Abstract
Coagulation factors, as factor VII, VIII, and IX, are complex proteins which are very difficult to express. Blood coagulation factor IX is a vitamin K-dependent protein, and it has become a valuable biopharmaceutical in the treatment of hemophilia B. Here, we describe the techniques used to generate human cell lines producing human recombinant factor IX, as an example of complex protein, as well as in vitro characterization of this coagulation factor.
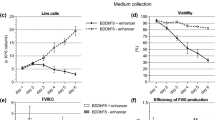
To produce the FIX human adherent 293T SK-Hep-1 cells were used and stably modified by a lentiviral vector carrying the hFIX and the eGFP genes. The eGFP was employed as a reporter protein.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kumar SR (2015) Industrial production of clotting factors: challenges of expression, and choice of host cells. Biotechnol J 10:995–1004
Schmidt AE, Bajaj SP (2003) Structure-function relationships in factor IX and factor IXa. Trends Cardiovasc Med 13:39–45
Hallgren KW, Hommema EL, McNally BA, Berkner KL (2002) Carboxylase overexpression effects full carboxylation but poor release and secretion of factor IX: implications for the release of vitamin K-dependent proteins. Biochemistry 41:15045–15055
Kaufman RJ, Wasley LC, Furie BC et al (1986) Expression, purification, and characterization of recombinant gamma-carboxylated factor IX synthesized in Chinese hamster ovary cells. J Biol Chem 261:9622–9628
Busby S, Kumar A, Joseph M et al (1985) Expression of active human factor IX in transfected cells. Nature 316:271–273
Messier TL, Pittman DD, Long GL et al (1991) Cloning and expression in COS-1 cells of a full-length cDNA encoding human coagulation factor X. Gene 99:291–294
Camire RM, Larson PJ, Stafford DW, High KA (2000) Enhanced gamma-carboxylation of recombinant factor X using a chimeric construct containing the prothrombin propeptide. Biochemistry 39:14322–14329
de Sousa Bomfim A, Cristina Corrêa de Freitas M, Picanço-Castro V et al (2016) Human cell lines: a promising alternative for recombinant FIX production. Protein Expr Purif 121:149–156
do Amaral RLF, de Sousa Bomfim A, de Abreu-Neto MS et al (2016) Approaches for recombinant human factor IX production in serum-free suspension cultures. Biotechnol Lett 38:385–394
Swiech K, de Freitas MCC, Covas DT, Picanço-Castro V (2015) Recombinant glycoprotein production in human cell lines. Methods Mol Biol 1258:223–240
Spencer HT, Denning G, Gautney RE et al (2011) Lentiviral vector platform for production of bioengineered recombinant coagulation factor VIII. Mol Ther 19:302–309
Bubsy S et al (1985) Expression of active human factor IX in transfected cells. Nature 316:271–273
Bradford MMA (1976) Rapid and sensitive method for the quantitation microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Neuhoff V, Arold R, Taube P, Ehrhardt W (1988) Improved staining of proteins in polyacrylamide gels including isoelectric focusing gels with clear background at nanogram sensitivity using Coomassie Brilliant Blue G-250 and R-250. Electrophoresis 9:255–262
Acknowledgments
The authors thank the National Council for Scientific and Technological Development (CNPq) and the Coordination for the Improvement of Higher Educational Personnel (CAPES) for funding the research.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
de Sousa Bomfim, A., Archangelo, B.S., Pereira, A.S., de Sousa Russo, E.M. (2022). Recombinant Expression of Complex Proteins in Human Cell Lines. In: Garcia Fruitós, E., Arís Giralt, A. (eds) Insoluble Proteins. Methods in Molecular Biology, vol 2406. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1859-2_19
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1859-2_19
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1858-5
Online ISBN: 978-1-0716-1859-2
eBook Packages: Springer Protocols