Summary
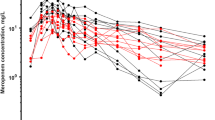
This study retrospectively evaluated the predictive performance of a 1-compartment Bayesian forecasting program in adult intensive care unit (ICU) patients with stable renal function. A comparison was made of the reliability of 3 sets of population-based parameter estimates and 2 serum concentration monitoring strategies. A larger mean error for prediction of peak gentamicin concentrations was seen with literature-derived parameters than when ICU population-based parameter estimates were used. Bias and precision improved when non-steady-state peak and trough concentrations were used to predict those at steady-state; the addition of steady-state values did not provide additional information for predictions once non-steady-state feedback concentrations were incorporated. The addition of 4 serial gentamicin concentrations obtained at both non-steady-state and steady-state did not noticeably improve the predictive performance.
The results demonstrate that initial ICU pharmacokinetic parameter estimates for a 1-compartment Bayesian model provide accurate prediction of steady-state gentamicin concentrations. Prediction bias and precision showed the greatest improvement when non-steady-state gentamicin concentrations were used to determine individualised pharmacokinetic parameters.
Similar content being viewed by others
References
Bodenham A, Shelly MP, Park GR. The altered pharmacokinetics and pharmacodynamics of drugs commonly used in critically ill patients. Clinical Pharmacokinetics 14: 347–373, 1988
Burton ME, Chow MSS, Platt DR, Day RB, Brater DC, et al. Accuracy of Bayesian and Sawchuk-Zaske dosing methods for gentamicin. Clinical Pharmacy 5: 143–149, 1986
Chelluri L, Jastremski MS. Inadequacy of standard aminoglycoside loading doses in acutely ill patients. Critical Care Medicine 15: 1143–1145, 1987
Chelluri L, Warren J, Jastremski MS. Pharmacokinetics of a 3 mg/kg body weight loading dose of gentamicin or tobramycin in critically ill patients. Chest 95: 1295–1297, 1989
Cockcroft DW, Gault MH. Prediction of Creatinine clearance from serum Creatinine. Nephron 16: 31–41, 1976
Dasla JF. Drug use in a surgical intensive care unit. Drug Intelligence and Clinical Pharmacy 20: 752–756, 1986
Datas JF, Armstrong DK. Variability in aminoglycoside pharmacokinetics in critically ill surgical patients. Critical Care Medicine 16: 327–330, 1988
Hassan E, Ober JD. Predicted and measured aminoglycoside pharmacokinetic parameters in critically ill patients. Antimicrobial Agents and Chemotherapy 31: 1855–1858, 1987
Kreger BE, Craven DE, McCabe WR. Gram-negative bacteremia: re-evaluation of clinical features and treatment in 612 patients. American Journal of Medicine 68: 344–355, 1980
Mann HJ, Fuhs DW, Awang R, Ndemo FA, Cerra FB. Altered aminoglycoside pharmacokinetics in critically ill patients with sepsis. Clinical Pharmacy 6: 148–153, 1987
Moore RD, Smith CR, Lielman PS. The association of aminoglycoside plasma levels with mortality in patients with Gramnegative bacteremia. Journal of Infectious Disease 149: 443–448, 1984
Niemiec PW, Allo MD, Miller CF. Effect of altered volume of distribution on aminoglcoside levels in patients in surgical intensive care. Archives of Surgery 122: 207–212, 1987
Noone P, Parsons TMC, Pattison JR, Slack RCB, Garfield-Davies D, et al. Experience in monitoring gentamicin therapy during serious Gram-negative sepsis. British Medical Journal 1: 477–481, 1974
Pryka RD, Rodvold KA, Garrison M, Rotschafer JC. Individualizing vancomycin dosage regimens: one- vs two-compartment Bayesian models. Therapeutic Drug Monitoring 11: 450–454, 1989
Rodvold KA, Blum RA. Predictive performance of Sawchuk-Zaske and Bayesian dosing methods for tobramycin. Journal of Clinical Pharmacology 27: 419–427, 1987
Rodvold KA, Pryka RD, Garrison M, Rotschafer JC. Evaluation of a two compartment Bayesian forecasting program for predicting vancomycin concentrations. Therapeutic Drug Monitoring 11: 269–275, 1989
Rodvold KA, Zokufa H, Rotschafer JC. Aminoglycoside pharmacokinetic monitoring: an integral part of patient care? Clinical Pharmacy 7: 608–613, 1988
Sawchuk RJ, Zaske DE. Pharmacokinetics of dosing regimens which utilize multiple intravenous infusions: gentamicin in burn patients. Journal of Pharmacokinetics and Biopharmaceutics 4: 183–195, 1976
Sheiner LB, Beal SL, Rosenberg B, Marathe VV. Forecasting individual pharmacokinetics. Clinical Pharmacology and Therapeutics 26: 294–305, 1979
Sheiner LB, Beal SL. Some suggestions for measuring predictive performance. Journal of Pharmacokinetics and Biopharmaceutics 9: 503–512, 1981
Schumacher GE, Barr JT. Bayesian approaches in pharmacokinetic decision making. Clinical Pharmacy 3: 525–530, 1984
Sultan E, Richard C, Pezzano M, Auzepy P, Singlas E. Pharmacokinetics of Pefloxacin and amikacin administered simultaneously to intensive care patients. European Journal of Clinical Pharmacology 34: 637–643, 1988
Summer WR, Michael JR, Lipsky JJ. Initial aminoglycoside levels in the critically ill. Critical Care Medicine 11: 948–950, 1983
Townsend PL, Fink MP, Stein KL, Murphy SG. Aminoglycoside pharmacokinetics: dosage requirements and nephrotoxicity in trauma patients. Critical Care Medicine 17: 154–157, 1989
Vožeh S, Uematsu T, Hauf GF, Follath F. Performance of Bayesian feedback to forecast lidocaine serum concentration: evaluation of the prediction error and the perdition interval. Journal of Pharmacokinetics and Biopharmaceutics 13: 203–211, 1985
Zaske DE. Aminoglycosides. In Evans et al. (Eds) Applied Pharmacokinetic Principles of Therapeutic Drug Monitoring. 2nd ed., pp. 331–381, Applied Therapeutics. Spokane. Washington, 1986
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rodvold, K.A., Pryka, R.D., Kuehl, P.G. et al. Bayesian Forecasting of Serum Gentamicin Concentrations in Intensive Care Patients. Clin Pharmacokinet 18, 409–418 (1990). https://doi.org/10.2165/00003088-199018050-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199018050-00005