Abstract
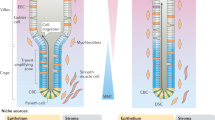
Organoid cultures have been developed to model intestinal stem cell (ISC) function in self-renewal and differentiation. Upon differentiation, the first fate decision for ISC and early progenitors to make is between secretory (Paneth cell, goblet cell, enteroendocrine cell, or tuft cell) and absorptive (enterocyte and M cell) lineages. Using genetic and pharmacological approaches, in vivo studies in the past decade have revealed that Notch signaling functions as a binary switch for the secretory vs. absorptive lineage decision in adult intestine. Recent breakthroughs in organoid-based assays enable real-time observation of smaller-scale and higher-throughput experiments in vitro, which have begun contributing to new understandings of mechanistic principles underlying intestinal differentiation. In this chapter, we summarize the in vivo and in vitro tools for modulating Notch signaling and assess its impact on intestinal cell fate. We also provide example protocols of how to use intestinal organoids as functional assays to study Notch activity in intestinal lineage decisions.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Barker N, van Oudenaarden A, Clevers H (2012) Identifying the stem cell of the intestinal crypt: strategies and pitfalls. Cell Stem Cell 11(4):452–460. https://doi.org/10.1016/j.stem.2012.09.009
Pellegrinet L, Rodilla V, Liu Z, Chen S, Koch U, Espinosa L, Kaestner KH, Kopan R, Lewis J, Radtke F (2011) Dll1- and dll4-mediated notch signaling are required for homeostasis of intestinal stem cells. Gastroenterology 140(4):1230–1240. e1231–1237. https://doi.org/10.1053/j.gastro.2011.01.005
Demitrack ES, Gifford GB, Keeley TM, Carulli AJ, VanDussen KL, Thomas D, Giordano TJ, Liu Z, Kopan R, Samuelson LC (2015) Notch signaling regulates gastric antral LGR5 stem cell function. EMBO J 34(20):2522–2536. https://doi.org/10.15252/embj.201490583
Tsai YH, VanDussen KL, Sawey ET, Wade AW, Kasper C, Rakshit S, Bhatt RG, Stoeck A, Maillard I, Crawford HC, Samuelson LC, Dempsey PJ (2014) ADAM10 regulates Notch function in intestinal stem cells of mice. Gastroenterology 147(4):822–834 e813. https://doi.org/10.1053/j.gastro.2014.07.003
van Es JH, van Gijn ME, Riccio O, van den Born M, Vooijs M, Begthel H, Cozijnsen M, Robine S, Winton DJ, Radtke F, Clevers H (2005) Notch/gamma-secretase inhibition turns proliferative cells in intestinal crypts and adenomas into goblet cells. Nature 435(7044):959–963. https://doi.org/10.1038/nature03659
Riccio O, van Gijn ME, Bezdek AC, Pellegrinet L, van Es JH, Zimber-Strobl U, Strobl LJ, Honjo T, Clevers H, Radtke F (2008) Loss of intestinal crypt progenitor cells owing to inactivation of both Notch1 and Notch2 is accompanied by derepression of CDK inhibitors p27Kip1 and p57Kip2. EMBO Rep 9(4):377–383. https://doi.org/10.1038/embor.2008.7
Fre S, Hannezo E, Sale S, Huyghe M, Lafkas D, Kissel H, Louvi A, Greve J, Louvard D, Artavanis-Tsakonas S (2011) Notch lineages and activity in intestinal stem cells determined by a new set of knock-in mice. PLoS One 6(10):e25785. https://doi.org/10.1371/journal.pone.0025785
Basak O, Beumer J, Wiebrands K, Seno H, van Oudenaarden A, Clevers H (2017) Induced quiescence of Lgr5+ stem cells in intestinal organoids enables differentiation of hormone-producing enteroendocrine cells. Cell Stem Cell 20(2):177–190 e174. https://doi.org/10.1016/j.stem.2016.11.001
Heuberger J, Kosel F, Qi J, Grossmann KS, Rajewsky K, Birchmeier W (2014) Shp2/MAPK signaling controls goblet/paneth cell fate decisions in the intestine. Proc Natl Acad Sci U S A 111(9):3472–3477. https://doi.org/10.1073/pnas.1309342111
