Abstract
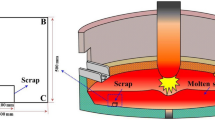
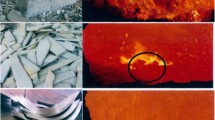
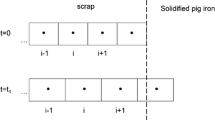
The melting kinetics of scrap is a limiting factor in controlling the temperature trajectory and scrap ratio of BOF process as well as the energy consumption and productivity of EAF steelmaking. Prediction of the melting rate of scrap, the mass transfer coefficient of carbon, and heat transfer coefficient between the melt and scrap can provide a theoretical basis for the process modeling of BOF and EAF. In this paper, the interface between melt and scrap in melting process is analyzed, and the moving boundary layer concept is optimized. Through Fick’s law and Fourier equation state, combined with the heat and mass balance of moving interface , the theoretical analysis model of scrap melting is established and programmed. The effects of bath temperature on the melting behavior of scrap in the liquid melt are studied. The results show that as the bath temperature increases, the formation time and maximum thickness of solidified layer decrease; furthermore, the time to reach the final stable melting rate and the initial heat transfer coefficient decreases. However, the final stable melting rate increases; moreover, the final stable heat transfer coefficient and mass transfer coefficient initially increase and then decrease.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- C b :
-
Carbon content of liquid melt (wt%)
- C i :
-
Carbon concentration corresponding to the liquidus at Tm (wt%)
- C sc :
-
Carbon content of the parent scrap (wt%)
- C s :
-
Carbon concentration corresponding to the solidus at Tm (wt%)
- C P :
-
The specific heat of the scrap (J/kg/K)
- D :
-
The diffusion coefficient for mass transfer inside the solid scrap (m2/s)
- D C :
-
The diffusion coefficient for mass transfer in the liquid melt (m2/s)
- h :
-
The heat transfer coefficient of the moving boundary (W/m2/K)
- h 0 :
-
The heat transfer coefficient for non-moving boundary (W/m2/K)
- k :
-
The thermal conductivity of liquid melt (W/m/K)
- R :
-
Scrap characteristic length (m)
- v :
-
The moving velocity of the interface /melting rate of scrap (m/s)
- ρ :
-
Mass density of the solid scrap (kg/m3)
- T b :
-
Bath temperature (K)
- T m :
-
Interface temperature (K)
- T 1 :
-
Center line temperature of scrap (K)
- ∆Hf:
-
The latent heat of melting (J/kg)
- β :
-
The mass transfer coefficient for the moving boundary (m/s)
- β 0 :
-
The mass transfer coefficient for non-moving boundary (m/s)
References
Sun H, Liu Y, Lin C, Lu M (2015) Experimental observation of spherical scrap melting in hot metal. Paper presented at the 6th international congress on the science and technology of steelmaking. Bei**g
Kruskopf A, Holappa LE (2018) Scrap melting model for steel converter founded on interfacial solid/liquid phenomena. Metallu Res Technol 115(2):1–7
Shukla AK, Deo B (2006) Coupled heat and mass transfer approach to simulate the scrap dissolution in steelmaking process. Paper presented at international symposium for research scholars on metallurgy, materials science and engineering. Chennai
Li JH (2007) Kinetics of steel scrap melting in liquid steel bath in an electric arc furnace. PhD thesis, McMaster University
Zhang YJ, Barr PV, Meadowcroft TR (2008) Continuous scrap melting in a short rotary furnace. Miner Eng 21(2):178–189
Dong K, Zhu R, Liu WJ (2015) Simplified calculation kinetic model for solid metal melting and decarburization process. High Temp Mater Process 34(5):447–456
Fruehan RJ (1998) The making, sha**, and treating of steel: steelmaking and refining volume. AISE Steel Foundation, Pittsburgh, pp 525–548
Kruskopf A (2015) A model for scrap melting in steel converter. Metall Mater Trans B 46(3):1195–1206
Li JH, Brooks G, Provatas N (2005) Kinetics of scrap melting in liquid steel. Metall Mater Trans B 36(2):293–302
Li JH, Provatas N (2008) Kinetics of scrap melting in liquid steel: multipiece scrap melting. Metall Mater Trans B 39(2):268–279
Li JH, Provatas N, Irons GA (2008) Modeling of late melting of scrap in an EAF. Iron Steel Technol 5(8):216–223
Shukla AK, Deo B, Robertson DGC (2013) Scrap dissolution in molten iron containing carbon for the case of coupled heat and mass transfer control. Metall Mater Trans B 44(6):1407–1427
Kruskopf A, Louhenkilpi S (2015) 1-Dimensional scrap melting model for steel converter (BOF). Paper presented at METEC and 2nd ESTAD. Dusseldorf
Sethi G, Shukla AK, Das PC et al (2004) Theoretical aspects of scrap dissolution in oxygen steelmaking converters. Paper presented at AISTech 2004—iron and steel technology conference proceedings. Nashville, TN
Sismanis PG, Argyropoulos SA (1988) Convective heat-transfer measurements in liquid metals under different fluid flow conditions. Metall Trans B 19(6):859–870
Li JH, Geoffery B, Provatas N (2004) Phase-field modeling of steel scrap melting in a liquid steel bath. Paper presented at AISTech 2004—iron and steel technology conference proceedings. Nashville, TN
Shukla AK, Deo B, Robertson DGC (2013) Role of air gap in scrap dissolution process. Metall Mater Trans B 44(6):1398–1406
Wanibe Y, Takai S, Fujisawa T et al (1982) Temperature-dependency of interdiffusion in molten Fe–C alloy. Trans Iron Steel Inst Jpn 22(7):560–565
Szekely J, Chuang YK, Hlinka JW (1972) The melting and dissolution of low-carbon steels in iron–carbon melts. Metall Trans 3(11):2825–2833
Chuang YK, Szekely J (1971) On the use of green’s functions for solving melting or solidification problems. Int J Heat Mass Transf 14(9):1285–1294
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 The Minerals, Metals & Materials Society
About this paper
Cite this paper
Liu, M., Ma, G., Zhang, X. (2020). Kinetics of Scrap Melting in Iron–Carbon Bath. In: TMS 2020 149th Annual Meeting & Exhibition Supplemental Proceedings. The Minerals, Metals & Materials Series. Springer, Cham. https://doi.org/10.1007/978-3-030-36296-6_98
Download citation
DOI: https://doi.org/10.1007/978-3-030-36296-6_98
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-36295-9
Online ISBN: 978-3-030-36296-6
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)