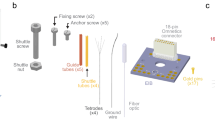
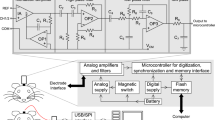
Abstract
Electroencephalography (EEG), an electrophysiological monitoring method to record the electrical activity of the brain, has been one of the most important noninvasive brain imaging tools in psychiatry, but surprisingly little is known about how the neural correlates of various EEG features are linked to cognition. Recent advances in neuroscience and related technologies make this an ideal time for new discoveries about the origin and significance of the contents of EEG. In particular, understanding the molecular and cellular mechanisms underlying diverse EEG features has been facilitated using mouse models under genetic, pharmacological, or optogenetic manipulations. A core challenge in mouse EEG was to obtain topographical neuroimaging that can be compared to human EEG. To overcome this challenge, we have developed a high-density EEG using a polyimide-based microarray that fits to the mouse skull and can be applied to various studies with a high spatiotemporal accuracy in free behavioral states. The benefits of mouse high-density EEG are not only that it provides cross-species neuroimaging data comparable to human EEG but also that it helps in dissecting enigmatic brain activity by probing the neural substrates of cognition when combined with optogenetics. The aim of this chapter is to introduce the methodological aspects of high-density EEG in mice. We explain the electrodes, surgery, recording, and analysis procedures and present applications in studying the origin of EEG signals. In addition, we point to potential areas where this technique will provide mechanical insight into circuit dysfunction in major psychiatric conditions.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Cohen MX (2017) Where does EEG come from and what does it mean? Trends Neurosci 40(4):208–218. https://doi.org/10.1016/j.tins.2017.02.004
Bragin A, Jando G, Nadasdy Z, Hetke J, Wise K, Buzsaki G (1995) Gamma (40-100 Hz) oscillation in the hippocampus of the behaving rat. J Neurosci 15(1 Pt 1):47–60
Buzsaki G, Wang XJ (2012) Mechanisms of gamma oscillations. Annu Rev Neurosci 35:203–225. https://doi.org/10.1146/annurev-neuro-062111-150444
O'Keefe J, Recce ML (1993) Phase relationship between hippocampal place units and the EEG theta rhythm. Hippocampus 3(3):317–330. https://doi.org/10.1002/hipo.450030307
Tort AB, Kramer MA, Thorn C, Gibson DJ, Kubota Y, Graybiel AM, Kopell NJ (2008) Dynamic cross-frequency couplings of local field potential oscillations in rat striatum and hippocampus during performance of a T-maze task. Proc Natl Acad Sci U S A 105(51):20517–20522. https://doi.org/10.1073/pnas.0810524105
Singer W (2013) Cortical dynamics revisited. Trends Cogn Sci 17(12):616–626. https://doi.org/10.1016/j.tics.2013.09.006
Choi JH, Koch KP, Poppendieck W, Lee M, Shin HS (2010) High resolution electroencephalography in freely moving mice. J Neurophysiol 104(3):1825–1834. https://doi.org/10.1152/jn.00188.2010
Lee M, Kim D, Shin HS, Sung HG, Choi JH (2011) High-density EEG recordings of the freely moving mice using polyimide-based microelectrode. J Vis Exp (47):2562. https://doi.org/10.3791/2562
Buzsaki G, Stark E, Berenyi A, Khodagholy D, Kipke DR, Yoon E, Wise KD (2015) Tools for probing local circuits: high-density silicon probes combined with optogenetics. Neuron 86(1):92–105. https://doi.org/10.1016/j.neuron.2015.01.028
Richardson RR Jr, Miller JA, Reichert WM (1993) Polyimides as biomaterials: preliminary biocompatibility testing. Biomaterials 14(8):627–635
Viventi J, Kim DH, Vigeland L, Frechette ES, Blanco JA, Kim YS, Avrin AE, Tiruvadi VR, Hwang SW, Vanleer AC, Wulsin DF, Davis K, Gelber CE, Palmer L, Van der Spiegel J, Wu J, ** brain activity in vivo. Nat Neurosci 14(12):1599–1605. https://doi.org/10.1038/nn.2973
Hwang E, Kim S, Han K, Choi JH (2012) Characterization of phase transition in the thalamocortical system during anesthesia-induced loss of consciousness. PLoS One 7(12):e50580. https://doi.org/10.1371/journal.pone.0050580
Bell AJ, Sejnowski TJ (1995) An information-maximization approach to blind separation and blind deconvolution. Neural Comput 7(6):1129–1159
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134(1):9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009
Ma Y, Smith D, Hof PR, Foerster B, Hamilton S, Blackband SJ, Yu M, Benveniste H (2008) In vivo 3D digital atlas database of the adult C57BL/6J mouse brain by magnetic resonance microscopy. Front Neuroanat 2:1. https://doi.org/10.3389/neuro.05.001.2008