Kober OI, Ahl D, Pin C, Holm L, Carding SR, Juge N (2014) Gammadelta T-cell-deficient mice show alterations in mucin expression, glycosylation, and goblet cells but maintain an intact mucus layer. Am J Physiol Gastrointest Liver Physiol 306(7):G582–G593. https://doi.org/10.1152/ajpgi.00218.2013
Yin X, Farin HF, van Es JH, Clevers H, Langer R, Karp JM (2014) Niche-independent high-purity cultures of Lgr5+ intestinal stem cells and their progeny. Nat Methods 11(1):106–112. https://doi.org/10.1038/nmeth.2737
Gracz AD, Samsa LA, Fordham MJ, Trotier DC, Zwarycz B, Lo YH, Bao K, Starmer J, Raab JR, Shroyer NF, Reinhardt RL, Magness ST (2018) Sox4 promotes Atoh1-independent intestinal secretory differentiation toward tuft and enteroendocrine fates. Gastroenterology 155(5):1508–1523. e1510. https://doi.org/10.1053/j.gastro.2018.07.023
de Lau W, Kujala P, Schneeberger K, Middendorp S, Li VS, Barker N, Martens A, Hofhuis F, DeKoter RP, Peters PJ, Nieuwenhuis E, Clevers H (2012) Peyer's patch M cells derived from Lgr5(+) stem cells require SpiB and are induced by RankL in cultured “miniguts”. Mol Cell Biol 32(18):3639–3647. https://doi.org/10.1128/MCB.00434-12
Cheng CW, Biton M, Haber AL, Gunduz N, Eng G, Gaynor LT, Tripathi S, Calibasi-Kocal G, Rickelt S, Butty VL, Moreno-Serrano M, Iqbal AM, Bauer-Rowe KE, Imada S, Ulutas MS, Mylonas C, Whary MT, Levine SS, Basbinar Y, Hynes RO, Mino-Kenudson M, Deshpande V, Boyer LA, Fox JG, Terranova C, Rai K, Piwnica-Worms H, Mihaylova MM, Regev A, Yilmaz OH (2019) Ketone body signaling mediates intestinal stem cell homeostasis and adaptation to diet. Cell 178(5):1115–1131 e1115. https://doi.org/10.1016/j.cell.2019.07.048
Lim JS, Ibaseta A, Fischer MM, Cancilla B, O'Young G, Cristea S, Luca VC, Yang D, Jahchan NS, Hamard C, Antoine M, Wislez M, Kong C, Cain J, Liu YW, Kapoun AM, Garcia KC, Hoey T, Murriel CL, Sage J (2017) Intratumoural heterogeneity generated by Notch signalling promotes small-cell lung cancer. Nature 545(7654):360–364. https://doi.org/10.1038/nature22323
Cheng CW, Yilmaz OH, Mihaylova MM (2020) Strategies for measuring induction of fatty acid oxidation in intestinal stem and progenitor cells. Methods Mol Biol 2171:53–64. https://doi.org/10.1007/978-1-0716-0747-3_4
Powell RH, Behnke MS (2017) WRN conditioned media is sufficient for in vitro propagation of intestinal organoids from large farm and small companion animals. Biol Open 6(5):698–705. https://doi.org/10.1242/bio.021717
Clinton J, McWilliams-Koeppen P (2019) Initiation, expansion, and cryopreservation of human primary tissue-derived normal and diseased organoids in embedded three-dimensional culture. Curr Protoc Cell Biol 82(1):e66. https://doi.org/10.1002/cpcb.66
Boonekamp KE, Dayton TL, Clevers H (2020) Intestinal organoids as tools for enriching and studying specific and rare cell types: advances and future directions. J Mol Cell Biol 12(8):562–568. https://doi.org/10.1093/jmcb/mjaa034
Schwank G, Andersson-Rolf A, Koo BK, Sasaki N, Clevers H (2013) Generation of BAC transgenic epithelial organoids. PLoS One 8(10):e76871. https://doi.org/10.1371/journal.pone.0076871
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Qiu, Y., Phanor, S.K., Pyo, S., Cheng, CW. (2023). Modeling Notch Activity and Lineage Decisions Using Intestinal Organoids. In: Ordóñez-Morán, P. (eds) Intestinal Differentiated Cells. Methods in Molecular Biology, vol 2650. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3076-1_10
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3076-1_10
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3075-4
Online ISBN: 978-1-0716-3076-1
eBook Packages: Springer Protocols