimation. J Am Stat Assoc. 2007;102(477):359-78.
Kuhn M, Johnson K. Applied predictive modeling: New York: Springer; 2013.
M4Competition. Competitor’s Guide: Prizes and Rules. Available from: https://www.m4.unic.ac.cy/wp-content/uploads/2018/03/M4-Competitors-Guide.pdf (accessed 04/01/2019) [
Funk S, Camacho A, Kucharski AJ, Lowe R, Eggo RM, Edmunds WJ. Assessing the performance of real-time epidemic forecasts: A case study of Ebola in the Western Area region of Sierra Leone, 2014-15. PLoS computational biology. 2019;15(2):e1006785.
COVID-19 coronavirus / cases [Internet]. 2020. Available from: https://www.worldometers.info/coronavirus/coronavirus-cases/.
Roosa K, Tariq A, Yan P, Hyman JM, Chowell G. Multi-model forecasts of the ongoing Ebola epidemic in the Democratic Republic of Congo, March-October 2019. Journal of the Royal Society, Interface/the Royal Society. 2020;17(169):20200447.
Chowell G, Viboud C, Simonsen L, Merler S, Vespignani A. Perspectives on model forecasts of the 2014-2015 Ebola epidemic in West Africa: lessons and the way forward. BMC medicine. 2017;15(1):42.
Viboud C, Sun K, Gaffey R, Ajelli M, Fumanelli L, Merler S, et al. The RAPIDD ebola forecasting challenge: Synthesis and lessons learnt. Epidemics. 2018;22:13-21. Worldometer. (Accessed May 11, 2020, at https://www.worldometers.info/coronavirus/.)
Acknowledgments
Funding: GC was supported by grants NSF 1414374 as part of the joint NSF-National Institutes of Health NIH-United States Department of Agriculture USDA Ecology and Evolution of Infectious Diseases program; UK Biotechnology and Biological Sciences Research Council [grant BB/M008894/1] and RAPID NSF 2026797.
Author contributions: GC conceived the study. KR and AT contributed to data analysis. All authors contributed to the interpretation of the results. GC and RR wrote the first draft of the manuscript. All authors contributed to writing subsequent drafts of the manuscript. All authors read and approved the final manuscript.
Competing interests: Authors declare no competing interests.
Data and materials availability: All data are publicly available.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Appendix
Appendix
The best fit of the sub-epidemic model to the COVID-19 epidemic in Italy. The sub-epidemic wave model successfully captures the multimodal pattern of the COVID-19 epidemic. Further, parameter estimates are well identified, as indicated by their relatively narrow confidence intervals. The top panels display the empirical distribution of r, p, K, and q. Bottom panels show the model fit (left), the sub-epidemic profile (center), and the residuals (right). Black circles correspond to the data points. The best model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. Cyan curves are the associated uncertainty from individual bootstrapped curves. Three hundred realizations of the sub-epidemic waves are plotted using different colors
The best fit of the sub-epidemic model to the COVID-19 epidemic in France. The sub-epidemic wave model successfully captures the multimodal pattern of the COVID-19 epidemic. Further, parameter estimates are well identified, as indicated by their relatively narrow confidence intervals. The top panels display the empirical distribution of r, p, K, and q. Bottom panels show the model fit (left), the sub-epidemic profile (center), and the residuals (right). Black circles correspond to the data points. The best model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. Cyan curves are the associated uncertainty from individual bootstrapped curves. Three hundred realizations of the sub-epidemic waves are plotted using different colors
The best fit of the sub-epidemic model to the COVID-19 epidemic in the United Kingdom. The sub-epidemic wave model successfully captures the multimodal pattern of the COVID-19 epidemic. Further, parameter estimates are well identified, as indicated by their relatively narrow confidence intervals. The top panels display the empirical distribution of r, p, K, and q. Bottom panels show the model fit (left), the sub-epidemic profile (center), and the residuals (right). Black circles correspond to the data points. The best model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. Cyan curves are the associated uncertainty from individual bootstrapped curves. Three hundred realizations of the sub-epidemic waves are plotted using different colors
The best fit of the sub-epidemic model to the COVID-19 epidemic in Louisiana, USA. The sub-epidemic wave model successfully captures the multimodal pattern of the COVID-19 epidemic. Further, parameter estimates are well identified, as indicated by their relatively narrow confidence intervals. The top panels display the empirical distribution of r, p, K, and q. Bottom panels show the model fit (left), the sub-epidemic profile (center), and the residuals (right). Black circles correspond to the data points. The best model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. Cyan curves are the associated uncertainty from individual bootstrapped curves. Three hundred realizations of the sub-epidemic waves are plotted using different colors
