Abstract
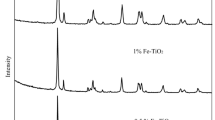
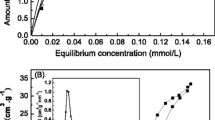
Different series of iron supported catalysts containing 10wt.% of iron, as oxide, on TiO2 and A12O3 have been prepared by the sol gel method and the traditional method based on the impregnation of the support with the metal precursor on commercial and sol-gel supports. The calcined solids were characterised by evaluating the specific surface area and porosity by nitrogen adsorption at 77 K, TPR, TPD of ammonia, X-ray diffraction, evaluation of band gap and isoelectric point by electrophoretic migration. Fe3+ ions in two situations were found, highly dispersed in which Fe3+ ions were placed in the network of the support and large Fe2O3 crystals were located on the surface catalysts. The catalytic activity of the prepared catalysts was measured in a batch reactor using ozone/UV as oxidizing agent. The catalysts displayed significant differences in initial reaction rate being the iron titania systems more active, specially when a higher extent of surface iron oxide species is present. After two hours on stream the conversion levels are similar in all the studied systems.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
O. Legrini, E. Oliveros and A.M. Braun, Chem. Rev., 93 (1993) 671
D. Ollis. Comparative aspects of advanced oxidation processes. Emerging Technologies in Waste Management II, ACS Symposium Series 518. Washington, DC, 1993. P 18–34.
E. Chammaro, A. Marco, J. Prado, S. Esplugas. Química & Industria. Sociedad Chilen de Química 1/2 (1996) 28.
W.H. Glaze and J-W Kang, Ind. Eng. Chem. Res. 28 (1989) 1573
D.F. Ollis, E. Pellizzetti and N. Serpone. Environ. Sci. Technol. 25 (1991) 1523
D. Bhatkhande, V. Pangarkar and A. Beenackers. J. Chem. Technol. Biotechnol 77 (2001) 102.
A.I. Lisenbigler, I. Guangquan and J.T. Yates Jr. Chem. Rev. 95 (1995) 735
W.H. Glaze, J.W Kang and Chapin D.H., Ozone Sci. Eng. 9 (1987) 335
G.R. Peyton, W.H. Glaze, Environ. Sci. Technol. 22 (1988) 761
J. Ma and N. Graham. Ozone Sci. Eng. 19 (1997) 227
R. Andreozzi, R. Marotta and R. Sanchirico. J. Chem. Technol. Biotechnol. 75 (2000) 59
R. Andreozzi, V. Caprio, R. Marotta and V. Tufano. Wat. Res. 35(1) (2001) 109
R. Gratia, S. Cortes, J. Sarasa P. Ormad, and J.L. Ovelleiro. Wat. Res. 34(5) (2000) 1525.
J.W. Chen, C. Hui, T. Keller and G. Smith. Water 73 (1976) 206
R. Capasso, G. Cristinzio, A. Evidente and F. Scognamiglio. Phytochemistry. 31(12) (1992) 4125
X. Fu, W. A. Zeltner, Q. Yang and M.A. Anderson, J.Catal., 168 (1997) 482.
G. Pecchi, P. Reyes, P. Sanhueza, J. Villaseñor, Chemosphere,43 (2001) 141.
P. Reyes, A. Figueroa, G. Pecchi and J.L.G.Fierro, Catal Today, 62 (2000) 209.
R.D. González, T. López and R. Gómez, Catal. Today, 35 (1997) 293.
E. E. Unmuth, L.H. Schwartz and J. B. Butt, J. Catal 63 (1980) 404.
X. Bokhimi, A. Morales, O. Novaro, E. Sánchez and R. Gómez. J. Mater Res. 10 (1995) 2788.
G. H. Haller and D. E. Resasco, Adv. Catal. 36 (1989) 173–235
J. Villaseñor, P. Reyes and G. Pecchi, J. Chem. Tech. Biotechnol., 72 (1998) 105–110)
P. V. Kamat, Surfaces Chem. Rev., 93 (1993) 227.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2003 Springer Science+Business Media New York
About this chapter
Cite this chapter
Pecchi, G., Reyes, P., Villaseñor, J. (2003). Phenol Degradation and Photodegradation Using O3/UV by Fe2O3 Supported on A12O3 and TiO2 prepared by Sol-Gel Method. In: López, T.M., Avnir, D., Aegerter, M. (eds) Emerging Fields in Sol-Gel Science and Technology. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-0449-8_5
Download citation
DOI: https://doi.org/10.1007/978-1-4615-0449-8_5
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4020-7458-5
Online ISBN: 978-1-4615-0449-8
eBook Packages: Springer Book Archive