Pinault D, O'Brien TJ (2005) Cellular and network mechanisms of genetically-determined absence seizures. Thalamus Relat Syst 3(3):181–203. https://doi.org/10.1017/S1472928807000209
Blumenfeld H (2005) Consciousness and epilepsy: why are patients with absence seizures absent? Prog Brain Res 150:271–286. https://doi.org/10.1016/S0079-6123(05)50020-7
Schomer DL, da Silva FL (2011) Niedermeyer’s electroencephalography: basic principles, clinical applications, and related fields, 6th edn. Lippincott Williams & Wilkins, Philadelphia
Pinault D, Leresche N, Charpier S, Deniau JM, Marescaux C, Vergnes M, Crunelli V (1998) Intracellular recordings in thalamic neurones during spontaneous spike and wave discharges in rats with absence epilepsy. J Physiol 509(Pt 2):449–456
Stam CJ (2005) Nonlinear dynamical analysis of EEG and MEG: review of an emerging field. Clin Neurophysiol 116(10):2266–2301. https://doi.org/10.1016/j.clinph.2005.06.011
Wong CH, Siah KW, Lo AW (2018) Estimation of clinical trial success rates and related parameters. Biostatistics. https://doi.org/10.1093/biostatistics/kxx069
Galambos R, Makeig S, Talmachoff PJ (1981) A 40-Hz auditory potential recorded from the human scalp. Proc Natl Acad Sci U S A 78(4):2643–2647
Conti G, Santarelli R, Grassi C, Ottaviani F, Azzena GB (1999) Auditory steady-state responses to click trains from the rat temporal cortex. Clin Neurophysiol 110(1):62–70
Kwon JS, O'Donnell BF, Wallenstein GV, Greene RW, Hirayasu Y, Nestor PG, Hasselmo ME, Potts GF, Shenton ME, McCarley RW (1999) Gamma frequency-range abnormalities to auditory stimulation in schizophrenia. Arch Gen Psychiatry 56(11):1001–1005
Spencer KM, Salisbury DF, Shenton ME, McCarley RW (2008) Gamma-band auditory steady-state responses are impaired in first episode psychosis. Biol Psychiatry 64(5):369–375. https://doi.org/10.1016/j.biopsych.2008.02.021
Thune H, Recasens M, Uhlhaas PJ (2016) The 40-Hz auditory steady-state response in patients with schizophrenia: a meta-analysis. JAMA Psychiat 73(11):1145–1153. https://doi.org/10.1001/jamapsychiatry.2016.2619
Schizophrenia Working Group of the Psychiatric Genomics C (2014) Biological insights from 108 schizophrenia-associated genetic loci. Nature 511(7510):421–427. https://doi.org/10.1038/nature13595
van Os J, Rutten BP, Poulton R (2008) Gene-environment interactions in schizophrenia: review of epidemiological findings and future directions. Schizophr Bull 34(6):1066–1082. https://doi.org/10.1093/schbul/sbn117
Geyer MA, McIlwain KL, Paylor R (2002) Mouse genetic models for prepulse inhibition: an early review. Mol Psychiatry 7(10):1039–1053. https://doi.org/10.1038/sj.mp.4001159
Powell CM, Miyakawa T (2006) Schizophrenia-relevant behavioral testing in rodent models: a uniquely human disorder? Biol Psychiatry 59(12):1198–1207. https://doi.org/10.1016/j.biopsych.2006.05.008
Wilson CA, Koenig JI (2014) Social interaction and social withdrawal in rodents as readouts for investigating the negative symptoms of schizophrenia. Eur Neuropsychopharmacol 24(5):759–773. https://doi.org/10.1016/j.euroneuro.2013.11.008
Brenner CA, Krishnan GP, Vohs JL, Ahn WY, Hetrick WP, Morzorati SL, O'Donnell BF (2009) Steady state responses: electrophysiological assessment of sensory function in schizophrenia. Schizophr Bull 35(6):1065–1077. https://doi.org/10.1093/schbul/sbp091
Sohal VS, Zhang F, Yizhar O, Deisseroth K (2009) Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 459(7247):698–702. https://doi.org/10.1038/nature07991
Cardin JA, Carlen M, Meletis K, Knoblich U, Zhang F, Deisseroth K, Tsai LH, Moore CI (2009) Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459(7247):663–667. https://doi.org/10.1038/nature08002
Shahriari Y, Krusienski D, Dadi YS, Seo M, Shin HS, Choi JH (2016) Impaired auditory evoked potentials and oscillations in frontal and auditory cortex of a schizophrenia mouse model. World J Biol Psychiatry 17(6):439–448. https://doi.org/10.3109/15622975.2015.1112036
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Choi, J.H., Hwang, E. (2022). High-Density Electroencephalography in Freely Moving Mice. In: Vertes, R.P., Allen, T. (eds) Electrophysiological Recording Techniques. Neuromethods, vol 192. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2631-3_1
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2631-3_1
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2630-6
Online ISBN: 978-1-0716-2631-3
eBook Packages: Springer Protocols