The best fit of the sub-epidemic model to the COVID-19 epidemic in Georgia, USA. The sub-epidemic wave model successfully captures the multimodal pattern of the COVID-19 epidemic. Further, parameter estimates are well identified, as indicated by their relatively narrow confidence intervals. The top panels display the empirical distribution of r, p, K, and q. Bottom panels show the model fit (left), the sub-epidemic profile (center), and the residuals (right). Black circles correspond to the data points. The best model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. Cyan curves are the associated uncertainty from individual bootstrapped curves. Three hundred realizations of the sub-epidemic waves are plotted using different colors
The best fit of the sub-epidemic model to the COVID-19 epidemic in Arizona, USA. The sub-epidemic wave model successfully captures the multimodal pattern of the COVID-19 epidemic. Further, parameter estimates are well identified, as indicated by their relatively narrow confidence intervals. The top panels display the empirical distribution of r, p, K, and q. Bottom panels show the model fit (left), the sub-epidemic profile (center), and the residuals (right). Black circles correspond to the data points. The best model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. Cyan curves are the associated uncertainty from individual bootstrapped curves. Three hundred realizations of the sub-epidemic waves are plotted using different colors
The best fit of the sub-epidemic model to the COVID-19 epidemic in Washington. The sub-epidemic wave model successfully captures the multimodal pattern of the COVID-19 epidemic. Further, parameter estimates are well identified, as indicated by their relatively narrow confidence intervals. The top panels display the empirical distribution of r, p, K, and q. Bottom panels show the model fit (left), the sub-epidemic profile (center), and the residuals (right). Black circles correspond to the data points. The best model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. Cyan curves are the associated uncertainty from individual bootstrapped curves. Three hundred realizations of the sub-epidemic waves are plotted using different color
The sub-epidemic decline function across countries and USA states based on results presented in Table 1
Mean performance of the sub-epidemic wave and the Richards models in 2–20 day ahead forecasts conducted during the epidemic in Spain. The sub-epidemic model outperformed the Richards model across all metrics and forecasting horizons, but the MSE and MAE reached similar values at longer forecasting horizons
Mean performance of the sub-epidemic wave and the Richards models in 2–20 day ahead forecasts conducted during the epidemic in the UK. The sub-epidemic model outperformed the Richards model across all metrics and forecasting horizons except for 2-day ahead forecasts for which the Richards model reached somewhat better performance
Mean performance of the sub-epidemic wave and the Richards models in 2–20 day ahead forecasts conducted during the epidemic in New York. The sub-epidemic model outperformed the Richards model across all forecasting horizons based on the PI Coverage and the MIS except for 2-day ahead forecasts. However, the Richards model more frequently outperformed the sub-epidemic wave model based on the MAE and MSE
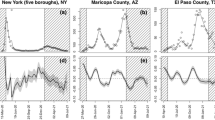
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in the USA. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in Italy. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in France. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in Spain. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in the UK. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in New York State. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sub-epidemic profiles of the sequential 20-day ahead forecasts for the COVID-19 epidemic in New York. Different colors represent different sub-epidemics of the epidemic wave profile. The aggregated trajectories are shown in gray and black circles correspond to the data points. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in Louisiana. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in Georgia. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in Arizona. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Sequential 20-day ahead forecasts of the sub-epidemic wave model for the COVID-19 epidemic in Washington. Black circles correspond to the data points. The model fit (solid red line) and 95% prediction interval (dashed red lines) are also shown. The vertical line separates the calibration period (left) from the forecasting period (right). The sequential forecasts were conducted on March 30, April 6, April 13, April 20, April 27, and May 4, 2020
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Chowell, G., Rothenberg, R., Roosa, K., Tariq, A., Hyman, J.M., Luo, R. (2022). Sub-epidemic Model Forecasts During the First Wave of the COVID-19 Pandemic in the USA and European Hotspots. In: Murty, V.K., Wu, J. (eds) Mathematics of Public Health. Fields Institute Communications, vol 85. Springer, Cham. https://doi.org/10.1007/978-3-030-85053-1_5
Download citation
DOI: https://doi.org/10.1007/978-3-030-85053-1_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-85052-4
Online ISBN: 978-3-030-85053-1
eBook Packages: Mathematics and StatisticsMathematics and Statistics (R0)