Abstract
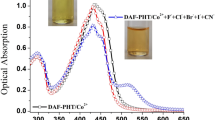
This paper reports that changing metal ions (Co2+, Ni2+, Cu2+, or Cd2+) in the 2-(2-thiazolylazo)-P-cresol (TAC)-metal ensemble may generate varied optical responses to anions. The TAC and Ni2+ ensemble can detect and quantify CN- in a highly sensitive and selective manner. CN- competes for the Ni2+ present in the ensemble during recognition events, which thereby triggers colorimetric and absorption spectral changes. CN- and S2- decomposed TAC-Cu2+ ensemble by forming [Cu(CN)X]n-species and CuS, respectively; however, the discrimination of CN- and S2- was not achieved. The TAC-Co2+ ensemble exhibited discriminated interaction with CN- through the absorption channel, but CN- was not quantitatively determined. Although the TAC-Cd2+ ensemble responded to different anions, it did not recognize each anion selectively. These results demonstrated that metal ions can powerfully modulate anion identification to some extent, which can be an effective strategy to achieve selectivity of certain anions by varying the metal ions in the ensemble.
Similar content being viewed by others
References
R. Martínez-Máñez and F. Sancenón, Chem. Rev., 2003, 103, 4419.
M. E. Moragues, R. Martínez-Máñez, and F. Sancenón, Chem. Soc. Rev., 2011, 40, 2593.
H. Khajehsharifi and M. M. Bordbar, Sens. Actuators, B, 2015, 209, 1015.
P. Singh, H. Singh, G. Bhargava, and S. Kumar, J. Mater. Chem. C, 2015, 3, 5524.
X. Li, X. Gao, W. Shi, and H. Ma, Chem. Rev., 2014, 114, 590.
P. A. Gale and C. Caltagirone, Chem. Soc. Rev., 2015, 44, 4212.
V. Amendola, L. Fabbrizzi, and L. Mosca, Chem. Soc. Rev., 2010, 39, 3889.
M. Chhatwal, A. Kumar, V. Singh, R. D. Gupta, and S. K. Awasthi, Coord. Chem. Rev., 2015, 292, 30.
J. Chen, Y. Li, W. Zhong, Q. Hou, H. Wang, X. Sun, and P. Yi, Sens. Actuators, B, 2015, 206, 230.
Q. Zhou, Y. Zhu, P. Sheng, Z. Wu, and Q. Cai, RSC Adv., 2014, 4, 46951.
A. K. Mahapatra, S. Mondal, S. K. Manna, K. Maiti, R. Maji, Md. R. Uddin, S. Mandal, D. Sarkar, T. K. Mondal, and D. K. Maiti, Dalton Trans., 2015, 44, 6490.
L. Tang, M. Cai, Z. Huang, K. Zhong, S. Hou, Y. Bian, and R. Nandhakumar, Sens. Actuators, B, 2013, 185, 188.
L. Tang, P. Zhou, Q. Zhang, Z. Huang, J. Zhao, and M. Cai, Inorg. Chem. Commun., 2013, 36, 100.
K. Ren, X. Shang, J. Fu, P. Zhao, and J. Zhang, Polyhedron, 2016, 104, 99.
A. Ojida, T. Sakamoto, M. Inoue, S. Fujishima, G. Lippens, and I. Hamachi, J. Am. Chem. Soc., 2009, 131, 6543.
S. Lee, K. K. Y. Yuen, K. A. Jolliffe, and J. Yoon, Chem. Soc. Rev., 2015, 44, 1749.
R. K. Pathak, J. Dessingou, and C. P. Rao, Anal. Chem., 2012, 84, 8294.
J. Wang and C. S. Ha, Tetrahedron, 2010, 66, 1846.
J. Wang and C. S. Ha, Analyst, 2010, 135, 1214.
A. Hens, P. Mondal, and K. K. Rajak, Polyhedron, 2015, 85, 255.
S. Biswas, A. K. Pramanik, and T. K. Mondal, J. Mol. Struct., 2015, 1088, 28.
V. A. Lemos, V. J. Ferreira, J. A. Barreto, and L. A. Meira, Water, Air, Soil Pollut., 2015, 226, 1.
V. A. Lemos, E. S. Santos, M. S. Santos, and R. T. Yamaki, Microchim. Acta, 2007, 158, 189.
X. J. Zhao and C. Z. Huang, TrAC, Trends Anal. Chem., 2010, 29, 354.
Z. Xu, X. Chen, H. N. Kim, and J. Yoon, Chem. Soc. Rev., 2010, 39, 127.
T. D. Ashton, K. A. Jolliffe, and F. M. Pfeffer, Chem. Soc. Rev., 2015, 44, 4547.
X. Cao, W. Lin, and L. He, Org. Lett., 2011, 13, 4716.
R. G. Upendar, D. Priyadip, S. Sukdeb, B. Mithu, K. G. Sudip, and D. Amitava, Chem. Commun., 2013, 49, 255.
W. Hao, A. McBride, S. McBride, J. P. Gao, and Z. Y. Wang, J. Mater. Chem., 2011, 21, 1040.
X. Lou, J. Qin, and Z. Li, Analyst, 2009, 134, 2071.
K. Sasakura, K. Hanaoka, N. Shibuya, Y. Mikami, Y. Kimura, T. Komatsu, T. Ueno, T. Terai, H. Kimura, and T. Naganot, J. Am. Chem. Soc., 2011, 133, 18003.
Y. Hao, W. Chen, L. Wang, X. Zhu, Y. Zhang, P. Qu, L. Liu, B. Zhou, Y. N. Liu, and M. Xu, Talanta, 2015, 143, 307.
Q. Zhou, Y. Zhu, P. Sheng, Z. Wu, and Q. Cai, RSC Adv., 2014, 4, 46951.
T. Momeni-Isfahani and A. Niazi, Spectrochim. Acta, Part A, 2014, 120, 630.
W. I. White and J. I. Legg, J. Am. Chem. Soc., 1975, 97, 3937.
A. Mirmohseni and A. Alipour, Sens. Actuators, B, 2002, 84, 245.
F. Meyer, R. F. Winter, and E. Kaifer, Inorg. Chem., 2001, 40, 4597.
A. Zheng, D. A. Dzombak, and R. G. Luthy, Environ. Sci. Technol., 2003, 37, 107.
A. K. Singh, P. K. Yadav, N. Kumari, R. Nagarajan, and L. Mishra, J. Mater. Chem. C, 2015, 3, 12123.
Y. Fu, Q. C. Feng, X. J. Jiang, H. Xu, M. Li, and S. Q. Zang, Dalton Trans., 2014, 43, 5815.
Acknowledgments
H. B. L. gratefully acknowledges financial support from the NSFC (Grant Nos. 31360020 and 31560014), GXNSFC (Grant No. 2014GXNSFCA118003), the Scientific Research Foundation of Guangxi University (Grant No. XGZ130080) and Hundred-Talent Program (Guangxi Province). J. W. thanks the NSFC (Grant No. 21567002), GXNSFC (Grant No. 2015GXNSFCA139005), as well as the Scientific Research Foundation of Guangxi University (Grant No. XGZ130081).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, J., Lan, B. & Liu, HB. Chemosensing Ensembles of 2-(2-Thiazolylazo)-p-cresol with Metal Ions in Colorimetric Detection of Anions. ANAL. SCI. 33, 677–682 (2017). https://doi.org/10.2116/analsci.33.677
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.33.677